Ma Haikuan, Zhang Xu, Zhong Shilei, Shi Xiaofeng, Ma Lizhen, Ma Jun. Detection of Antibiotics Based on Hyphenated Technique of Electrostatic-Preconcentration and Surface-Enhanced-Raman-Spectroscopy[J]. Chinese Journal of Lasers, 2018, 45(2): 207028
Search by keywords or author
- Chinese Journal of Lasers
- Vol. 45, Issue 2, 207028 (2018)
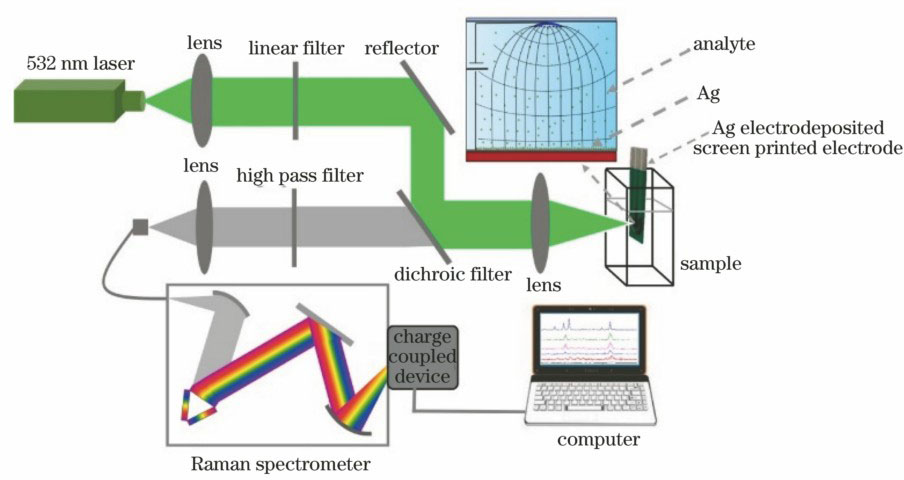
Fig. 1. Schematic of Raman detection system
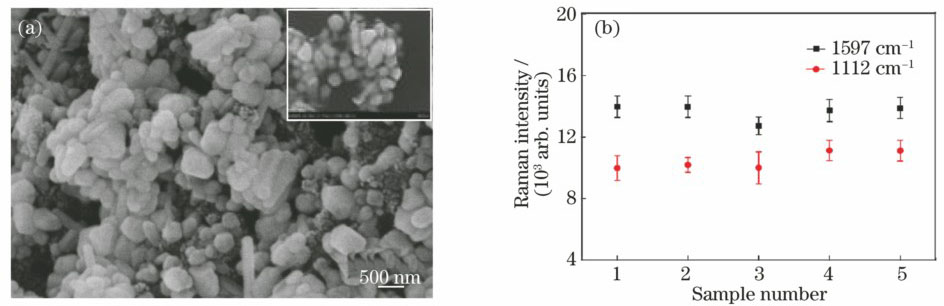
Fig. 2. (a) Morphology of Ag nano-film on carbon electrode; (b) test results of repeatability and stability of SERS electrode
Fig. 3. Raman spectra of sulfamerazine solutions with different pH values
Fig. 4. Relationship between Raman spectral intensity and pH value of sulfamethazine solution
Fig. 5. Raman spectra of sulfamerazine solutions under different working voltages
Fig. 6. Relationship between Raman spectral intensity and working voltage of sulfamethazine solution
Fig. 7. Raman spectra of sulfamerazine solutions under different accumulation time
Fig. 8. Relationship between Raman spectral intensity and accumulation time of sulfamethazine solution
Fig. 9. Raman spectra of sulfamerazine solutions with different concentrations
Fig. 10. Relationship between Raman spectral intensity and concentration of sulfamethazine solution
Fig. 11. Raman spectra of amikacin solutions with different concentrations
Fig. 12. Relationship between Raman spectral intensity and concentration of amikacin solution
Fig. 13. Raman spectra of enrofloxacin solutions with different concentrations
Fig. 14. Relationship between Raman spectral intensity and concentration of enrofloxacin solution
Fig. 15. Raman spectra of ciprofloxacin solutions with different concentrations
Fig. 16. Relationship between Raman spectral intensity and concentration of ciprofloxacin solution
|
Table 1. Linear relationships between Raman intensity and concentration of 4 kinds of antibiotics under low concentrations

Set citation alerts for the article
Please enter your email address



