Xiangkun YU, Kun LIU, Zhipeng LI, Yulu ZHAO, Jinyou SHEN, Ping MAO, Aiwu SUN, Jinlong JIANG. Efficient Adsorption of Radioactive Iodide by Copper/Palygorskite Composite [J]. Journal of Inorganic Materials, 2021, 36(8): 856
Search by keywords or author
- Journal of Inorganic Materials
- Vol. 36, Issue 8, 856 (2021)
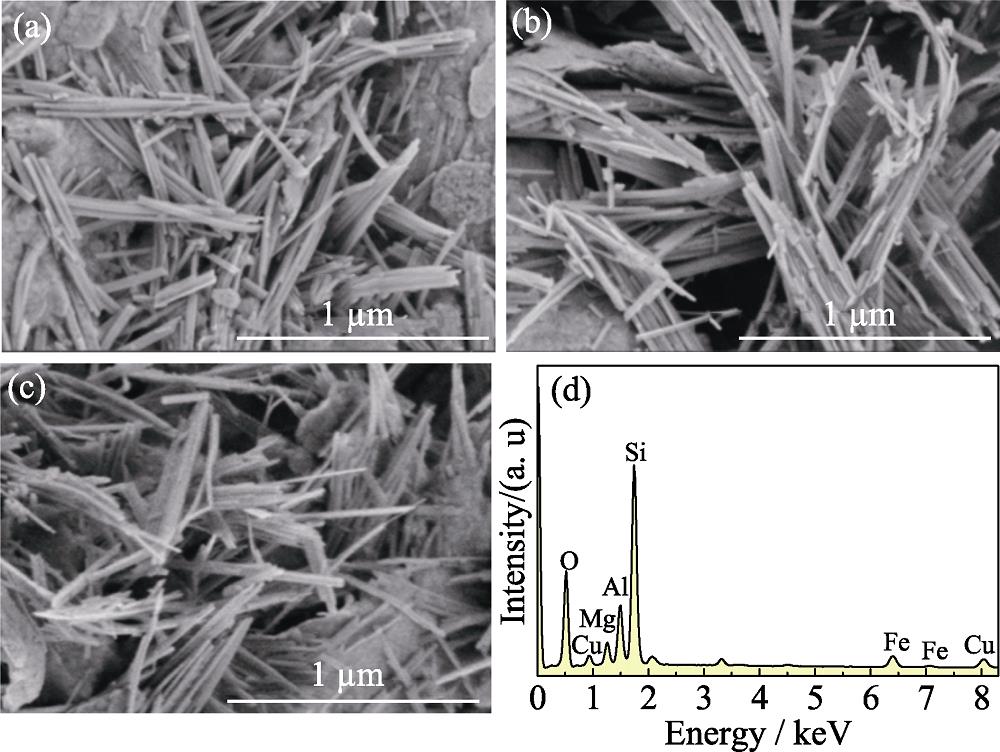
1. SEM images of the samples (a) PAL, (b) Cu 2+@PAL, (c) Cu@PAL and (d) EDX pattern of Cu@PAL
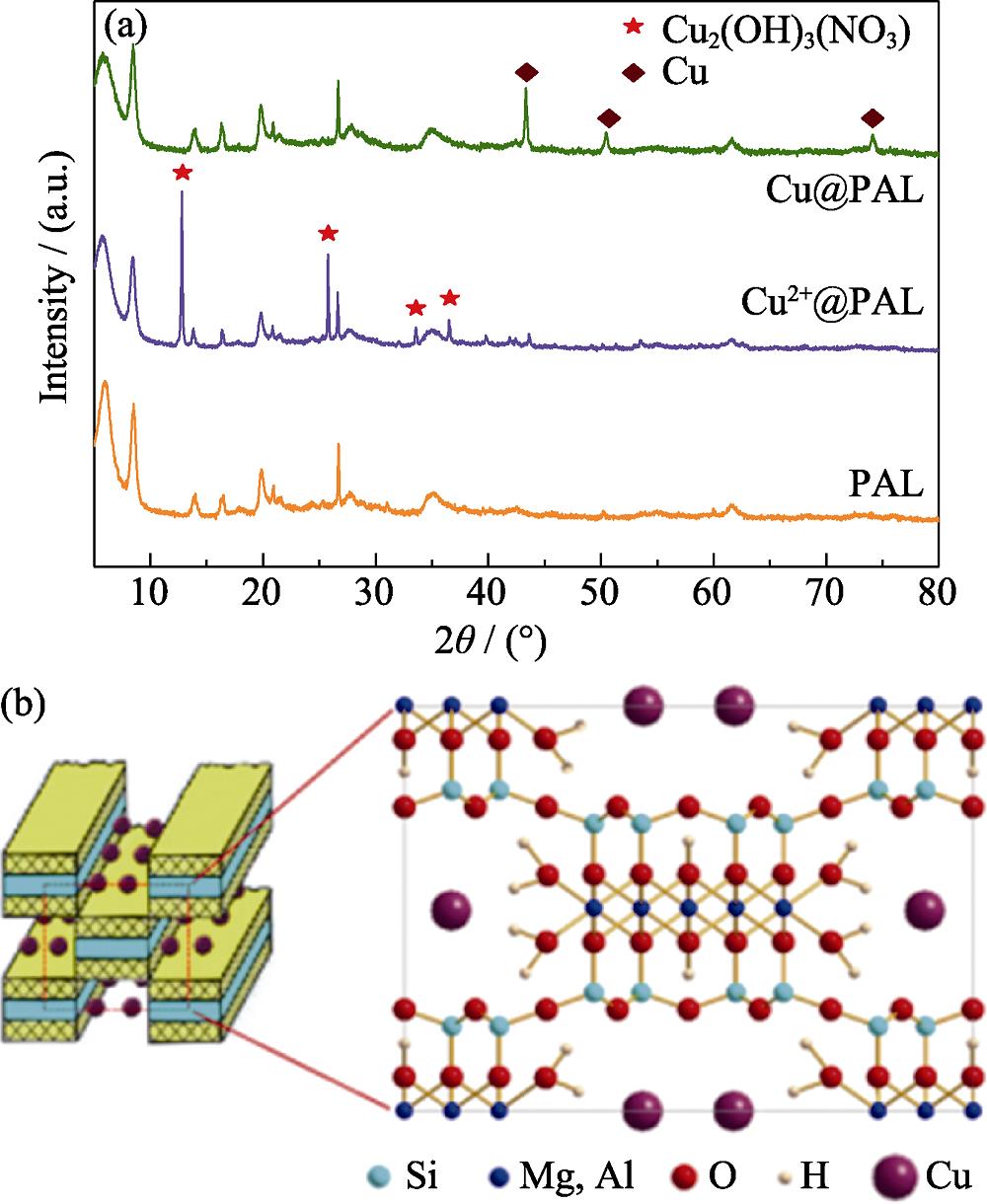
2. (a) XRD patterns of the samples and (b) structural schematic diagram of Cu@PAL
3. (a) TEM image of Cu2+@PAL and elemental mappings of (b) N, (c) O, (d) Al, (e) Si, and (f) Cu
4. (a) TEM image of Cu@PAL and elemental mappings of (b) N, (c) O, (d) Al, (e) Si, and (f) Cu; and high magnification TEM (g) and HRTEM (h) images of Cu@PAL
5. XPS spectra of the samples
6. (a) TGA curves and (b) FT-IR spectra of the samples
7. (a) N2 adsorption-desorption isotherms and (b) pore diameter distributions of the samples
