- Journal of Semiconductors
- Vol. 41, Issue 9, 091704 (2020)
Abstract
1. Introduction
Lithium-ion batteries (LIBs) are one of the most successful commercial rechargeable energy storage devices with high energy density and long cycle life[
Due to the possibility of using Zn metal as anodes, the study of cathode materials to match with the Zn anode is of the greatest interest for ZIBs[
On the other hand, although aqueous electrolytes have a lot of advantages and most of the reported ZIBs adopted aqueous electrolytes, it should be noted that the aqueous electrolytes face the challenge of decomposition of water under larger electrochemical window, which would lead to poor cycleability of aqueous ZIBs (AZIBs). However, the organic cathode materials have the merits of tunable electrode potential through molecular design, which therefore are particularly suitable for aqueous ZIBs. To date, the reported ZIBs with organic cathodes are mostly based on aqueous electrolytes, and the relevant research progress is focused in this review. However, to comprehensively summarize the progress of organic cathodes for ZIBs, the investigation of organic ZIBs (OZIBs) based on non-aqueous electrolytes are also discussed (the mentioned electrolytes in this review are aqueous unless the solvent is specifically noted). The challenges and strategies to improve the performance of organic cathode materials are also addressed in each part. Finally, the perspectives on OZIBs are put forward in hope of developing high-performance ZIBs and energy storage devices.
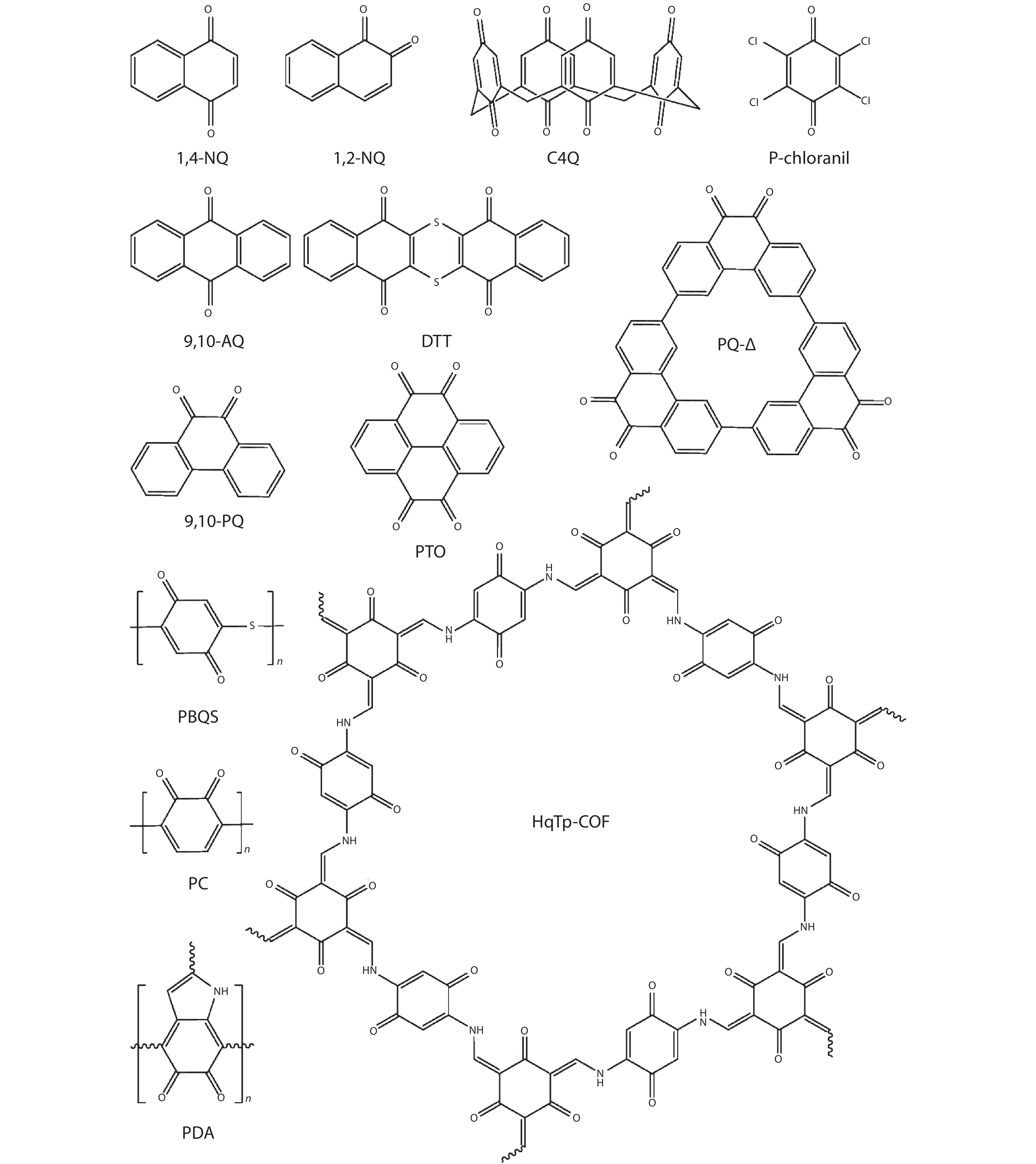
2. Quinones
Quinones are a kind of representative organic electrode materials and have exhibited decent electrochemical performance (high capacity, high rate capability, long cycleability etc.) as cathodes in LIBs based on the stable redox reaction of carbonyls[
![]()
Figure 1.The molecular structures of reported quinones as cathodes for ZIBs.
