Xiya Guo, Jinling Song, Baoying Wang, Xueling Zhu. Preparation of BiOClxBryIz Composite Catalysts and Its Visible Light Catalytic Degradation of Methyl Orange[J]. Acta Optica Sinica, 2021, 41(11): 1116003
Search by keywords or author
- Acta Optica Sinica
- Vol. 41, Issue 11, 1116003 (2021)
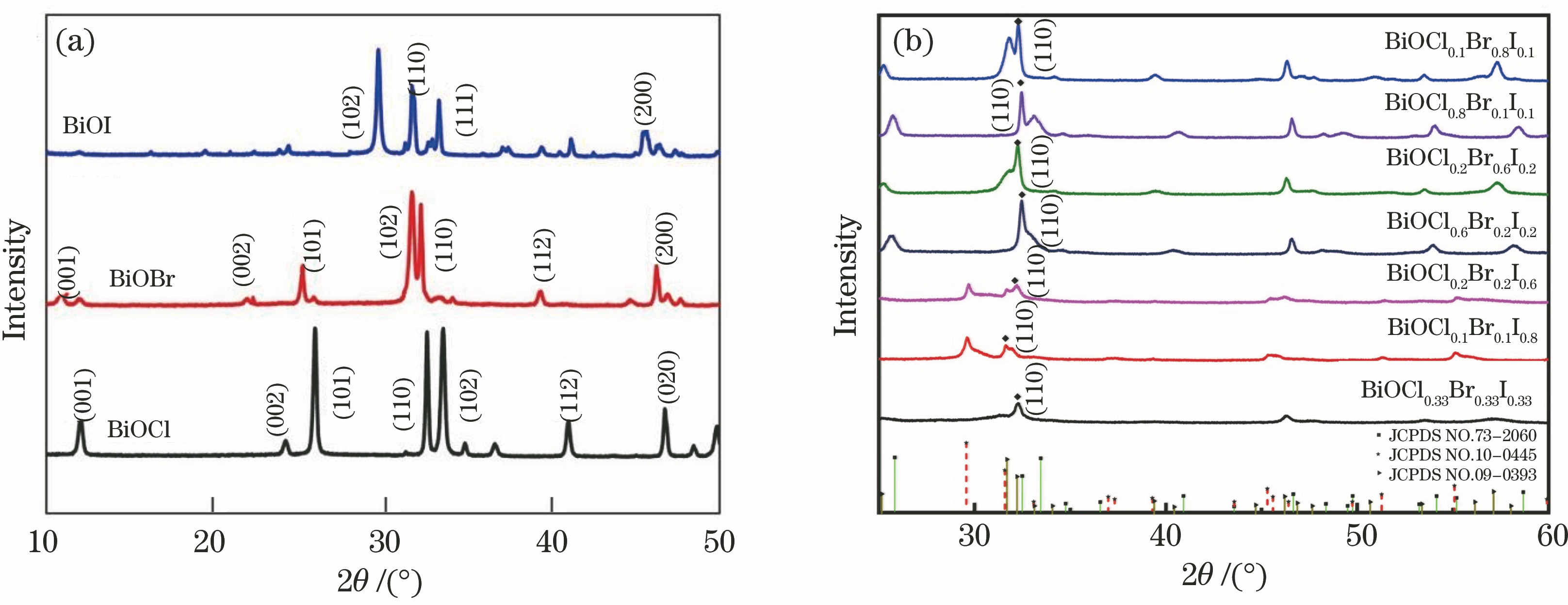
Fig. 1. XRD patterns of pure BiOX and BiOClxBryIz composite catalysts. (a) Pure BiOX; (b) BiOClxBryIz composite catalysts

Fig. 2. SEM images of BiOCl0.33Br0.33I0.33 composite catalyst
Fig. 3. EDS spectrum and mapping images of BiOCl0.33Br0.33I0.33 catalyst. (a) SEM image; (b) EDS spectrum; (c)--(h) mapping images
Fig. 4. TEM images of BiOCl0.33Br0.33I0.33 catalyst
Fig. 5. XPS spectra of BiOCl0.33Br0.33I0.33 composite catalyst. (a) Survey spectrum; (b) Bi 4f; (c) O 1s; (d) Cl 2p; (e) Br 3d; (f) I 3d
Fig. 6. UV-Vis diffuse reflectance spectra and (αhν)1/2-hν diagrams (inside) of BiOClxBryIz composite catalysts
Fig. 7. PL spectra of BiOClxBryIz composite catalysts
Fig. 8. Efficiency graphs of BiOClxBryIz composite catalysts and pure BiOX degrading 15 mg/L MO
Fig. 9. Reaction kinetics diagrams of BiOClxBryIz composite catalysts degrading 15 mg/L MO
|
Table 1. Dosage table of NaX in the preparation of BiOClxBryIz catalysts
|
Table 2. EDS analysis of each element in BiOCl0.33Br0.33I0.33 composite catalyst
|
Table 3. Band gaps, degradation efficiencies and kinetic constants of BiOClxBryIz composite catalysts
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 4. Photocatalytic activity of different BiOXs catalysts

Set citation alerts for the article
Please enter your email address



