Shuai TANG, Wentai ZHANG, Junyu QIAN, Peng XIAN, Xiaoshan MO, Nan HUANG, Guojiang WAN. Long-term in Vitro Corrosion Behavior of Zinc in Ringer’s Solution [J]. Journal of Inorganic Materials, 2020, 35(4): 461
Search by keywords or author
- Journal of Inorganic Materials
- Vol. 35, Issue 4, 461 (2020)
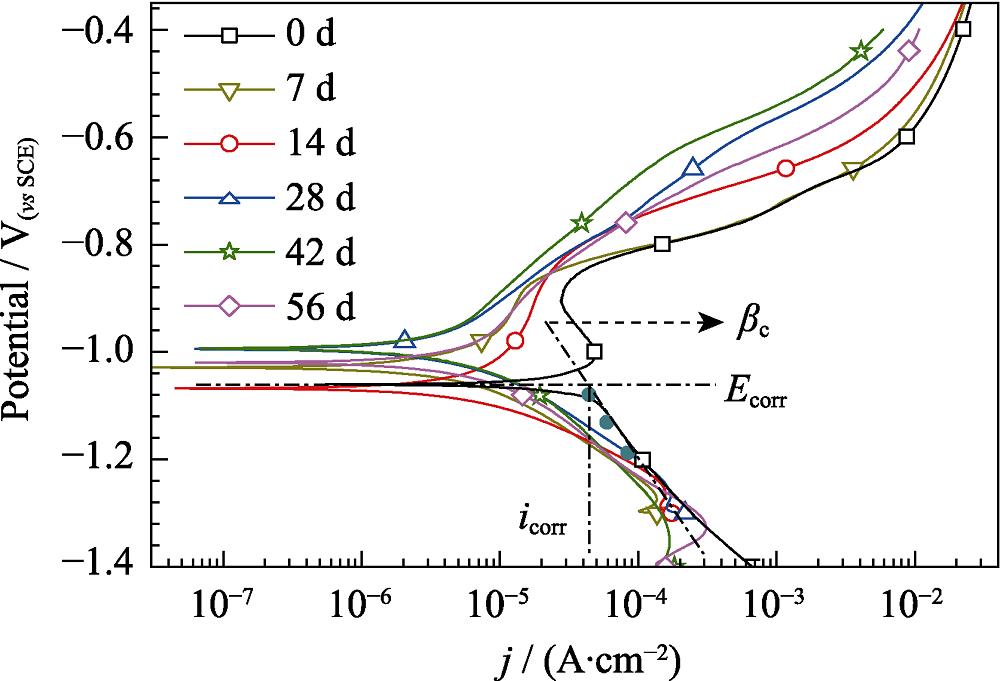
1. Potentiodynamic polarization curves of Zn immersed in Ringer’s solution at (37±0.5) ℃ for 7, 14, 28, 42 and 56 d
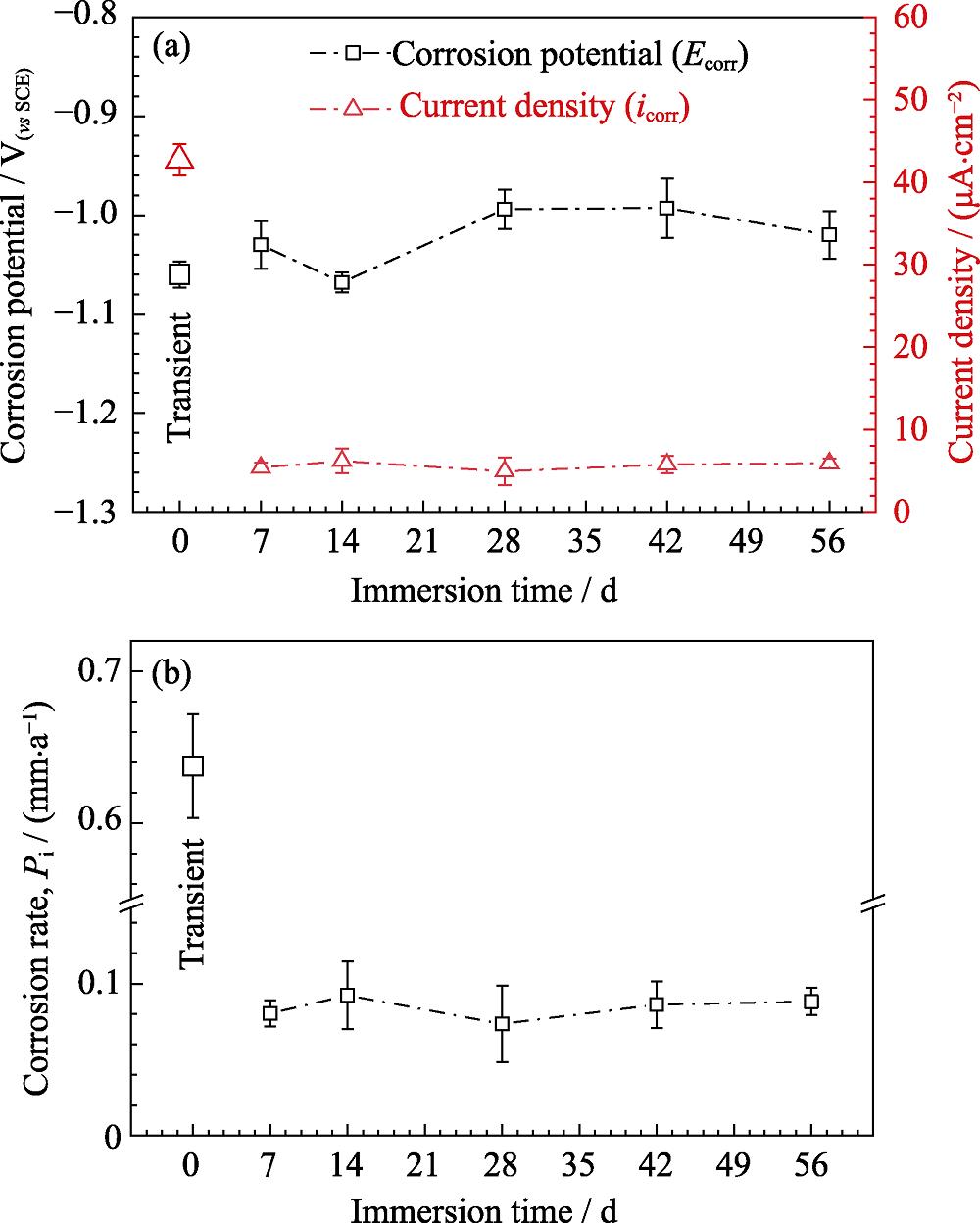
2. Parameters of corrosion potential and corrosion current density obtained from PDP curves (a), and corrosion rate P i obtained from current density (b)
3. Nyquist plots measured by Electrochemical Impedance Spectroscopy (EIS) (a), bode plots of |Z| vs . frequency (b) and bode plots of phase angle vs . frequency (c) of Zn immersed in Ringer’s solution at (37±0.5) ℃ for 7, 14, 28, 42 and 56 d
4. EIS data fitted with Equivalent electrical circuit (EEC) for 0 d (a) and EEC for 7 to 56 d (b), interfacial charge transfer resistance R ct and the corrosion products resistance R p obtained from fitted results of the EIS spectra (c), and polarization resistance R polar calculated from EIS components as a function of time (d)
