Xiang-Xue WANG, Shu-Jun YU, Xiang-Ke WANG, [in Chinese], [in Chinese], [in Chinese]. Removal of Radionuclides by Metal-organic Framework-based Materials[J]. Journal of Inorganic Materials, 2019, 34(1): 17
Search by keywords or author
- Journal of Inorganic Materials
- Vol. 34, Issue 1, 17 (2019)
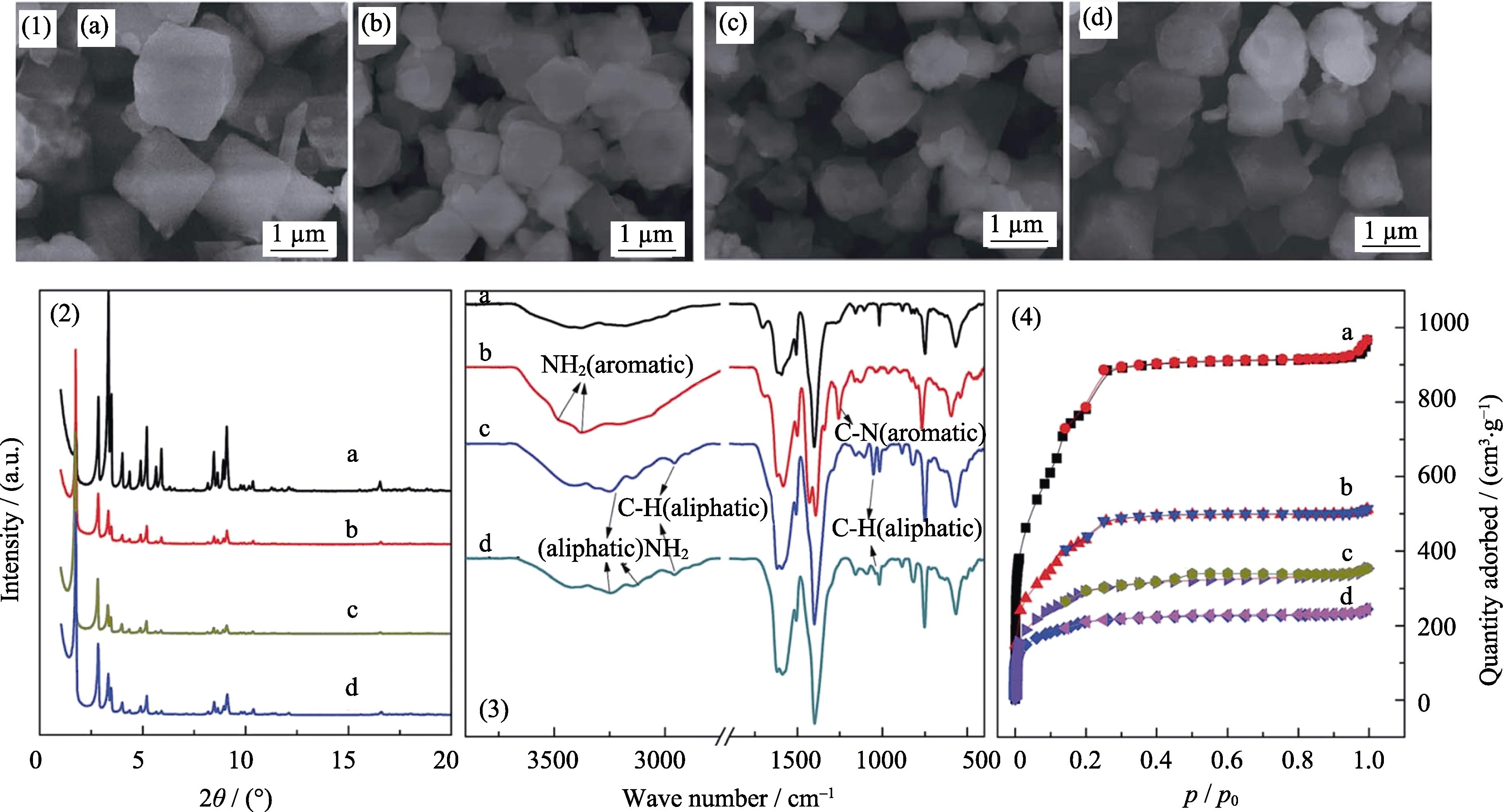
![(1) SEM images, (2) XRD patterns, (3) FT-IR spectra, (4) N2 sorption isotherms[26] of MIL-101 and its amino derivatives, (a) MIL-101; (b) MIL-101-NH2; (c) MIL-101-ED; (d) MIL-101-DETA](/richHtml/jim/2019/34/1/17/img_1.png)
. (1) SEM images, (2) XRD patterns, (3) FT-IR spectra, (4) N2 sorption isotherms[26] of MIL-101 and its amino derivatives, (a) MIL-101; (b) MIL-101-NH2; (c) MIL-101-ED; (d) MIL-101-DETA
![(a) UV-Vis absorption spectra of TcO4- during the anion exchange; (b) Sorption kinetics of TcO4- by SCU-101 compared with Purolite A530E and A532E; (c) Sorption isotherms of ReO4- by SCU-101, Mg-Al-LDH, and NDTB-1; (d) Effect of competing anions on the removal percentage of TcO4- by SCU-101; (e) Effect of SO42- on the anion exchange of ReO4- by SCU-101; (f) Removal percentage of ReO4- after irradiation as compared with the original SCU-101 sample[28]](/richHtml/jim/2019/34/1/17/img_2.png)
. (a) UV-Vis absorption spectra of TcO4- during the anion exchange; (b) Sorption kinetics of TcO4- by SCU-101 compared with Purolite A530E and A532E; (c) Sorption isotherms of ReO4- by SCU-101, Mg-Al-LDH, and NDTB-1; (d) Effect of competing anions on the removal percentage of TcO4- by SCU-101; (e) Effect of SO42- on the anion exchange of ReO4- by SCU-101; (f) Removal percentage of ReO4- after irradiation as compared with the original SCU-101 sample[28]