2.1. Quinone-based cathodes with insertion of Zn2+ ions
Most quinones as cathodes for ZIBs are reported to bind with Zn2+ ions during discharge–charge process. A series of small molecular quinone-based cathodes for ZIBs exhibited decent Zn-storage performance (Fig. 2(a))[
![]()
Figure 2.(Color online) (a) The voltages and capacities of 1,2-NQ, 9,10-PQ, 1,4-AQ, 9,10-AQ and C4Q in ZIBs. (b) The charge-discharge profiles of C4Q at 0.02 A/g and (c) cycling performance at 0.5 A/g in ZIBs with a Nafion separator. (d) The ESP mapping of C4Q. (e) The optimized structure of the C4Q and Zn3C4Q. Reproduced with permission from Ref. [
As mentioned above, small molecular quinones usually suffered capacity decay due to the dissolution of discharged salts in aqueous electrolytes. However, it was reported that a pyrene-4,5,9,10-tetraone (PTO) cathode for AZIBs was inherently insoluble in aqueous electrolytes, as well as the discharged products, permitting high cycleability[
![]()
Figure 3.(Color online) (a) The charge-discharge profiles of flexible Zn//PTO battery at flat state and 180° bending state at 1 A/g. (b) the cycling performance of flexible Zn//PTO battery at different bending state at 1 A/g. (c) the photos of LEDs and fan powered by the flexible Zn//PTO battery. Repoduced with permission from Ref. [
It is accepted that polymers usually have low solubilities in electrolytes and hence are expected to exhibit decent cycleability[
Thus, compositing polymers with conductive carbon materials is helpful for confining the polymers in the carbon materials and further inhibiting the dissolution of active materials. Moreover, the conductive carbon materials can increase the electrical conductivity of composite electrodes and thus benefits the rate capability[
![]()
Figure 4.(Color online) (a) The schematic diagram of the synthesis (top), photo (bottom left), and Zn-storage mechanism (bottom right) of PDA/CNTs. (b) The schematic diagram of the synthesis of PC/graphene. (c) Cycling performance of PDA/CNTs at 0.2 A/g in ZIBs. (d) Rate capability of PC/graphene. (a), (c) Reproduced with permission from Ref. [
It was widely accepted that the volume change of flexible organic electrode materials is relatively small in comparison with inorganic materials[
![]()
Figure 5.(Color online) (a) The charge-discharge curves of p-chloranil at 0.2 C at different cycles. (b) The cycling performance of p-chloranil/CMK-3 at 1 C. (c) The calculated crystal structure of p-chloranil and Zn2+-inserted p-chloranil; (d) SEM images of p-chloranil electrode and (e) p-chloranil/CMK-3 composite electrode at pristine (left), discharged (middle), and charged (right) state. Reproduced with permission from Ref. [
2.2. Quinone-based cathodes with insertion of more than Zn2+ ions
As mentioned above, most reported quinone-based cathodes for ZIBs are regarded to undergo the reversible coordination/de-coordination with Zn2+ ions during the discharge–charge process. However, some groups found that apart from Zn2+ ions, other ions in electrolytes can also bind with quinones and contribute capacity. Nam et al. believed that the discharge process of a triangular phenanthrenequinone-based macrocycle (PQ-Δ) was involved in the insertion of hydrated Zn2+ ions rather than the desolvated Zn2+ ions, which was revealed by the Fourier transform infrared spectroscopy (FTIR) and X-ray photoelectron spectroscopy (XPS) results (Figs. 6(a)–6(c))[
![]()
Figure 6.(Color online) (a) The FTIR spectra and (b) XPS spectra of PQ-Δ at pristine, discharged and charged states. (c) The energy storage mechanism of PQ-Δ in 2 M ZnSO4. Reproduced with permission from Ref. [
In view of the smaller size of H+ ion than that of Zn2+ ion and the previous report that the MnO2-based cathode for ZIBs displayed insertion of H+ ions[
In a short summary, quinones exhibit fine Zn-storage performance with high capacity. However, it is worth noting that the research of quinone-based cathodes in ZIBs is in infancy, and they face some challenges. For instance, the dissolution behavior needs to be overcome by polymerization or compositing with carbon materials to achieve stable cycleability. Besides, the Zn-storage mechanism requires further investigation. And the redox voltages of quinones are relatively low, which restricts the energy density of ZIBs.
3. Conducting polymers
Conducting polymers (CPs) are promising electrode materials for secondary batteries due to their high electrical conductivity and reversible redox reactions, which generally involve the anion-insertion in the polymer backbone. They have exhibited fine anion-storage performance as electrodes for metal-ion batteries[
3.1. Polyaniline
Among various CPs, PANI is an attractive electrode material due to its fine redox reversibility, easy synthesis from chemical or electrochemical methods, and the stability in air. PANI has exhibited fine electrochemical performance in ZIBs, however, there are still some problems that restrict its further application.
It has been accepted that among three states (leucoemeraldine, pernigraniline, emeraldine) of PANI, the emeraldine form of PANI at the half-oxidized state can be doped (protonated) after acid doping and the resulting emeraldine salt possesses high electrical conductivity; while leucoemeraldine (fully reduced state) and pernigraniline (fully oxidized state) are insulators even when doped with acid (Fig. 7)[
![]()
Figure 7.The schematic diagram of the redox mechanism of PANI.
3.1.1. Doped polyaniline
Forming doped PANI is the most common strategy for improving the electrochemical performance, which can be realized via chemical synthesis (called self-doped PANI) or forming composites with suitable materials that can provide protons. In this case, doped PANI can keep electrochemical activity in weakly acidic electrolytes, even neutral or basic electrolytes.
It is an effective method to synthesize self-doped PANI by introducing substituent groups (as shown in Fig. 8, e.g., –COOH, –SO3H, –OH, etc) as proton reservoirs.
![]()
Figure 8.Reported monomers for co-polymerization with aniline to form self-doped PANI.
