Ying-Jin Cheng, Chao-Fei Yang, Gang Xue, Tao Wang, Lei Zhang, Mei-E Li. Investigation of interaction between α-Fe metal and H atom by ab-initio method [J]. Acta Physica Sinica, 2020, 69(5): 053101-1
Search by keywords or author
- Acta Physica Sinica
- Vol. 69, Issue 5, 053101-1 (2020)
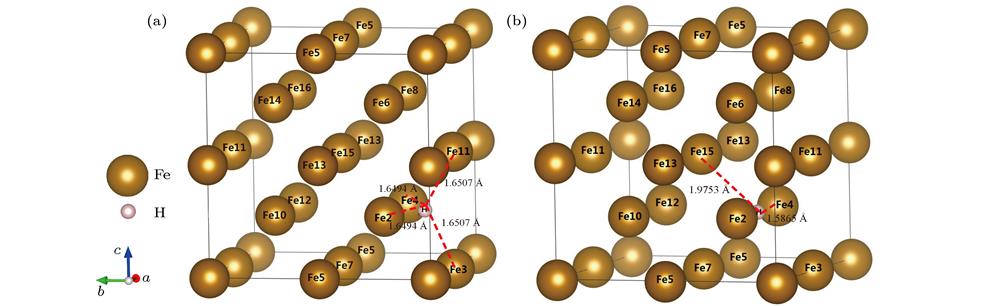
Fig. 1. Crystalline structure of α-Fe+H: (a) T-site; (b) O-site.
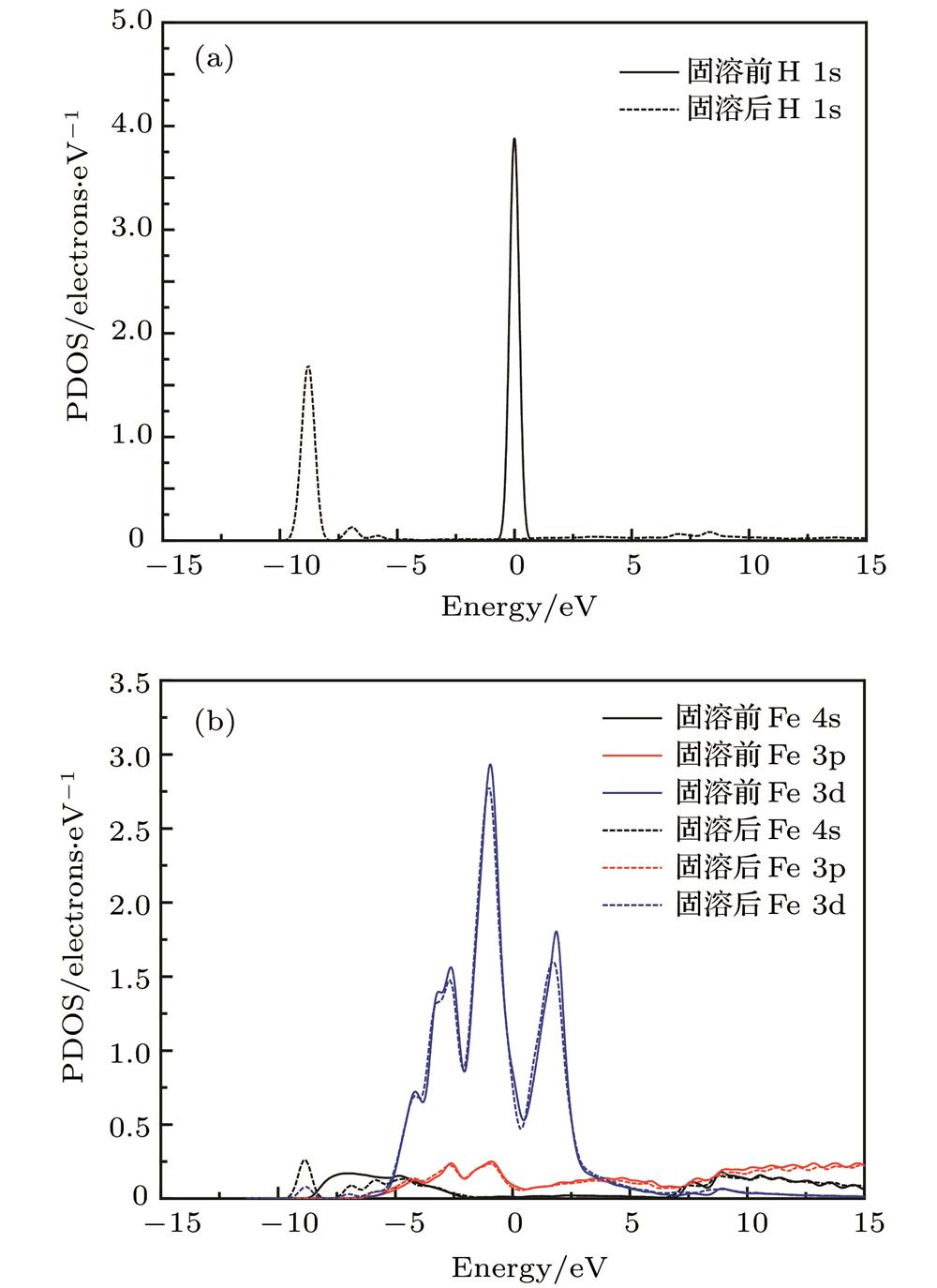
Fig. 2. Partial electronic density of state of α -Fe+H (T-site): (a) Free H atom and interstitial H atom; (b) Fe atom in perfect α -Fe crystal and the nearest neighbour of interstitial H atom; (c) H atom in tetrahedral interstice; (d) the nearest neighbour Fe atom of interstitial H atom.
Fig. 3. Electron density difference of α -Fe+H(T-site).
Fig. 4. Crystalline structure of α -Fe+(n H-Vac): (a) α -Fe+Vac; (b)α -Fe+(3H-Vac); (c) α -Fe+(4H-Vac); (d) α- Fe+(5H-Vac); (e) α -Fe+(1H-Vac); (f) α -Fe+(2H-Vac); (g) α -Fe+(6H-Vac).
Fig. 5. Isoelectronic density surface and electron density difference of α -Fe+(n H-Vac): (a) Electron density difference of α -Fe+Vac in surface (100); (b) electron density difference of α -Fe+Vac in surface (110); (c) isoelectric density surface of α -Fe+Vac; (d) electron density difference of α -Fe+(2H-Vac) in surface (010); (e) isoelectric density surface of α -Fe+(2H-Vac).
Fig. 6. Crystalline structure of α -Fe+Vac+H(T-site).
