Dengfeng Li, Yachao Zhang, Chao Liu, Jiangbo Chen, Dong Sun, Lidai Wang, "Review of photoacoustic imaging for microrobots tracking in vivo [Invited]," Chin. Opt. Lett. 19, 111701 (2021)
Search by keywords or author
- Chinese Optics Letters
- Vol. 19, Issue 11, 111701 (2021)
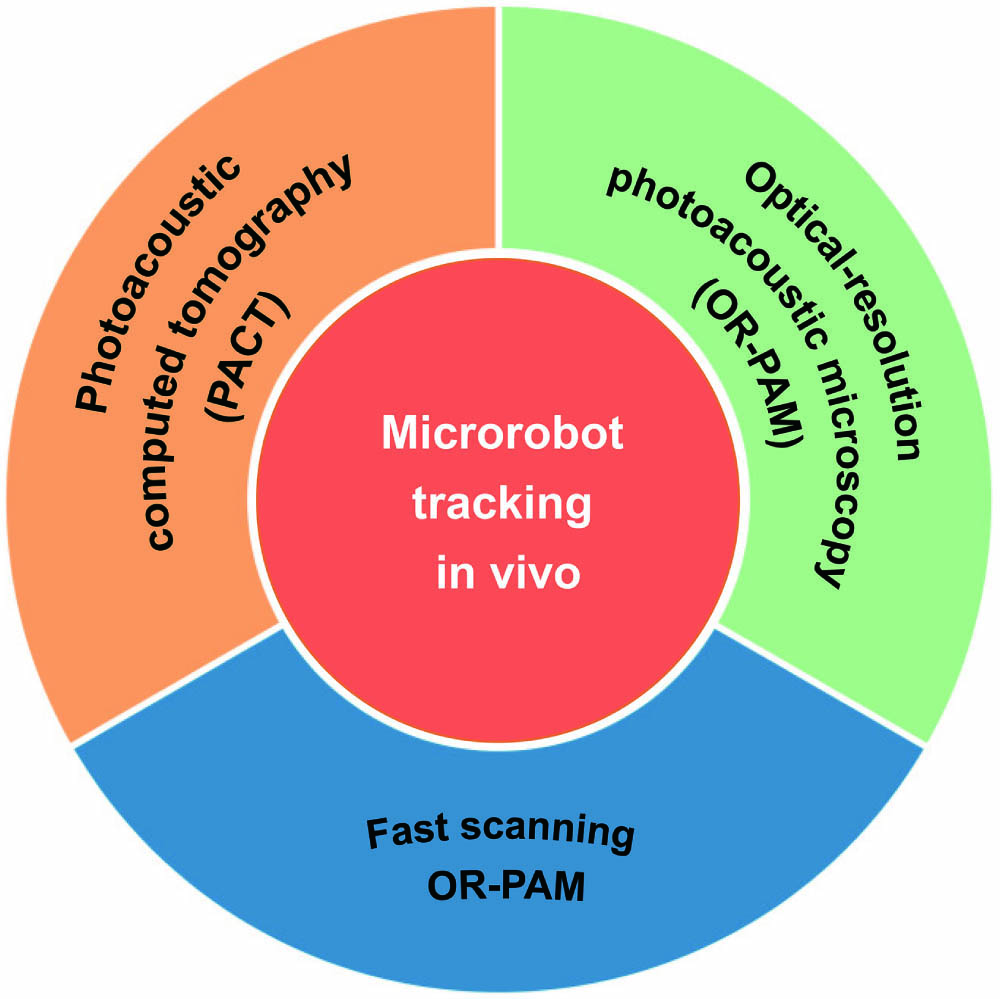
Fig. 1. Schematic summary of microrobots tracking in vivo based on PAI.