For example, three aniline derivatives, o-aminobenzoic acid (o-ABA), m-aminobenzoic acid (m-ABA) and m-aminobenzenesulphonic acid (m-ABS) were employed to synthesize self-doped PANI via electro-polymerization with aniline[
![]()
Figure 9.(Color online) (a) The d
It was reported that the meta-substituted ABA-based PANI derivatives showed higher electrochemical properties than the ortho-substituted counterparts[
Apart from the co-polymerization of anilines with the substituted anilines, other redox-active monomers with proton-supply ability are also taken into consideration to synthesize self-doped PANI. PANMTh, which was synthesized by electrochemical copolymerization of aniline with N-methylthionine (also called as azure C), exhibited fine electrochemical activity in aqueous electrolytes with pH value of 10[
In addition to the chemical synthesis, mixing PANI with materials that possess proton-supply ability is also effective to form the composite cathodes that are less dependent on the pH values of electrolytes. For example, poly(3,4-ethylenedioxythiophene) polystyrene sulfonate (PEDOT:PSS) is an electrically conductive polymer, where the –SO3H group in PSS can function as a proton reservoir and provide enough H+ ions for the protonation of PANI (Fig. 10(c))[
![]()
Figure 10.(Color online) (a) The schematic diagram of the synthesis process, (b) long-term cycling performance at 10 A/g, and (c) proposed reduction mechanism of PANI-PEDOT:PSS-CNTs composite cathode in 2 M ZnSO4. Reproduced with permission from Ref. [
The doping of PANI can also be achieved by using other materials. For example, after the electro-deposition of PANI on carbon cloth (CC), soaking them into aqueous solution of K3[Fe(CN)6], and CC-PANI-FeCN composite was formed, where [Fe(CN)6]3– ions were reduced to [Fe(CN)6]4– ions by PANI during the soak process. Owing to the interactions between –NH+–/–NH+= groups in PANI and [Fe(CN)6]4– ions, the cycling performance of the CC-PANI-FeCN cathode (the capacity retention was 71% after 1000 cycles at 5 A/g) was superior to CC-PANI (capacity retention of 17% at same conditions) without the doping. Besides, fine rate capability (e.g. 162 and 125 mAh/g at 1 and 5 A/g, respectively) was achieved[
3.1.2. Compositing polyaniline with conductive materials
The above examples indicated that the electrochemical performance of PANI not only relies on its electrochemical activity, but also can be influenced by its electrical conductivity. In addition to the method by synthesizing doped PANI, another effective strategy for improving the electrical conductivity of PANI is mixing PANI with special conductive materials.
As one of the most efficient ways, PANI could be synthesized on the conductive materials during the electrochemical polymerization process, and thus the composite electrode with enhanced electrical conductivity can be directly used as electrodes for ZIBs. Different conductive substrates (Pt, carbon cloth (CC)[
![]()
Figure 11.(Color online) (a) Ex situ XPS spectra of PANI/CFs for ZIB at different state. (b) The schematic diagram of the ion storage mechanism of PANI/CFs. (c) The proposed redox mechanism of PANI/CFs in 1 M Zn(CF3SO3)2. Reproduced with permission from Ref. [
Furthermore, it has been reported that the functionalization of carbon materials via pre-treatment or in situ process during polymerization of PANI is helpful to strengthen the interactions between carbon materials and PANI. For example, electron-cyclotron-resonance (ECR) plasma treatment is an effective method to oxidize the surface of carbon materials homogeneously. After the treatment of ECR O2 plasma, the surface of carbon fibers (CF) had oxygen-containing functional groups (–OH, –COOH, –C=O). The functional groups enhanced the interactions with the deposited PANI, when such carbon fibers were used as substrates[
The aforementioned methods, including doping of PANI and mixing with conductive materials, are mainstream ways for improving the Zn-storage performance of PANI. Beyond these, the synthesis conditions of PANI, such as the control of monomer concentration[
3.1.3. Polyaniline-based flexible Zn-ion batteries
In view of the fine Zn-storage performance of PANI and the rise of flexible electronics, flexible AZIBs based on PANI have attracted increasing attention based on gel polymer electrolytes (GPEs) recently. A sandwich-like flexible ZIB was fabricated with polyacrylamide (PAM)-ZnSO4 GPE, nanowire array PANI cathode and nanosheet array Zn anode[
![]()
Figure 12.(Color online) (a) The schematic diagram of the self-healing process of the all-in-one ZIB. (b) The CV curves of the original ZIB and the ZIB after self-healing. (c) The cycling performance of the ZIB after several self-healing. (d) The practical presentation of the self-healing ZIB. Repoduced with permission from Ref. [
In summary, PANI is validated to be a superior Zn-storage electrode material with fine performance. However, the electrochemical activity of PANI highly depends on the pH value of electrolytes, which should be acidic and hence helpful for the easy protonation. Unfortunately, under such circumstances, the Zn anode faces corrosion. Extensive efforts have been reported to solve this challenge and effective strategies include the doping of PANI by chemical synthesis or mixing with proper materials. Besides, the capacity of PANI is relatively low and limited by the doping level. Moreover, the energy storage mechanism of PANI in AZIBs needs further investigation. Recently, Zhao et al. reported a novel poly(1,5-naphthalenediamine, NAPD) cathode for ZIBs (the repeating unit was naphthalene for poly(1,5-NAPD) and benzene for PANI; both linkages are –NH– or =N–)[
3.2. Other conducting polymers
Apart from PANI, many other CPs (Fig. 13) have also demonstrated fine Zn-storage performance, such as polypyrrole (PPy)[
![]()
Figure 13.The molecular structures of reported CPs as cathodes for ZIBs apart from PANI.
