Zhousu Xu, Jiazhi Xia, Xiaofeng Liu, Jianrong Qiu. Research Progress in Modulation of Optical Properties and Applications of CsPbX3 Perovskite Quantum Dot Doped Glasses[J]. Laser & Optoelectronics Progress, 2022, 59(15): 1516013
Search by keywords or author
- Laser & Optoelectronics Progress
- Vol. 59, Issue 15, 1516013 (2022)
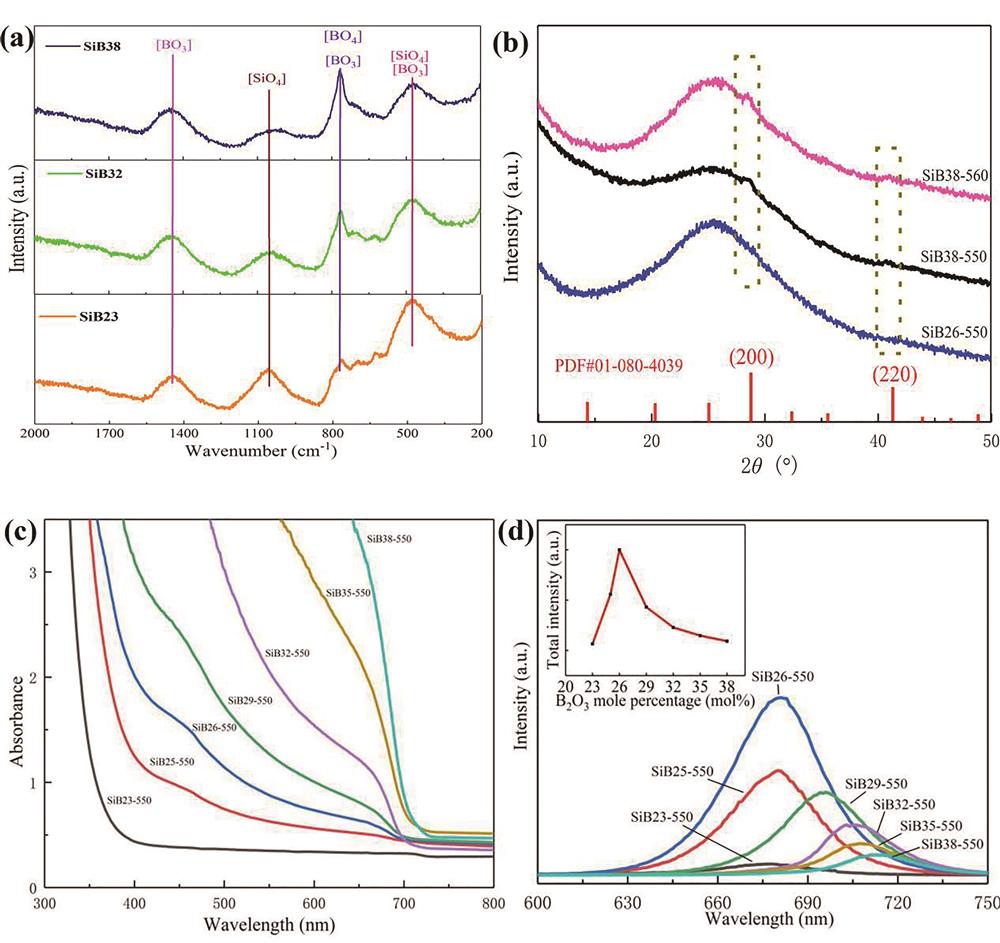
![Influence of the molar ratio of SiO2 to B2O3 on crystallization and optical properties of CsPbI3 quantum dots glass. (a) Raman spectra and (b) XRD patterns of CsPbI3 quantum dots; (c) absorption spectra and (d) photoluminescence spectra of quantum dots at different B2O3 contents (samples were marked as SiBx-y, x is the molar ratio of B2O3, and y is the heat treatment temperature) [27]](/richHtml/lop/2022/59/15/1516013/img_01.jpg)
Fig. 1. Influence of the molar ratio of SiO2 to B2O3 on crystallization and optical properties of CsPbI3 quantum dots glass. (a) Raman spectra and (b) XRD patterns of CsPbI3 quantum dots; (c) absorption spectra and (d) photoluminescence spectra of quantum dots at different B2O3 contents (samples were marked as SiBx-y, x is the molar ratio of B2O3, and y is the heat treatment temperature) [27]
![Influence of ZnO contents on crystallization and optical properties of CsPbI3 quantum dots glass. (a) FT-IR spectra[28]; (b) XRD patterns[28]; (c) PL spectra[28]; (d) PL quantum yield[28] (samples were marked as ZnOx-y-z, x is ZnO molar ratio, y is heat treatment temperature, and z is heat treatment time). Influence of Al2O3 contents on optical properties of CsPbBr3 quantum dots glass. (e) Excitation spectra[30]; (f) PL spectra[30] (samples were marked as Al2O3-x, x is Al2O3 molar ratio)](/richHtml/lop/2022/59/15/1516013/img_02.jpg)
Fig. 2. Influence of ZnO contents on crystallization and optical properties of CsPbI3 quantum dots glass. (a) FT-IR spectra[28]; (b) XRD patterns[28]; (c) PL spectra[28]; (d) PL quantum yield[28] (samples were marked as ZnOx-y-z, x is ZnO molar ratio, y is heat treatment temperature, and z is heat treatment time). Influence of Al2O3 contents on optical properties of CsPbBr3 quantum dots glass. (e) Excitation spectra[30]; (f) PL spectra[30] (samples were marked as Al2O3-x, x is Al2O3 molar ratio)
Fig. 3. Effect of different metal oxides (SrO, CaO, and MgO) on crystallization and optical properties of CsPbBr3 quantum dots glass. (a) XRD patterns; (b) TEM image; (c) PL spectra; (d) PL delay carves (S1, S2, and S3 represent samples doped with SrO, CaO, and MgO, respectively) [31]
