Bo ZHANG, Ning ZHANG, Jianhua YANG, Jiancheng LAN, Jinqu WANG. High Performance a&b Oriented T Zeolite Membrane by a Two-stage Crystallization Synthesis [J]. Journal of Inorganic Materials, 2020, 35(8): 939
Search by keywords or author
- Journal of Inorganic Materials
- Vol. 35, Issue 8, 939 (2020)

1. SEM images of α -Al2O3 support photographed from surface (a) and cross section (b), large seeds (c), small seeds (d), modified by large seeds photographed from surface (e) and cross section (f), and the small seed layer photographed from surface (g) and cross section (h)
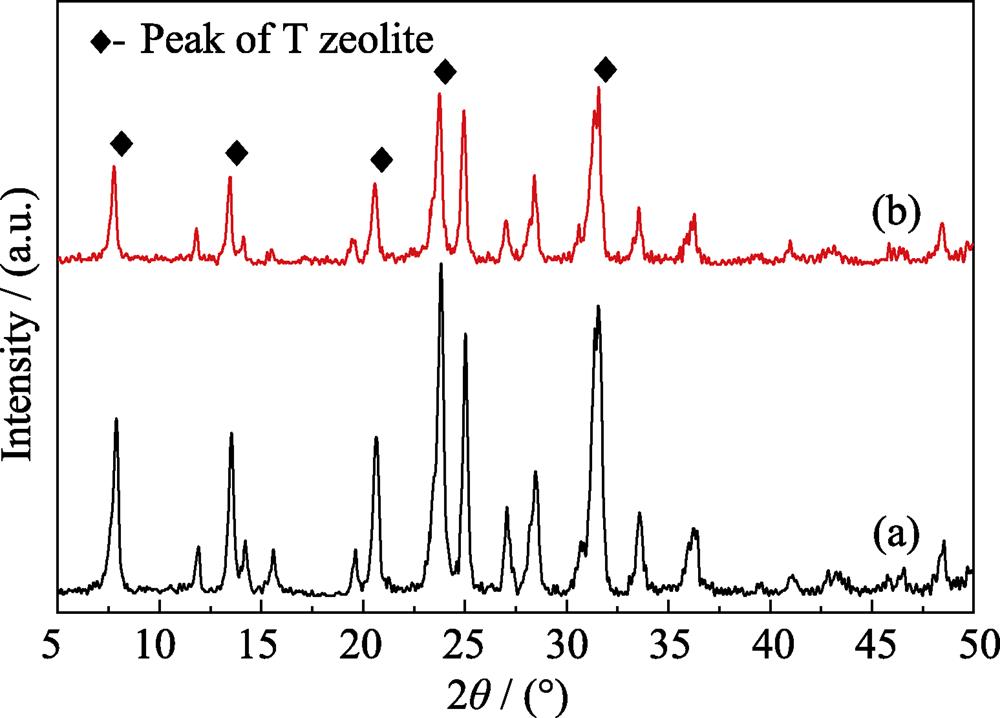
2. XRD patterns of large seeds (a) and small seeds (b)
3. SEM images of T-type zeolite membranes after first-step crystallization prepared at different first-step crystallization temperatures
4. SEM images of T-type zeolite membranes after two-step crystallizations prepared at different first-step crystallization temperature
5. XRD patterns of zeolite T membrane M4 (a) and small zeolite T seeds (b)
6. SEM images of T-type zeolite membranes after first-step crystallization prepared with varying first-step crystallization time
7. SEM images of T-type zeolite membranes after two-step crystallizations prepared with different first-step crystallization time
8. SEM images of T-type zeolite membranes after two-step crystallizations prepared at different second-step crystallization temperatures
| ||||||||||||||||||||||||||||||||||||||||||||||
Table 1. Pervaporation performance of T zeolite membranes prepared under different first-step crystallization temperatures
| ||||||||||||||||||||||||||||||||||||||||||||||
Table 2. Pervaporation performance of T zeolite membranes prepared with different first-step crystallization time
| ||||||||||||||||||||||||||||||||||||||||||||||
Table 3. Pervaporation performance of T zeolite membranes prepared under different second-step crystallization temperatures
|
Table 4. Pervaporation performance of zeolite T membranes for 90wt% isopropanol/water mixture at 75 ℃

Set citation alerts for the article
Please enter your email address