PPy is known as a bipolar CP, and both cations and anions could be inserted into PPy. Grgur et al. believed that the energy storage mechanism of PPy cathode for ZIBs was the insertion of anions from the electrolytes[
Normally, the cycleability of PPy electrode was inferior[
![]()
Figure 14.(Color online) (a) The schematic diagram of the fabricating process of flexible Zn and PPy electrode on PET. (b) The color change of flexible Zn//PPy battery at different voltages. (c) The cycling performance of flexible Zn//PPy battery. Repoduced with permission from Ref. [
Similar to PANI and PPy, PIn is also a N-heterocyclic CP. It shows less dependence on the pH value of the electrolytes than PANI. In the 1990s, Pandey et al. reported PIn cathode for ZIB with 1 M ZnSO4 electrolyte[
PTh, a S-heterocyclic CP, is also a potential cathode material for ZIB. The energy storage mechanism of PTh in ZIBs is considered as the doping/dedoping of anions from the electrolytes. The Zn//PTh battery showed an average discharge voltage of 1.2 V with 0.1 M Zn(ClO4)2 and 1 M LiClO4 in propylene carbonate electrolyte and ClO4– anions could insert/extract into/from PTh reversibly during cycling[
Poly(acetylene) (PAc) is the first reported CP and its application as electrode materials for secondary batteries can date back to 1980s[
In short, CPs are endowed with advantages of relatively high redox voltage as cathodes for ZIBs. Besides, fine rate capability could be expected owing to the high electrical conductivity; which, however, is affected by the doping level and the chemical environment (e.g. the pH value of the electrolytes). Furthermore, the capacity is also limited by the doping level. Thus, the modification of CPs needs further investigation for high-performance ZIBs.
4. Other redox compounds
In addition to the aforementioned organic/polymeric electrode materials, there are also many other redox-active organic materials that can be used as electrodes for ZIBs.
As representative carbonyl-based materials, quinones have been demonstrated as fine Zn-storage electrode materials and were studied widely. Other materials containing carbonyl groups were also reported. For example, anhydrides and imides are also potential cathode materials for ZIBs due to the redox activity of carbonyls. Wang et al. first reported 1,4,5,8-naphthalenetetracarboxylic dianhydride (NTCDA) and 1,4,5,8-naphthalene diimide (NTCDI) as cathodes for ZIBs[
Apart from the carbonyl compounds, C=N groups containing materials are also expected to possess Zn-storage capacity due to the redox of C=N groups. For example, diquinoxalino [2,3-a:2’,3’-c] phenazine (HATN) and a HATN-based polymer have exhibited fine energy storage performance in LIBs[
In view of the variety of organic materials, a series of bio-materials with C=N and C=O groups, riboflavin (RF), alloxazine (ALX) and lumazine (LMZ), were reported as electrodes for ZIBs[
Organic materials containing C=O or/and C=N groups are both considered as cation-insertion electrode materials for ZIBs, and they have exhibited high capacity but the voltage is relatively lower than the anion-insertion electrode materials. In addition to conducting polymers, arylamine compounds can also be utilized as anion-insertion cathodes for ZIBs. For example, Glatz et al. reported a 1,4-bis(diphenylaminobenzene) (BDB) cathode for ZIBs in a high-concentrated electrolyte (19 M LiTFSI and 1 M Zn(CF3SO3)2)[
5. Conclusions and perspectives
The investigation of organic electrode materials for ZIBs is appealing due to the flexibility, eco-friendliness, and designable molecular structure of organic compounds. Organic electrode materials have exhibited fine Zn-storage performance (as shown in Table 1). However, the research of OZIBs is still in infancy and the further development is hampered by many issues. Aimed at these challenges, various strategies have been proved to be effective. The challenges and corresponding solutions are specifically summarized as follows:
(1) The dissolution behavior: Small molecular organic materials often suffer capacity decay due to the dissolution of discharged products in electrolytes. Fortunately, such an issue can be alleviated by many methods. Polymerization is the most effective way to inhibit the dissolution of small molecules. Applying functional separators[
(2) The unclear energy storage mechanism: The Zn-storage mechanism is essential for further enhancing the performance OZIBs (particularly in aqueous electrolytes due to the various ions) and needs further exploration. For example, the Zn-storage mechanism of carbonyl compounds is considered as the coordination of Zn2+ ions with carbonyls, but some reports showed that quinones could also bind with H+ ions (e.g. DTT) or hydrated Zn2+ ions (e.g. PQ-Δ) during discharge.
(3) Inherent restrictions resulting from the organic electrode materials: Various organic materials have been validated to be promising cathodes for ZIBs and they have exhibited fine performance. However, they often suffer their own shortcomings. For example, quinones possess high capacity due to the abundant Zn-storage active sites, but the redox voltage is not high. The potential could be elevated to a certain extent, by introducing electron-withdrawing groups. Furthermore, the low electrical conductivity of quinones restricts their rate capability. It seems that the CPs with high electrical conductivity and high voltage are more promising as cathodes for ZIBs. But the capacity of CPs is low. Particularly, the acidic electrolytes are necessary to guarantee the electrochemical activity of PANI, but such electrolytes would cause Zn corrosion.
(4) The universal problems in ZIB, such as the low electrochemical window of aqueous electrolytes, the corrosion of Zn metal and the possible growth of Zn dendrites, are out of the scope of this review and will not be further illustrated here.
In conclusion, rapid development of OZIBs is expected if the above challenges can be solved well. We hope this review can provide insight into the development of high performance OZIBs.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 51773071), the National 1000-Talents Program, Innovation Fund of WNLO and the Fundamental Research Funds for the Central Universities (HUST : 2017KFYXJJ023, 2017KFXKJC002, 2018KFYXKJC018, and 2019kfyRCPY099).
