Objective Infrared (3--5 μm) lasers have various applications in the military and civilian areas. Nonlinear optical crystals are important in the field of infrared technology for implementing frequency conversion of the infrared laser. Recently, the synthetic and optical performance of quaternary lithium-sulfur compounds, Li2XMS4 (X stands for Ba, Mn, and Cd; M stands for Ge and Sn) with tetrahedron units are widely researched because of their outstanding nonlinear optical coefficient and large energy gap. Besides, thermal conductivity is another important parameter to evaluate the performance of nonlinear optical crystals. High thermal conductivity will decrease the heat loss of crystals and keep the optical device working normally. However, the quaternary lithium-sulfur compounds and suitable crystal size are difficult to obtain experimentally, thus the thermal conductivity is difficult to measure. Theoretical predictions on the thermal transport behavior of quaternary lithium-sulfur compounds are imperative. In this study, the representative Li2BaSnS4 is studied and compared with LiGaS2, to reveal the microcosmic influence factor of thermal conductivity.
Methods Lattice thermal conductivity, which is caused by lattice vibration, is the main component of thermal conductivity. By solving the linearized phonon Boltzmann equation with the relaxation time approximated (RTA) method, the lattice thermal conductivity of Li2BaSnS4 and LiGaS2 can be obtained. The phonon group velocity and lifetime play an important role in lattice thermal conductivity (Formula 1). Phonon lifetime is the representation of the phonon anharmonic effect. Phonon scattering rates are positive correlation with scattering intensity and the number of scattering channels, which are evaluated by Grüneisen parameters and weighted joint density of states (w-JDOS) (Formula 2). The harmonic and anharmonic phonon effects are calculated using PHONOPY and PHONO3PY programs combined with ab initio Simulation Package, which is based on density functional theory and projector-augmented-wave formalism. For exchange-correlation functional, electron-electron interactions are treated using a generalized gradient approximation in the form of Perdew, Burke, and Ernzerhof. The Li: 1s 22s 1, S: 3s 23p 4, Ba: 6s 2, Sn: 5s 25p 2, and Ga: 4s 24p 1 are treated as valence electrons. The cutoff energy used for plane-wave expansion of electron wave functions is set to 700 eV. The convergence criteria of force and energy are set to 0.01 eV/nm and 10 -7 eV, respectively. Then, using a finite-difference approach, 2×2×1 and 2×2×2 supercells for Li2BaSnS4 and LiGaS2, respectively, are considered. To make the calculation affordable, the interaction range of third-order interatomic force constants (IFCs) is both set to 0.6 nm for Li2BaSnS4 and LiGaS2. To calculate thermal conductivity κL, the q-point meshes for Li2BaSnS4 and LiGaS2 are selected at 8×8×6 and 7×8×9, respectively.
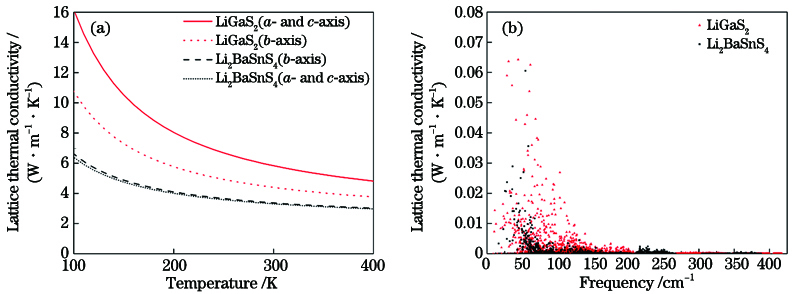
Results and Discussions After calculations, it is found that the thermal conductivity of Li2BaSnS4 and LiGaS2 is anisotropic and the lattice thermal conductivity of Li2BaSnS4 is lower than LiGaS2 in three directions (Fig.1). The representative value along the c-axis is studied. Also, it is shown that the three acoustic phonon modes and low-frequency (~50--150 cm -1) optical phonon modes (Fig.2) are the main contribution to total lattice thermal conductivity. The greatest difference in their lattice thermal conductivity appears naturally in the low-frequency region. We focus on Ba, Sn, and S of Li2BaSnS4 and Ga and S of LiGaS2 because their vibrational modes are located at low frequencies. When we analyze harmonic phonon properties, we find that the maximum difference of phonon group velocity also appears in the low-frequency region. The distribution of electron localization functions of Li2BaSnS4 and LiGaS2 and crystal orbital Hamilton population is used to evaluate the bond strength of Li2BaSnS4 and LiGaS2, which is an important influence factor of phonon group velocity. In Li2BaSnS4, Ba-S are the weaker ionic bonds, while Sn-S are the covalent bonds, and Ga-S are the stronger covalent bonds in LiGaS2. BaS8 dodecahedron and SnS4 tetrahedrons are alternately arranged in Li2BaSnS4; whereas, GaS4 tetrahedrons are linked directly in LiGaS2, which leads to the phonon group velocity of Li2BaSnS4 is lower than that of LiGaS2. However, the phonon lifetime of Li2BaSnS4 is lower than that of LiGaS2, especially in low-frequency optical phonon (Fig.6), which reveals that the phonon scattering effect of Li2BaSnS4 is stronger than that of LiGaS2. The reason is that the absolute value of Grüneisen parameters and w-JDOS of Li2BaSnS4 are larger than those of LiGaS2 (Fig.7).
Conclusions In this study, the lattice thermal conductivities of chalcogenides Li2BaSnS4 and LiGaS2 are obtained using linearized phonon Boltzmann equation and first-principle calculation. It is found that the lattice along the c-axis of ternary chalcogenides LiGaS2 at room temperature is higher than that of the quaternary lithium-sulfur compounds Li2BaSnS4. The lower contribution of thermal conductivity of acoustic phonon modes and low-frequency optical modes in Li2BaSnS4 is the main reason for the lower thermal conductivity of Li2BaSnS4. To further reveal microcosmic reason, harmonic and anharmonic phonon properties are calculated by obtaining the second and third IFCs. Although Sn-S and Ga-S are covalent bonds, Ba-S bonds are weaker, and SnS4 tetrahedrons are all linked with BaS8 dodecahedron, which leads to a lower phonon group velocity of Li2BaSnS4. Meanwhile, the addition of Ba increases phonon anharmonicity. These two factors combine to lower the thermal conductivity of Li2BaSnS4.