Shuguang Yao, Yunhui Dong, Xianglong Li, Minhan Xie. Causes of Defects in Selective Laser Melting of AlSi10Mg[J]. Chinese Journal of Lasers, 2024, 51(16): 1602307
Search by keywords or author
- Chinese Journal of Lasers
- Vol. 51, Issue 16, 1602307 (2024)
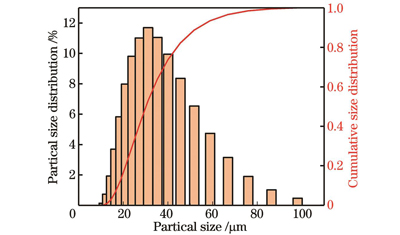
Fig. 1. Particle size distribution of AlSi10Mg alloy powder

Fig. 2. FS271M laser selective melting (SLM) equipment
Fig. 3. Powder bed and forming melt channel
Fig. 4. Formed melt channels and their microscopic morphologies
Fig. 5. Change of melt channel width with scanning speed
Fig. 6. SLM forming. (a) SLM forming physical process; (b) mesoscopic numerical model of SLM process
Fig. 7. Profile view of laser action
Fig. 8. Thermal conductivity, specific heat capacity, dynamic viscosity, and density as a function of temperature for AlSi10Mg
Fig. 9. Comparison of single-channel appearances under 100 W power and different scanning speeds
Fig. 10. Microscopic appearance of single-channel formed under 100 W laser power and different scanning speeds
Fig. 11. Comparison of small particle spheroidization (left: particle sputtering area; right: no sputtering area)
Fig. 12. Microscopic appearance of double-channel (left: 800 mm/s; right: 1000 mm/s)
Fig. 13. Temperature evolution diagram at a point in the powder bed
Fig. 14. Section of molten pool at 500 mm/s scanning speed
Fig. 15. Timing diagrams of single-channel forming section at 200 mm/s scanning speed
Fig. 16. Surface and section images of single-channel at 200 mm/s scanning speed
Fig. 17. Single-channel appearance at different preheating temperatures
Fig. 18. Variation of molten pool sizes at different preheating temperatures
|
Table 1. Chemical composition of AlSi10Mg alloy powder
|
Table 2. Melt channel forming parameters
|
Table 3. Thermophysical properties of materials

Set citation alerts for the article
Please enter your email address



