Chaofeng ZHAO, Jiaren JIN, Yingzhong HUO, Lu SUN, Yuejie AI. Adsorption of Phenolic Organic Pollutants on Graphene Oxide: Molecular Dynamics Study [J]. Journal of Inorganic Materials, 2020, 35(3): 277
Search by keywords or author
- Journal of Inorganic Materials
- Vol. 35, Issue 3, 277 (2020)
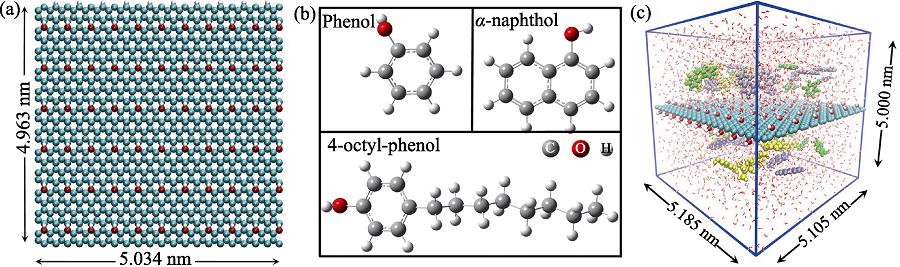
1. (a) GO model, (b) structures of phenol, α -naphthol and 4-octyl-phenol molecules in MD simulations, and (c) initial configuration of phenol (green), α -naphthol (purple) and 4-octyl-phenol (yellow) molecules in the competitive system
2. Equilibrium structures of (a) phenol, (b) α -naphthol and (c) 4-octyl-phenol molecules adsorbed on GO surface
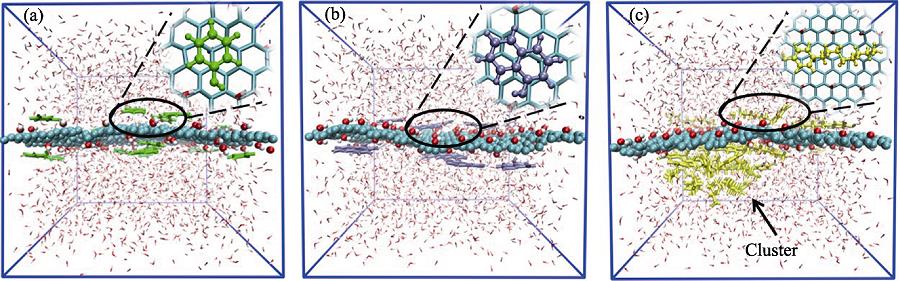
3. Snapshots of competitive system from simulation process at different time Phenol, α -naphthol and 4-octyl-phenol molecules are shown as green, purple and yellow molecules, respectively. Water molecules are not shown to highlight the configuration
4. Distances of centers of mass between GO and each (a) phenol, (b) α -naphthol and (c) 4-octyl-phenol molecule, as function of time
5. (a) Interaction energies between different POPs molecules in competitive system; The maximal cluster size of POPs in (b) independent and (c) competitive systems, respectively; The radial distribution functions (g(r )) and coordination numbers (n(r )) of POPs in (d) independent and (e) competitive systems, respectively
6. (a) Potential of mean force of POPs molecules; The interaction energies between GO and POPs molecules in (b) independent and (c) competitive systems, respectively; The hydrophobic areas of POPs molecules in (d) independent and (e) competitive systems, respectively; The hydrogen bonds between GO and POPs molecules in (f) independent and (g) competitive systems, respectively
S1. Initial configurations of (a) phenol, (b) α -naphthol and (c) 4-octyl-phenol molecules in the independent system
S2. SASAs of (a) phenol, (b) α -naphthol and (c) 4-octyl-phenol molecules in the independent system
S3. SASAs of (a) phenol, (b) α -naphthol and (c) 4-octyl-phenol molecules in the competitive system
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 1. Interaction energies between GO and POPs molecules in independent system at different periods
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 2. Interaction energies between GO and POPs molecules in competitive system at different periods

Set citation alerts for the article
Please enter your email address