References
[1] M Armand, J M Tarascon. Building better batteries. Nature, 451, 652(2008).
[2] B Dunn, H Kamath, J M Tarascon. Electrical energy storage for the grid: A battery of choices. Science, 334, 928(2011).
[3] J Y Hwang, S T Myung, Y K Sun. Sodium-ion batteries: Present and future. Chem Soc Rev, 46, 3529(2017).
[4] T Hosaka, K Kubota, A S Hameed et al. Research development on K-ion batteries. Chem Rev, 120, 6358(2020).
[5]
[6] Y Marcus. Ionic radii in aqueous solutions. Chem Rev, 88, 1475(1988).
[7] B Wang, Y C Wu, S M Zhuo et al. Synergistic effect of organic plasticizer and lepidolite filler on polymer electrolytes for all-solid high-voltage Li–metal batteries. J Mater Chem A, 8, 5968(2020).
[8] C Jiang, Y M Gu, M Tang et al. Toward stable lithium plating/strippingby successive desolvation and exclusive transport of Liions. ACS Appl Mater Interfaces, 12, 10461(2020).
[9] Y Y Liu, D C Lin, P Y Yuen et al. An artificial solid electrolyte interphase with high Li-ion conductivity, mechanical strength, and flexibility for stable lithium metal anodes. Adv Mater, 29, 1605531(2017).
[10] X Q Zhang, X B Cheng, X Chen et al. Fluoroethylene carbonate additives to render uniform Li deposits in lithium metal batteries. Adv Funct Mater, 27, 1605989(2017).
[11] Q Li, S P Zhu, Y Y Lu. 3D porous Cu current collector/Li-metal composite anode for stable lithium-metal batteries. Adv Funct Mater, 27, 1606422(2017).
[12] Z M Zhao, J W Zhao, Z L Hu et al. Long-life and deeply rechargeable aqueous Zn anodes enabled by a multifunctional brightener-inspired interphase. Energy Environ Sci, 12, 1938(2019).
[13] Y X Zeng, X Y Zhang, R F Qin et al. Dendrite-free zinc deposition induced by multifunctional CNT frameworks for stable flexible Zn-ion batteries. Adv Mater, 31, 1903675(2019).
[14] A Naveed, H J Yang, J Yang et al. Highly reversible and rechargeable safe Zn batteries based on a triethyl phosphate electrolyte. Angew Chem Int Ed, 58, 2760(2019).
[15] A Konarov, N Voronina, J H Jo et al. Present and future perspective on electrode materials for rechargeable zinc-ion batteries. ACS Energy Lett, 3, 2620(2018).
[16] L N Chen, Q Y An, L Q Mai. Recent advances and prospects of cathode materials for rechargeable aqueous zinc-ion batteries. Adv Mater Interfaces, 6, 1900387(2019).
[17] J Cui, Z W Guo, J Yi et al. Organic cathode materials for rechargeable zinc batteries: Mechanisms, challenges, and perspectives. ChemSusChem, 13, 2160(2020).
[18] D Yang, H T Tan, X H Rui et al. Electrode materials for rechargeable zinc-ion and zinc-air batteries: Current status and future perspectives. Electrochem Energy Rev, 2, 395(2019).
[19] D Selvakumaran, A Q Pan, S Q Liang et al. A review on recent developments and challenges of cathode materials for rechargeable aqueous Zn-ion batteries. J Mater Chem A, 7, 18209(2019).
[20] D L Chao, W H Zhou, C Ye et al. An electrolytic Zn-MnO2 battery for high-voltage and scalable energy storage. Angew Chem Int Ed, 58, 7823(2019).
[21] H Pan, Y Shao, P Yan et al. Reversible aqueous zinc/manganeseoxide energy storage from conversion reactions. Nat Energy, 1, 16039(2016).
[22] Z L Li, S Ganapathy, Y L Xu et al. Mechanistic insight into the electrochemical performance of Zn/VO2 batteries with an aqueous ZnSO4 electrolyte. Adv Energy Mater, 9, 1900237(2019).
[23] F Wan, Z Q Niu. Design strategies for vanadium-based aqueous zinc-ion batteries. Angew Chem Int Ed, 58, 16358(2019).
[24] L Y Zhang, L Chen, X F Zhou et al. Towards high-voltage aqueous metal-ion batteries beyond 1.5 V: The zinc/zinc hexacyanoferrate system. Adv Energy Mater, 5, 1400930(2015).
[25] R Trócoli, Mantia F La. An aqueous zinc-ion battery based on copper hexacyanoferrate. ChemSusChem, 8, 481(2015).
[26] J H Huang, Z Wang, M Y Hou et al. Polyaniline-intercalated manganesedioxide nanolayers as a high-performance cathode materialfor an aqueous zinc-ion battery. Nat Commun, 9, 2906(2018).
[27] B Lee, H R Lee, H Kim et al. Elucidating the intercalation mechanism of zinc ions into α-MnO2 for rechargeable zinc batteries. Chem Commun, 51, 9265(2015).
[28] C Wang, X C Ren, C H Xu et al. N-type 2D organic single crystals for high-performance organic field-effect transistors and near-infrared phototransistors. Adv Mater, 30, 1706260(2018).
[29] Q Q Wang, F X Yang, Y Zhang et al. Space-confined strategy toward large-area two-dimensional single crystals of molecular materials. J Am Chem Soc, 140, 5339(2018).
[30] Y Lu, J Chen. Prospects of organic electrode materials for practical lithium batteries. Nat Rev Chem, 4, 127(2020).
[31] M Tang, C Jiang, S Y Liu et al. Small amount COFs enhancing storage of large anions. Energy Storage Mater, 27, 35(2020).