8. (a) Adsorption capacity of I- anions on Cu@PAL and (b) XRD patterns of adsorption products in the solution with different pH
9. (a) Adsorption isotherm of the samples, fitting curves of (b) Langmuir model and (c) Frenudlich model for adsorption isotherm of Cu@PAL
10. SEM (a) and TEM (b) and HRTEM (c) images of I-Cu@PAL
11. Absorption kinetic curve (a), fitting curves of the pseudo first order kinetic model (b) and pseudo second order kinetic model (c) for adsorption kinetic of Cu@PAL
12. Effect of (a) anion and (b) cation on the adsorption of Cu@PAL
|
Table 1. Chemical analysis (XRF) of PAL and Cu@PAL/wt%
|
Table 2. Comparison of several Cu based adsorbents for iodide adsorption
| ||||||||||||||||||
Table 3. Isotherm parameters for the adsorption of I- anions by Cu@PAL
| |||||||||||||||||||||||
Table 4. Adsorption properties of Cu@PAL and nano Cu exposed to air
| ||||||||||||||||||
Table 5. Kinetic parameters for the adsorption of I- by Cu@PAL
|
Table 6. Leaching or desorption efficiencies of Cu@PAL and nano-Cu after adsorption

Set citation alerts for the article
Please enter your email address