Fig. 4. Influence of CaF2 contents on crystallization and optical properties of CsPbBr3 quantum dots glass. (a) FT-IR spectra [32]; (b) Raman spectra[32]; (c) XRD patterns[32]; (d) images of CsPbBr3 quantum dot glass at different heat treatment temperatures and different doping concentrations of CaF2 under UV lamp[32]; (e) absorption spectra[32]; (f) PL spectra of CsPbBr3 quantum dots[32]; (g) quantum yields curves of CsPbBr3 quantum dots under different heat treatment temperature and different CaF2 contents[32] (samples were marked as CPBx-y-z,x is CaF2 doping molar ratio, y is heat treatment temperature, and z is heat treatment time); (h) XRD patterns of the perovskite QDs glasses [33]
Fig. 5. Effect of AgO contents on crystallization and optical properties of CsPbBr3 quantum dots glass. (a) XRD patterns[34]; (b) TEM image[34]; (c) PL spectra[34]; (d) PL delay spectra[34] (samples were marked as CPBAx-y, x is the molar ratio of AgO doping, and y is the heat treatment temperature). Effect of Ag2O contents on CsPbBr3 quantum dots glass. (e) XRD patterns[35]; (f) infrared spectra[35]; (g) absorption spectra[35]; (h) PL spectra[35] (samples were marked as Gx,x is the AgO doping molar ratio)
Fig. 6. Effect of Dy3+ contents on optical properties of CsPbBr3 quantum dots glass. (a) PL spectra[37]; (b) CIE color coordinates of glass in different temperature[37] (illustration is glass sample picture excited in 365 nm) (samples were marked as BSx, x is the molar ratio of Dy3+ doping). Effect of Tb3+ contents on optical properties of CsPbBr3 quantum dots glass. (c) PL spectra[38]; (d) PL delay spectrum[38]; (e) fluorescence images during temperature rise[39]. Influence of Eu3+/Tm3+ co-doping on CsPbBr3 quantum dots glass. (f) PL spectra of samples excited at 375 nm[40]; (g) digital images[40]; (h) corresponding CIE color coordinates[40]
Fig. 7. Effect of K+ contents on optical properties of CsPbBr3 quantum dots glass. (a) PL spectra[41]; (b) thermal stability cycles images[41]. Effect of Rb+ contents on optical properties of CsPbBr3 quantum dots glass. (c) PL spectra[42]; (d) absorption spectra[42](x is Rb+ doping ratio). Effect of Sn2+ contents on optical properties of CsPbBr3 quantum dots glass. (e) PL spectra[43]; (f) PL quantum yield curves[43] (x is Sn2+ doping ratio). Effect of Cd2+ contents on crystallization and optical properties of CsPbBr3 quantum dots glass. (g) XRD patterns[44]; (h) PL spectra[44]
Fig. 8. Laser induced crystallization of CsPbX3 quantum dots. (a) Schematic diagram of direct photolithography of color perovskite nanocrystals and patterns[45]; (b)-(d) multi-pattern based on CsPbBr3-xIx nanocrystals[45]; (e) demonstration of optical information storage application[47]
Fig. 9. X ray imaging. (a) Schematic of experimental setup; (b)-(d) X ray image of sample[49]
Fig. 10. White LED is prepared by blue InGaN chip, Ce3+∶YAG phosphor, and Gd3+ doped CsPbBrI2 quantum dot glass. (a) PL spectra and corresponding images; (b) CIE coordinates and WLED device photographs; (c) schematic diagram of WLED structure and fluorescence images of glass powder under an UV lamp[53]
Set citation alerts for the article
Please enter your email address