. Linear pseudo-first-order kinetic (a), pseudo-second-order (b), intraparticle diffusion (c) and elovich equation (d) for adsorption of Cs+ on MOF/KNiFC and MOF/Fe3O4/KNiFC[44]; (e) Isotherm model of U(VI) adsorption on UiO-66 (inset) and GO-COOH/UiO-66 composites; (f) Langmuir model, (g) Freundlich model, and (h) Dubinin-Radushkevich model[30]
. (a) Comparison of experimental U L3-edge XANES spectra for pristine MIL-101(Cr), and different ED contents grafting ED-MIL-101(Cr) samples after the adsorption of U(VI), (b) Experimental Fourier transform of the U L3-edge EXAFS data for different samples and their corresponding fits[54]
. MD simulations on the process of uranyl sorption into SZ-2. The top (a) and side (b) view of the simulation system-1 (uranyl cation approaching along the c axis); (c) The final snapshot (at t ¼ 100 ns) of run 1 (out of total 6) to show the importance of equatorial water of uranyl cation in mediating its binding to the SZ-2 (the blue dash line indication the hydrogen bond between equatorial water molecules and the dangling hydrogen bond acceptors); (d) Time evolution of the electrostatic and vdW interaction energies of uranyl cation with SZ-2 and water; (e) The number of equatorial water molecules of uranyl cation (pink curve) and the number of hydrogen bonds formed between equatorial coordinating water molecules and other acceptors (including F and O in main framework) as the function of simulation time[58]
|
Table 1. Radionuclides adsorption on different materials
|
Table 2. The main purpose, advantages and disadvantages of main adsorption characterization techniques mentioned above
Set citation alerts for the article
Please enter your email address



