- Spectroscopy and Spectral Analysis
- Vol. 41, Issue 1, 327 (2021)
Abstract
Introduction
Dioxins are general of chlorine volatile organic pollutants (CVOCs) with 75 kinds of Polychlorinated Dibenzo-ρ-dioxins(PCDDs) and 135 species of Polychlorinated Dibenzo-ρ-dioxins(PCDFs). It has the characteristics of environmental persistence, biological chemicals, and long-term persistent with the harm of teratogenic, mutagenic and carcinogenic, and so on, which is regarded as one of the most toxic poisons on earth[
Lots of techniques have been developed for solving the release of dioxins from iron ore sintering flue gas. Among them, catalytic combustion is an effective method to complete mineralization of dioxins into CO2, H2O and HCl/CL2 at relatively low temperature[
Herein, the comparisons of and characterization of Ce-V-Ti prepare with the number of Ce/V weight ratio by sol-gel method were carried out. Chlorobenzene was used as the probe reactant to study the catalytic activity of catalyst under simulating iron ore sintering flue gas. Then, a one-step sol-gel method was developed to form Ce-V-Ti catalysts with a mass ratio of Ce:V from 2:1 to 8:1. The catalysts were characterized in detail by XRD, TEM, BET, XPS and TPR and catalytic activity. In addition, the structure and morphology properties of the catalysts were investigated. The main goal of this study is to present an optimal mass ratio of Ce/V to prepare Ce-V-Ti catalysts with high catalytic activity and resistance to water and sulfur.
1 Experimental
1.1 Catalysts synthesis
Ce-V-Ti catalysts were prepared by a sol-gel method. Ce(NO3)3·6H2O ethanol solution was added to Ti[O(CH2)3CH3]4 ethanol solution with about 15 wt% ratio of Ce/(Ti+Ce+V) under stirring. Then the solution of NH4VO3 and oxalic acid ( :noxalic acid=1:1) were added with about 2.5 Wt% ratio of V/(Ti+Ce+V) under stirring. The condensation solution was slowly gelled after finishing the reaction between Ti[O(CH2)3CH3]4 and H2O at room temperature. The gel was dried at 110 ℃ for 12 h and then calcined at 450 ℃ for 3 h. The obtained samples were labeled as Ce(x)-V(y)-Ti catalysts, where x and y represent the weight percent of Ce and V, respectively.
1.2 Characterization of catalysts
Characterization by XRD, BET, XPS and FTIR was carried out. The phase structures of catalysts were analyzed by a German D8ADVANCE X-ray diffractometer (XRD) with CuKα radiation (CuKα=0.154 06 nm). The surface area and distribution of pore size were carried on an ASAP-2020 surface analyzer from Micromeritics, USA. X-ray photoelectron spectroscopy (XPS) was used to analyze the oxidation states on the surface of the CeO2-VOx-TiO2 catalysts with AXIS ULTRP, which using AlKα(1 486.6 eV) radiation as the excitation source (powered at 10 mA and 15 kV). FT-IR spectra were recorded at room temperature on a Fourier Transform Infrared. Spectrometer (FT-IR, Nicolet, 5DXC) in the 500~4 000 cm-1 range and a resolution of 4 cm-1.
1.3 Catalytic activity measurement
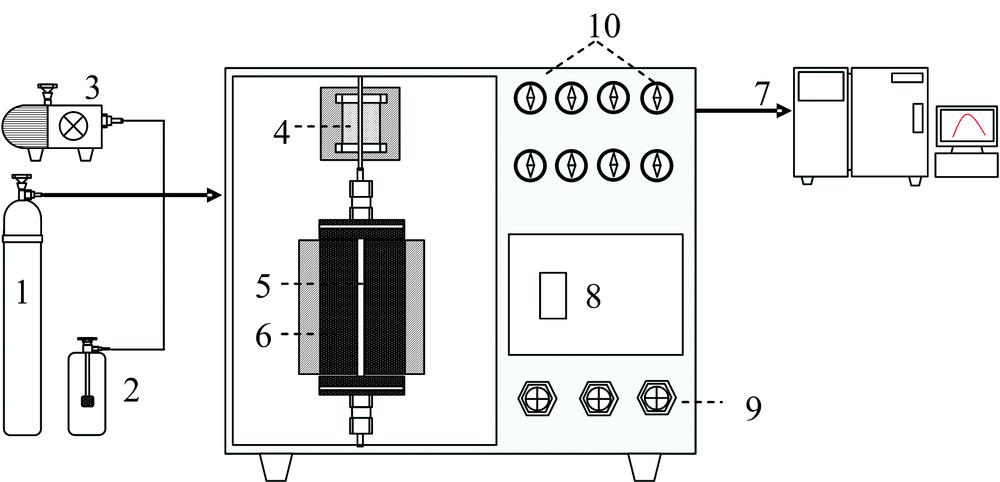
Catalytic activity was carried out in a self-designed apparatus at atmospheric pressure of 100~400 ℃ (Fig.1). Ce-V-Ti catalysts were placed in a silica tube with 3 mm inner diameter. As dioxins are very highly toxic and hard to handle, chlorobenzene (CB) was employed to be model compounds to predicted the catalytic activity of dioxins over different catalysts. The CB containing feed stream to the reactor was carried by N2 and controlled by a flow controller with a typical GHSV (gas hourly space velocity) of 30 000 h-1. The reaction flue gas was composed of 16% O2 and 84% N2, and the CB concentration was set to 100 ppm to simulate iron ore sintering flue gas. The recent gases were analyzed under a given temperature by using an online gas chromatograph (GC) equipped with flame ionization detector (FID1) and SE-54 capillary column for the quantitative analysis the inlet and outlet of CB. During the CB analysis, the temperature of the column and detector of GC with nitrogen as carrier gas was set up as 100 ℃ and 150 ℃, respectively.
