Li LI, Xiaojie GUO, Yang JIN, Chaogui CHEN, M Asiri Abdullah, M Marwani Hadi, Qingzhou ZHAO, Guodong SHENG. Distinguished Cd(II) Capture with Rapid and Superior Ability using Porous Hexagonal Boron Nitride: Kinetic and Thermodynamic Aspects [J]. Journal of Inorganic Materials, 2020, 35(3): 284
Search by keywords or author
- Journal of Inorganic Materials
- Vol. 35, Issue 3, 284 (2020)
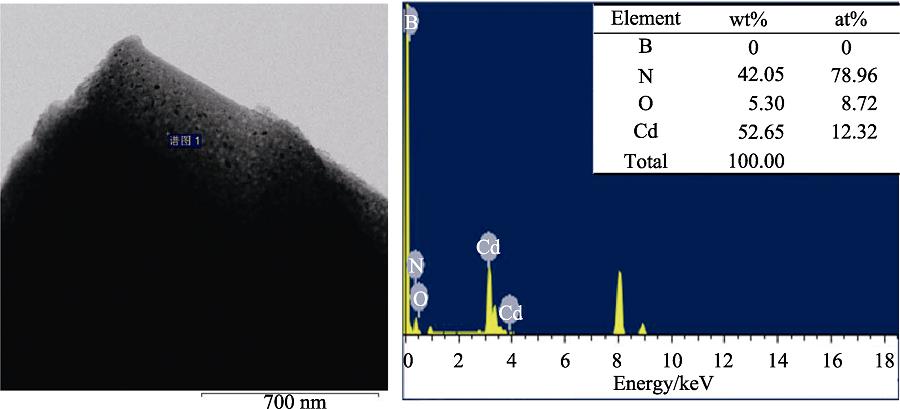
. Energy spectrum analysis (ESA) for unidentified black dots adsorbed on p-BN

. (A) SEM and (B) HRTEM images of p-BN, (C) SEM and (D) HRTEM images of p-BN after adsorption, (E) EDS analysis and (F) high-magnification HRTEM image of p-BN after adsorption
. XRD patterns (A) and FT-IR spectra (B) of p-BN before and after adsorption
. BET measurement of p-BN
. (A) Effect of initial pH on Cd(II) adsorption capacity (q e) and adsorption percentage at equilibrium, and (B) effect of p-BN dosage on the adsorption capacity (q e) and adsorption percentage of Cd(II)
. Variation of distribution of Cd(II) hydrolyzate species with pH
. (A) Adsorption capacities of Cd(II) with various contact times at different initial concentrations of Cd(II), and (B) adsorption percentages of Cd(II) on p-BN with various contact time at different initial concentrations of Cd(II)
. Kinetics models for adsorption of Cd(II) on p-BN
. (A) Adsorption isotherms of Cd(II) on p-BN at T =303, 313 and 323 K, equilibrium adsorption isotherms fitted by (B) Langmuir model, (C) Freundlich model, (D) Tempkin model
. Linear plots of lnK dversus 1/T for Cd(II) adsorption on p-BN adsorbent
. (A) XPS surveys for p-BN and adsorbed p-BN(inset: high resolution Cd3d XPS spectrum and background); (B) Experimental bonding enerygy peaks of Cd(II) and the comparisons of primary peaks of Cd3d5/2 and Cd3d3/2 for free Cd(II), CdCO3, Cd(OH)2
. High resolution spectra of B1s (a) and O1s (b) for p-BN before and after adsorption
| ||||||||||||||||||||||||||||||||||||||||||
Table 1.
Adsorption kinetics models parameters
| |||||||||||||||||||||||||||||||||||||||||||||||||||
Table 1.
Adsorption isotherm models parameters of Cd(II) on p-BN
| |||||||||||||||||||||||||||||||||||||||||
Table 2.
Values of thermodynamic parameters for the adsorption of Cd(II) on p-BN

Set citation alerts for the article
Please enter your email address