[32] Y W Wu, R H Zeng, J M Nan et al. Quinone electrode materials for rechargeable lithium/sodium ion batteries. Adv Energy Mater, 7, 1700278(2017).
[33] Y R Wang, C X Wang, Z G Ni et al. Binding zinc ions by carboxyl groups from adjacent molecules toward long-life aqueous zinc-organic batteries. Adv Mater, 32, 2000338(2020).
[34] Q Zhao, W W Huang, Z Q Luo et al. High-capacity aqueous zinc batteries using sustainable quinone electrodes. Sci Adv, 4, eaao1761(2018).
[35] Z W Guo, Y Y Ma, X L Dong et al. An environmentally friendly and flexible aqueous zinc battery using an organic cathode. Angew Chem Int Ed, 57, 11737(2018).
[36] G Dawut, Y Lu, L C Miao et al. High-performance rechargeable aqueous Zn-ion batteries with a poly(benzoquinonyl sulfide) cathode. Inorg Chem Front, 5, 1391(2018).
[37] X J Yue, H D Liu, P Liu. Polymer grafted on carbon nanotubes as a flexible cathode for aqueous zinc ion batteries. Chem Commun, 55, 1647(2019).
[38] S Q Zhang, W T Zhao, H Li et al. Cross-conjugated polycatechol organic cathode for aqueous zinc-ion storage. ChemSusChem, 13, 188(2020).
[39] C L Wang, C Jiang, Y Xu et al. A selectively permeable membrane for enhancing cyclability of organic sodium-ion batteries. Adv Mater, 28, 9182(2016).
[40] M Tang, S L Zhu, Z T Liu et al. Tailoring π-conjugated systems: From π-π stacking to high-rate-performance organic cathodes. Chem, 4, 2600(2018).
[41] S L Zhu, M Tang, Y C Wu et al. Free-standing protective films for enhancing the cyclability of organic batteries. Sustain Energy Fuels, 3, 142(2019).
[42] Y Chen, M Tang, Y C Wu et al. A one-dimensional π-d conjugated coordination polymer for sodium storage with catalytic activity in Negishi coupling. Angew Chem Int Ed, 58, 14731(2019).
[43] H Y Li, M Tang, Y C Wu et al. Large π-conjugated porous frameworks as cathodes for sodium-ion batteries. J Phys Chem Lett, 9, 3205(2018).
[44] C L Wang, H L Dong, W P Hu et al. Semiconducting π-conjugated systems in field-effect transistors: A material odyssey of organic electronics. Chem Rev, 112, 2208(2012).
[45] C L Wang, H L Dong, L Jiang et al. Organic semiconductor crystals. Chem Soc Rev, 47, 422(2018).
[46] M A Khayum, M Ghosh, V Vijayakumar et al. Zinc ion interactions in a two-dimensional covalent organic framework based aqueous zinc ion battery. Chem Sci, 10, 8889(2019).
[47] C L Wang. Weak intermolecular interactions for strengthening organic batteries. Energy Environ Mater(2020).
[48] Y Chen, H Y Li, M Tang et al. Capacitive conjugated ladder polymers for fast-charge and -discharge sodium-ion batteries and hybrid supercapacitors. J Mater Chem A, 7, 20891(2019).
[49] C L Wang, Y Xu, Y G Fang et al. Extended π-conjugated system for fast-charge and -discharge sodium-ion batteries. J Am Chem Soc, 137, 3124(2015).
[50] Z L Li, H L Zhao. Recent developments of phosphorus-based anodes for sodium ion batteries. J Mater Chem A, 6, 24013(2018).
[51] D P Kundu, P Oberholzer, C Glaros et al. Organic cathode for aqueous Zn-ion batteries: Taming a unique phase evolution toward stable electrochemical cycling. Chem Mater, 30, 3874(2018).
[52] M Tang, Y C Wu, Y Chen et al. An organic cathode with high capacities for fast-charge potassium-ion batteries. J Mater Chem A, 7, 486(2019).
[53] K W Nam, H Kim, Y Beldjoudi et al. Redox-active phenanthrenequinone triangles in aqueous rechargeable zinc batteries. J Am Chem Soc, 142, 2541(2020).
[54] N Wang, X L Dong, B L Wang et al. Zinc-organic battery with a wide operation-temperature window from −70 to 150 °C. Angew Chem Int Ed, 132, 14685(2020).
[55] W Sun, F Wang, S Hou et al. Zn/MnO2 battery chemistry with H+ and Zn2+ coinsertion. J Am Chem Soc, 139, 9775(2017).
[56] J J Shea, C Luo. Organic electrode materials for metal ion batteries. ACS Appl Mater Interfaces, 12, 5361(2020).
[57] Y Chen, S M Zhuo, Z Y Li et al. Redox polymers for rechargeable metal-ion batteries. EnergyChem, 2, 100030(2020).
[58] K Ghanbari, M F Mousavi, M Shamsipur. Preparation of polyaniline nanofibers and their use as a cathode of aqueous rechargeable batteries. Electrochim Acta, 52, 1514(2006).
[59] N L D Somasiri, A G MacDiarmid. Polyaniline: characterization as a cathode active material in rechargeable batteries in aqueous electrolytes. J Appl Electrochem, 18, 92(1988).
[60] K Yamamoto, M Yamada, T Nishiumi. Doping reaction of redox-active dopants into polyaniline. Polym Adv Technol, 11, 710(2000).
[61] N Gospodinova, L Terlemezyan. Conducting polymers prepared by oxidative polymerization: Polyaniline. Prog Polym Sci, 23, 1443(1998).
[62] P Jiménez, E Levillain, O Alévêque et al. Lithium n-doped polyaniline as a high-performance electroactive material for rechargeable batteries. Angew Chem, 129, 1575(2017).
