Xiaowen Kou, Bin Zhou, Xunchen Liu, Haixuan Chen, Meng Zhang, Pengfei Liu. Measurement of Trace NH3 Concentration in Atmosphere by Cavity Ring-Down Spectroscopy[J]. Acta Optica Sinica, 2018, 38(11): 1130001
Search by keywords or author
- Acta Optica Sinica
- Vol. 38, Issue 11, 1130001 (2018)
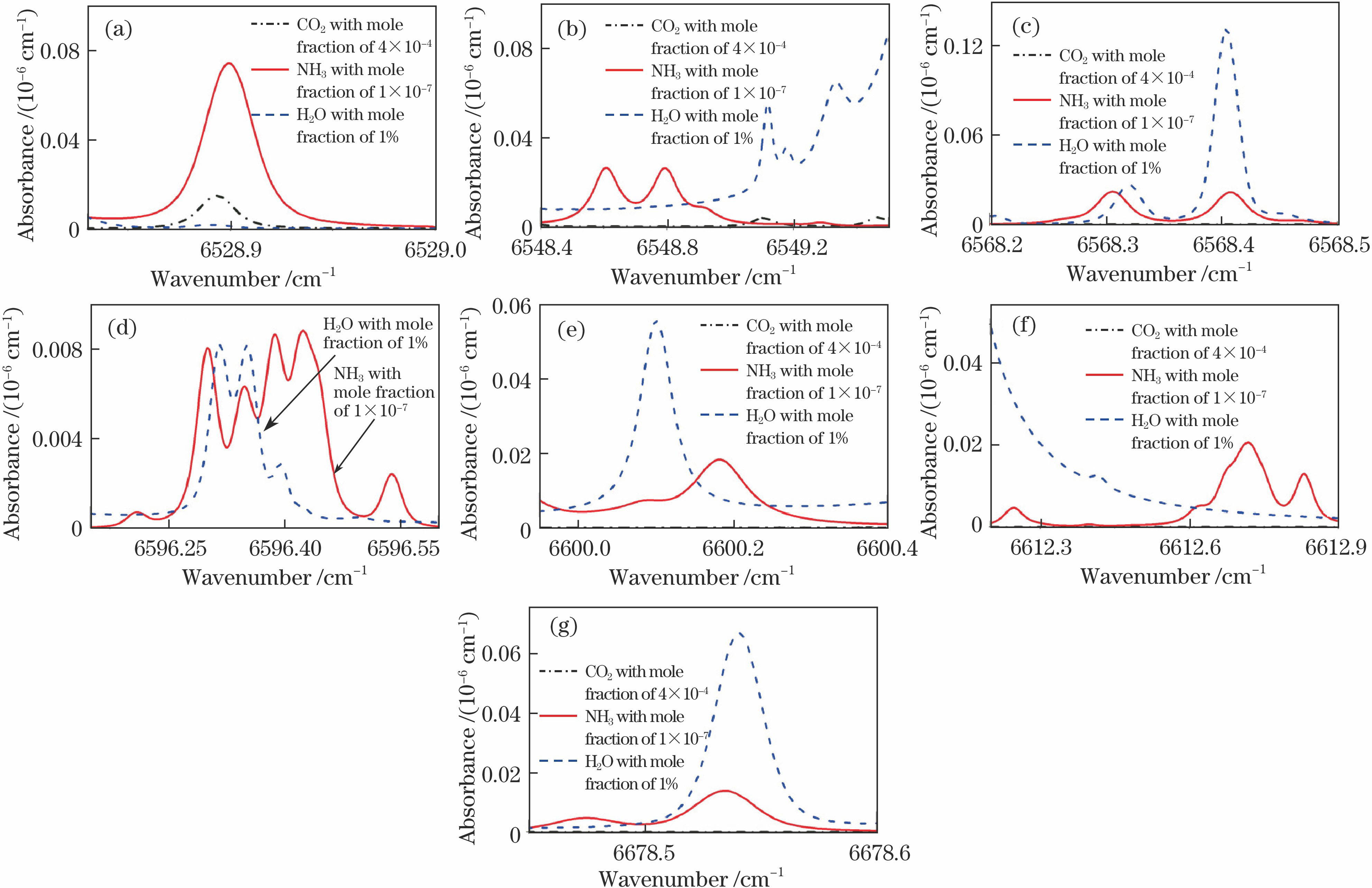
Fig. 1. Simulated absorbance of different gases near 1.5 μm wavelength (T=300 K, p=9.5 kPa, L=1.6×104 m). (a) 6528.9 cm-1; (b) 6548.7 cm-1; (c) 6568.4 cm-1; (d) 6596.4 cm-1; (e) 6600.2 cm-1; (f) 6612.7 cm-1; (g) 6678.5 cm-1
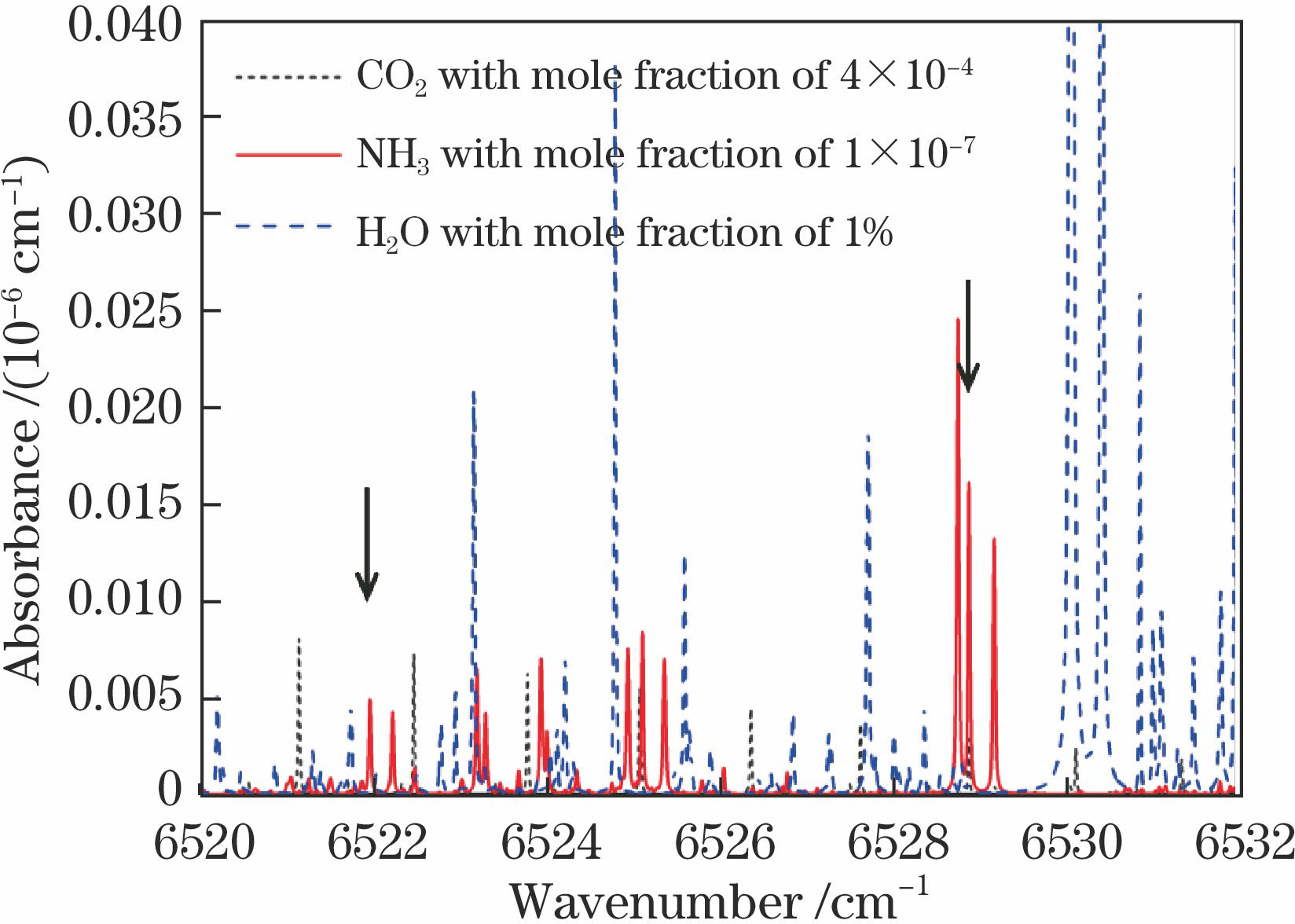
Fig. 2. Simulated absorbance of different gases in range of 6520-6532 cm-1 (T=300 K, p=9.5 kPa, L=1.6×104 m)
Fig. 3. Simulated absorbance of different gases near 6521.97 cm-1 (T=300 K, p=9.5 kPa, L=1.6×104 m)
Fig. 4. Schematic of laser mode matching
Fig. 5. Transmitted cavity mode signal
Fig. 6. Schematic of CRDS experimental system
Fig. 7. Experimental gas path
Fig. 8. Acquisition and fitting of ring-down curve
Fig. 9. Absorption lines of NH3, CO2 and H2O in atmosphere in range of 6520-6530 cm-1 (T=300 K, p=9.5 kPa, L=1.6×104 m). (a) Experimental measurement data; (b) simulation results in HITRAN database
Fig. 10. Absorption coefficient of 55 sampling points obtained by measurement
Fig. 11. Absorption results of 6521.97 cm-1 NH3 and 6522.04 cm-1 H2O spectral lines after baseline deduction
Fig. 12. (a) Voigt multi-peak fitting results of NH3 and H2O absorbance; (b) fitting residual
Fig. 13. (a) NH3 concentration obtained by a continuous measurement for 400 s in outdoor environment; (b) Allan deviation
|
Table 1. Cavity ring-down time obtained after acquisition and measurement for 1000 times
|
Table 2. Measurement results of NH3 and CO2 concentrations under different conditions

Set citation alerts for the article
Please enter your email address