![In vivo PAI from organelles to organism. (a) A typical tubular-shaped mitochondrion in mouse embryonic fibroblasts[28]. (b) Capillary beds and RBCs in mouse ears[29]. (c) Heartbeat-encoded arterial network mapping of a 27-year-old healthy female volunteer’s breast cross-sectional image[30]. (d) Complete maximum intensity projection (MIP) PA image of the hand[31].](/richHtml/col/2021/19/11/111701/img_002.jpg)
Fig. 2. In vivo PAI from organelles to organism. (a) A typical tubular-shaped mitochondrion in mouse embryonic fibroblasts[28]. (b) Capillary beds and RBCs in mouse ears[29]. (c) Heartbeat-encoded arterial network mapping of a 27-year-old healthy female volunteer’s breast cross-sectional image[30]. (d) Complete maximum intensity projection (MIP) PA image of the hand[31].
Fig. 3. Multi-contrast and multi-parameter PAI. (a) Labeled tumor and surrounding vascular vessels[33]. (b) Oxygen saturation (sO2) in mouse ear[29]. (c) BF in mouse ears[34]. (d) Labeled lymphatic and blood vessels[35].
Fig. 4. PAI-trackable magnetic microswimmers. Reproduced with permission from Ref. [42].
Fig. 5. In vivo US/PACT imaging of the liver and kidney. (a) US image, bipolar PA image, and unipolar PA image at the cross section of the liver (P1) and kidney (P2); arrow 1 denotes the tracked and recorded artery blood vessel position. (b) Respiration motion time interval estimation according to the correlation coefficient calculation. (c) PA amplitude changing as a function of time at the position marked by an arrow [P1 in (a)] and its corresponding spectral analysis. Reproduced with permission from Ref. [60].
Fig. 6. Microrobotic system guided by PACT in vivo. (a) Microrobots in the mouse with PA excitation and generation. (b) PACT imaging setup. (c) Enteric coating protection in the stomach. (d) CW NIR laser irradiation for micromotor release in the intestine. (e) Microrobots propulsion to the therapy region. (f) Dynamics evaluation of PACT-guided microrobots movement in the intestine. (g) Movement displacement caused by the microrobots migration. (h) Movement displacement caused by respiration. (i) Speed comparison of microrobots migration and the respiration-induced movement. Reproduced with permission from Ref. [70].
Fig. 7. Microrobots imaging and navigation using developed PACT/US dual-modal system. (a) Comparison of PA signal amplitude in the left lateral lobe with and without injection microrobots clusters. The microrobots were injected through the portal vein, and the PA images were overlayed on the US images. (b) PA/US imaging guiding microrobots in the inferior vena cava of the mouse. Reproduced with permission from Ref. [23].
Fig. 8. PAI of a single microrobot in blood. (a) Optical resolution PA system. (b) High-resolution PA tracking of a single micro-rocket in the blood vessel model. Reproduced with permission from Ref. [22].
Fig. 9. Voice-coil-driven fast-scanning OR-PAM. (a) Schematic of the system. (b) Snapshots of single RBCs releasing oxygen in a mouse brain. (c) sO2 images without continuous visual stimulation. (d) sO2 images with 1 Hz continuous optical flashing stimulations on the left mouse eye. Reproduced with permission from Ref. [83].
Fig. 10. Polygon-scanning fast OR-PAM. (a) Schematic of the system. (b) sO2 image of the mouse ear; C-scan time is ∼5 s. (c) Flowing iron particle tracking. Reproduced with permission from Ref. [85].
Fig. 11. Single-breath-hold PACT system for breast cancer diagnosis and screening. (a) Perspective cut-away view of patient bed and optical components of the system. (b) Data acquisition components of the system. Four sets of 128-channel data acquisition systems were mapping a custom-built 512-element ring-array transducer. Reproduced with permission from Ref. [30].
|
Table 1. Scanning Speed and Range of Fast-Scanning PAM Systems
|
Table 2. Summary of PAI Systems in the Review for Microrobots Tracking

Set citation alerts for the article
Please enter your email address