5. Surface morphology of Zn immersed in Ringer’s solution at (37±0.5)℃ for 7 (a), 14 (b), 28 (c), 42 (d), and 56 (e) d
6. Cross-sectional images (a) and EDS line profile (b) of Zn immersed in Ringer’s solution at (37±0.5) ℃ for 56 d
7. XRD spectra of Zn immersed in Ringer’s solution at (37±0.5) ℃ for 7, 14, 28, 42, and 56 d
8. XPS spectra (a) and high-resolution O 1s (b) and (c) Ca 2p spectra of bare Zn and Zn after being immersed in Ringer’s solution for 56 d
9. Surface morphology of Zn immersed in Ringer’s solution at (37±0.5) ℃ for 7 (a), 14 (b), 28, (c) 42 (d), and 56 d (e) after removal of corrosion products
10. Cross-sectional SEM images of Zn immersed in Ringer’s solution at (37±0.5) ℃ for 56 d after removal of corrosion products
11. Corrosion rate P w calculated from weight loss of Zn after immersion in Ringer’s solution at (37±0.5) ℃ for 7, 14, 28, 42 and 56 d
|
Table 1. The evolution of fitted results of electrochemical impedance spectroscopy of Zn in Ringer’s solution at (37±0.5) ℃

Set citation alerts for the article
Please enter your email address