Fig. 7. Formation energy of defects at 0 K for different H chemical potentials.
Fig. 8. Equilibrium concentration of defects for different temperature and H chemical potentials: (a) Equilibrium concentration of vacancies; (b) equilibrium concentration of H occupying in vacancies; (c) equilibrium concentration of H occupying in interstitial positions.
Fig. 9. Equilibrium total concentration of H of α -Fe containing vacancies.
Fig. 10. Schematic diagram of Devnathan-Stachurski double electrolytic cell.
|
Table 1. [in Chinese]
|
Table 2. [in Chinese]
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 3. [in Chinese]
|
Table 4. [in Chinese]
|
Table 5.
Lattice parameters and crystalline structure of α-Fe+(nH-Vac).
α-Fe+(nH-Vac)的晶格常数和晶体结构
|
Table 6.
Binding energy, formation energy, and heat of solution of α-Fe+(nH-Vac).
α-Fe+(nH-Vac)的结合能、形成能和溶解热
| |||||||||||||||||||||||
Table 7.
Hydrogen trapping energy of α-Fe+(nH-Vac)
α-Fe+(nH-Vac)对H原子的陷阱能
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 8.
Atomic orbital population of α-Fe+(nH-Vac).
α-Fe+(nH-Vac)晶体原子轨道布居
|
Table 9.
Binding energy, formation energy, and heat of solution of α-Fe+Vac+H(T-site).
α-Fe+Vac+H(T-site)的结合能、形成能和溶解热
| ||||||||||
Table 10.
Calculated and test value of equilibrium concentration of H atom.
H平衡溶解度计算值和实验值
Set citation alerts for the article
Please enter your email address