[63] A A Karyakin, A K Strakhova, A K Yatsimirsky. Self-doped polyanilines electrochemically active in neutral and basic aqueous solutions: Electropolymerization of substituted anilines. J Electroan Chem, 371, 259(1994).
[64] M S Rahmanifar, M F Mousavi, M Shamsipur. Effect of self-doped polyaniline on performance of secondary Zn-polyaniline battery. J Power Sources, 110, 229(2002).
[65] H Y Shi, Y J Ye, K Liu et al. A long-cycle-life self-doped polyaniline cathode for rechargeable aqueous zinc batteries. Angew Chem Int Ed, 57, 16359(2018).
[66] S L Mu, Q F Shi. Controllable preparation of poly(aniline-co-5-aminosalicylic acid) nanowires for rechargeable batteries. Synth Met, 221, 8(2016).
[67] S L Mu. Rechargeable batteries based on poly(aniline-co-o-aminophenol) and the protonation of the copolymer. Synth Met, 143, 269(2004).
[68] J Zhang, D Shan, S L Mu. A rechargeable Zn- poly(aniline-co-m-aminophenol) battery. J Power Sources, 161, 685(2006).
[69] C X Chen, X Z Hong, T T Xu et al. Electrosynthesis and electrochemical and electrochromic properties of poly(aniline-co-N-methylthionine). J Electrochem Soc, 162, G54(2015).
[70] C X Chen, X Z Hong, A K Chen et al. Electrochemical properties of poly(aniline-co-N-methylthionine) for zinc-conducting polymer rechargeable batteries. Electrochim Acta, 190, 240(2016).
[71] C X Chen, Z Y Gan, C Xu et al. Electrosynthesis of poly(aniline-co-azure B) for aqueous rechargeable zinc-conducting polymer batteries. Electrochim Acta, 252, 226(2017).
[72] Y Liu, L Y Xie, W Zhang et al. A conjugated system of PEDOT: PSS induced self-doped PANI for flexible zinc-ion batteries with enhanced capacity and cyclability. ACS Appl Mater Interfaces, 11, 30943(2019).
[73] Z Wang, J J Han, N Zhang et al. Synthesis of polyaniline/graphene composite and its application in zinc-rechargeable batteries. J Solid State Electrochem, 23, 3373(2019).
[74] W C Du, J F Xiao, H B Geng et al. Rational-design of polyaniline cathode using proton doping strategy by graphene oxide for enhanced aqueous zinc-ion batteries. J Power Sources, 450, 227716(2020).
[75] H Yao, Q J Li, M S Zhang et al. Prolonging the cycle life of zinc-ion battery by introduction of [Fe(CN)6]4− to PANI via a simple and scalable synthetic method. Chem Eng J, 392, 123653(2020).
[76] P Li, Z S Fang, Y Zhang et al. A high-performance, highly bendable quasi-solid-state zinc –organic battery enabled by intelligent proton-self-buffering copolymer cathodes. J Mater Chem A, 7, 17292(2019).
[77] F Trinidad. Performance study of Zn/ZnCl2, NH4Cl/polyaniline/carbon battery. J Electrochem Soc, 138, 3186(1991).
[78] K Ghanbari, M F Mousavi, M Shamsipur et al. Synthesis of polyaniline/graphite composite as a cathode of Zn-polyaniline rechargeable battery. J Power Sources, 170, 513(2007).
[79] Y Xia, D R Zhu, S H Si et al. Nickel foam-supported polyaniline cathode prepared with electrophoresis for improvement of rechargeable Zn battery performance. J Power Sources, 283, 125(2015).
[80] F Wan, L L Zhang, X Y Wang et al. An aqueous rechargeable zinc-organic battery with hybrid mechanism. Adv Funct Mater, 28, 1804975(2018).
[81] H Yu, G Liu, M Wang et al. Plasma-assisted surface modification on the electrode interface for flexible fiber-shaped Zn–polyaniline batteries. ACS Appl Mater Interfaces, 12, 5820(2020).
[82] X C Xiao, W J Liu, K Wang et al. High-performance solid-state Zn batteries based on a free-standing organic cathode and metal Zn anode with an ordered nano-architecture. Nanoscale Adv, 2, 296(2020).
[83] H M Cao, F Wan, L L Zhang et al. Highly compressible zinc-ion batteries with stable performance. J Mater Chem A, 7, 11734(2019).
[84] Y Zhang, Q R Wang, S S Bi et al. Flexible all-in-one zinc-ion batteries. Nanoscale, 11, 17630(2019).
[85] S Huang, F Wan, S S Bi et al. A self-healing integrated all-in-one zinc-ion battery. Angew Chem, 131, 4357(2019).
[86] S S Bi, F Wan, S Huang et al. A flexible quasi-solid-state bifunctional device with zinc-ion microbattery and photodetector. ChemElectroChem, 6, 3933(2019).
[87] Y Zhao, Y N Wang, Z M Zhao et al. Achieving high capacity and long life of aqueous rechargeable zinc battery by using nanoporous-carbon-supported poly(1, 5-naphthalenediamine) nanorods as cathode. Energy Storage Mater, 28, 64(2020).
[88] J Q Wang, J Liu, M M Hu et al. A flexible, electrochromic, rechargeable Zn//PPy battery with a short circuit chromatic warning function. J Mater Chem A, 6, 11113(2018).
[89] Z X Li, Y Huang, J Y Zhang et al. One-step synthesis of MnO
[90] X W Li, X L Xie, R Lv et al. Nanostructured polypyrrole composite aerogels for a rechargeable flexible aqueous Zn-ion battery with high rate capabilities. Energy Technol, 7, 1801092(2019).
