Lu Gao, Beibei Gao, Fu Wang. Applications of Super-Resolution Microscopy Techniques in Living Brain Imaging[J]. Chinese Journal of Lasers, 2022, 49(20): 2007301
Search by keywords or author
- Chinese Journal of Lasers
- Vol. 49, Issue 20, 2007301 (2022)
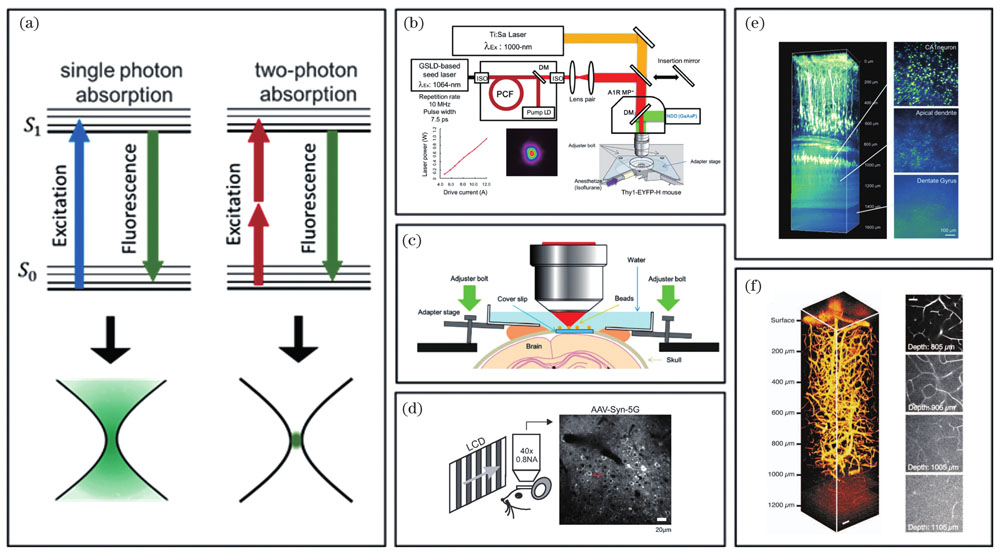
![Principle of two-photon excitation fluorescence scanning microscopy (2PLSM) and its applications in living brain imaging. (a) Jablonski diagram of single photon absorption and two-photon absorption and spatial distribution of their stimulated luminescence; (b) principle of 2PLSM imaging in mouse brain based on gain switched semiconductor laser diode[6]; (c) installation diagram of cranial window in mouse; (d) 2PLSM imaging of cortical activity induced by visual stimulation in mouse[24]; (e) 2PLSM imaging of cortical and hippocampal neurons in an adult mouse under gain excitation with a wavelength of 1064 nm[6]; (f) 2PLSM imaging of cerebral cortical blood vessels in a mouse under excitation light with a wavelength of 1280 nm[35], scaled bar: 50 μm](/richHtml/zgjg/2022/49/20/2007301/img_01.jpg)
Fig. 1. Principle of two-photon excitation fluorescence scanning microscopy (2PLSM) and its applications in living brain imaging. (a) Jablonski diagram of single photon absorption and two-photon absorption and spatial distribution of their stimulated luminescence; (b) principle of 2PLSM imaging in mouse brain based on gain switched semiconductor laser diode[6]; (c) installation diagram of cranial window in mouse; (d) 2PLSM imaging of cortical activity induced by visual stimulation in mouse[24]; (e) 2PLSM imaging of cortical and hippocampal neurons in an adult mouse under gain excitation with a wavelength of 1064 nm[6]; (f) 2PLSM imaging of cerebral cortical blood vessels in a mouse under excitation light with a wavelength of 1280 nm[35], scaled bar: 50 μm
![Principle of stimulated emission depletion (STED) microscopy and its applications in living brain imaging. (a) Jablonski diagram of STED and schematics of excitation, STED, and emission spots; (b) application of STED in mouse brain imaging[50]; (c) principle of three dimensional two-photon STED (3D-2P-STED) microscopy with aberration correction[58]; (d) schematic of 2P-STED imaging of mouse brain in vivo and imaging comparison of 2P-STED and 2PLSM[57]; (e) repetitive super-resolution imaging of mouse brain cortex using STED microscopy[56]](/richHtml/zgjg/2022/49/20/2007301/img_02.jpg)
Fig. 2. Principle of stimulated emission depletion (STED) microscopy and its applications in living brain imaging. (a) Jablonski diagram of STED and schematics of excitation, STED, and emission spots; (b) application of STED in mouse brain imaging[50]; (c) principle of three dimensional two-photon STED (3D-2P-STED) microscopy with aberration correction[58]; (d) schematic of 2P-STED imaging of mouse brain in vivo and imaging comparison of 2P-STED and 2PLSM[57]; (e) repetitive super-resolution imaging of mouse brain cortex using STED microscopy[56]
Fig. 3. Principle of structure illumination microscopy (SIM) and its applications in living brain imaging. (a) Principle of SIM[70]; (b) application of adaptive optics SIM (AO-SIM) in living brain imaging[59]; (c) schematic of optical slice SIM (OS-SIM) with adaptive optics (AO) and its imaging results[72]; (d) principle of patterned activation nonlinear SIM (PANL-SIM) and its imaging results[74]
Fig. 4. Experiments of brain activity in awake mouse. (a) Setup of an imaging system for brain activity in awake mouse based on 2PLSM[88]; (b) setup of an imaging system for brain activity in anesthesia mouse based on optical coherence tomography (OCT)[89]; (c) design and imaging of micro endoscope for deep imaging of the brain in a live mouse[84]; (d) system and imaging for head-fixed awake mouse[83]
|
Table 1. Comparison of various super-resolution imaging techniques applied to living brain imaging
Set citation alerts for the article
Please enter your email address