[91] S Li, I Sultana, Z P Guo et al. Polypyrrole as cathode materials for Zn-polymer battery with various biocompatible aqueous electrolytes. Electrochim Acta, 95, 212(2013).
[92] A Lahiri, L Yang, G Z Li et al. Mechanism of Zn-ion intercalation/deintercalation in a Zn-polypyrrole secondary battery in aqueous and bio-ionic liquid electrolytes. ACS Appl Mater Interfaces, 11, 45098(2019).
[93] B N Grgur, M M Gvozdenović, J Stevanović et al. Polypyrrole as possible electrode materials for the aqueous-based rechargeable zinc batteries. Electrochim Acta, 53, 4627(2008).
[94] G Cxiricx-Marjanovicx, S Mentus. Charge-discharge characteristics of polythiopheneas a cathode active material in a rechargeable battery. J Appl Electrochem, 28, 103(1998).
[95] T J Simons, M Salsamendi, P C Howlett et al. Rechargeable Zn/PEDOT battery with an imidazolium-based ionic liquid as the electrolyte. ChemElectroChem, 2, 2071(2015).
[96] A Fdz de Anastro, N Casado, X E Wang et al. Poly(ionic liquid) iongels for all-solid rechargeable zinc/PEDOT batteries. Electrochim Acta, 278, 271(2018).
[97] P C Pandey. Electrochemical synthesis of polyindole and its evaluation for rechargeable battery applications. J Electrochem Soc, 145, 999(1998).
[98] Z J Cai, C W Hou. Study on the electrochemical properties of zinc/polyindole secondary battery. J Power Sources, 196, 10731(2011).
[99] Z Liu, A Prowald, O Höfft et al. An ionic liquid-surface functionalized polystyrene spheres hybrid electrolyte for rechargeable zinc/conductive polymer batteries. ChemElectroChem, 5, 2321(2018).
[100] Z J Cai, J Guo, H Z Yang et al. Electrochemical properties of electrospun poly(5-cyanoindole) submicron-fibrous electrode for zinc/polymer secondary battery. J Power Sources, 279, 114(2015).
[101] P J Nigrey. Lightweight rechargeable storage batteries using polyacetylene, (CH)
[102] B Häupler, C Rössel, A M Schwenke et al. Aqueous zinc-organic polymer battery with a high rate performance and long lifetime. NPG Asia Mater, 8, e283(2016).
[103] X S Wang, L Chen, F Lu et al. Boosting aqueous Zn2+ storage in 1, 4, 5, 8-naphthalenetetracarboxylic dianhydride through nitrogen substitution. ChemElectroChem, 6, 3644(2019).
[104] C Peng, G H Ning, J Su et al. Reversible multi-electron redox chemistry of π-conjugated N-containing heteroaromatic molecule-based organic cathodes. Nat Energy, 2, 17074(2017).
[105] R R Kapaev, I S Zhidkov, E Z Kurmaev et al. Hexaazatriphenylene-based polymer cathode for fast and stable lithium-, sodium- and potassium-ion batteries. J Mater Chem A, 7, 22596(2019).
[106] Z W Tie, L J Liu, S Z Deng et al. Proton insertion chemistry of a zinc-organic battery. Angew Chem Int Ed, 59, 4920(2020).
[107] L W Cheng, Y H Liang, Q N Zhu et al. Bio-inspired isoalloxazine redox moieties for rechargeable aqueous zinc-ion batteries. Chem - Asian J, 15, 1290(2020).
[108] H Glatz, E Lizundia, F Pacifico et al. An organic cathode based dual-ion aqueous zinc battery enabled by a cellulose membrane. ACS Appl Energy Mater, 2, 1288(2019).
[109] K Koshika, N Sano, K Oyaizu et al. An aqueous, electrolyte-type, rechargeable device utilizing a hydrophilic radical polymer-cathode. Macromol Chem Phys, 210, 1989(2009).
[110] Y W Luo, F P Zheng, L J Liu et al. A high-power aqueous zinc-organic radical battery with tunable operating voltage triggered by selected anions. ChemSusChem, 13, 2239(2020).
[111] C Jiang, M Tang, S L Zhu et al. Constructing universal ionic sieves via alignment of two-dimensional covalent organic frameworks (COFs). Angew Chem Int Ed, 57, 16072(2018).
[112] C Jiang, C L Wang. 2D materials as ionic sieves for inhibiting the shuttle effect in batteries. Chem - Asian J, 15, 2294(2020).
[113] L J Yan, C X Zhao, Y Sha et al. Electrochemical redox behavior of organic quinone compounds in aqueous metal ion electrolytes. Nano Energy, 73, 104766(2020).
[114] L J Yan, X M Zeng, Z H Li et al. An innovation: Dendrite free quinone paired with ZnMn2O4 for zinc ion storage. Mater Today Energy, 13, 323(2019).
[115] A Guerfi, J Trottier, I Boyano et al. High cycling stability of zinc-anode/conducting polymer rechargeable battery with non-aqueous electrolyte. J Power Sources, 248, 1099(2014).
[116] A Kitani. Performance study of aqueous polyaniline batteries. J Electrochem Soc, 133, 1069(1986).
[117] P F Liu, R Lv, Y He et al. An integrated, flexible aqueous Zn-ion battery with high energy and power densities. J Power Sources, 410/411, 137(2019).
[118] D Shan, S L Mu. Electrochemical characteristics of polyaniline synthesized in the presence of ferrocenesulfonic acid. Synth Met, 126, 225(2002).
[119] H Karami, M F Mousavi, M Shamsipur. A new design for dry polyaniline rechargeable batteries. J Power Sources, 117, 255(2003).
Set citation alerts for the article
Please enter your email address