![]()
Figure 1.Diagram of catalyst activity test system
1: Gas cylinder; 2: CB generator; 3: Air pumps; 4: Preheating furnaces; 5: Silica tube; 6: Holding furnace; 7: GC; 8: Controlling panel; 9: Controlling valve; 10: Pressure gages
The conversion ratio of CB (η) was calculated as follows
where, the CBin and CBout were the inlet and outlet concentration of CB in the system at steady-state, respectively.
2 Results and discussion
2.1 Overview of catalysts
The CB conversion over Ce-V-Ti catalysts with a different weight ratio of Ce was present in Fig.2(a). Ce(15)-V(2.5)-Ti catalysts performed a best catalytic activity in all catalysts. It can remove 58% CB at 150 ℃. However, the CB conversion over Ce-V-Ti catalysts with a higher or lower composition of Ce than 15Wt% were decline obviously. It indicated that adding too little of Ce had no obvious effect on improving the catalytic activity of CB combustion[
![]()
Figure 2.The catalytic activity of CB over Ce-V-Ti catalysts with different ratioof Ce/V
The CB conversion over Ce-V-Ti catalysts with a different weight ratio of V was present in Fig.2(b). The CB conversion was improved obviously by adding a little of V ac co-activity content. In general, the amount of V added to commercial catalysts is between 0~4%. When 2.5% V was introduced into Ce(15)-Ti catalysts, the catalytic activity was effectively promoted. The order of catalytic activity at 200 ℃ was Ce(15)-V(2.5)-Ti>Ce(15)-V(2)-Ti>Ce(15)-V(5)-Ti>Ce(15)-V(10)-Ti. It indicated that VOx might aggrege on the surface of catalysts when adding much more V than 5 Wt%[
2.2 Characterization of the catalysts
Fig.3(a) shows the XRD pattern of Ce-Ti, V-Ti and Ce-V-Ti catalysts. Among them, Ce-Ti and V-Ti catalysts were also prepared by sol-gel method, and the weight ratio of Ce/V was the same as that of Ce-V-Ti catalysts. The strong peak of anatase phase TiO2 (PDF 01-084-1286) was detected in all samples. There were no peaks corresponded to CeO2 and V2O5 because it’s covered by strong support peaks of anatase and low crystallinity due to good dispersion of Ce and V on the surface of Ti. As we can see from Fig.3(b),the intensity of Ce-V-Ti at 23°~28° was wider and shorter than Ce-Ti and V-Ti catalysts due to the solid solution reaction of Ce and Ti. The XRD pattern of TiO2 at (101) crystal plane shifted to the left (from 25.33° to 25.18°). It indicated that the lattice constant “a” (from 3.782 to 3.808 Å) and the constant “c” increased together (from 9.502 to 9.545 Å) in the Ce-V-Ti catalysts. When the lattice constants “a” and “c” increased simultaneously, the chemical bond between metal elements and oxygen in the catalysts would become longer[
![]()
Figure 3.XRD patterns of Ce-V-Ti catalysts (a);vitiation of the main peak of anatase (b)
Table 1 showed the specific surface area, pore volume and pore diameter of Ce-V-Ti catalyst. Surface and pore properties were changed with different metal oxides loaded on based-Ti. The SBET of V-Ti catalysts was 23.08 m2·g-1, which was the smallest one. When Ce is loaded, the specific surface area of the Ce-Ti catalysts increased to 90.53 m2·g-1. The specific surface area of Ce-V-Ti catalysts further increased to 95.53 m2·g-1 by introducing transition metal of V into Ce-Ti. It indicated that V made for the solid solution reaction of Ce into Ti[
The information on the atomic concentration and element chemical state on the catalyst surface was investigated by XPS. Fig.4 (a—c) illustrated the obtained XPS spectra for Ce(3d), O(1s) and V(2p), respectively. As shown in Fig.4(a), the Ce(3d) spectra reveled that the Ce(3d) orbit was composed of two multiples (v and u). The u, u″, u‴, v, v″ and v‴ peaks were attributed to Ce4+ species, while u' and v' were assigned to Ce3+ species. During the recycle reaction of catalytic combustion, active oxygen species are released through reduction reaction of Ce4+→ Ce3+. Then Ce2O3 captures O from oxygen on the surface of catalysts, and re-oxidized to CeO2.
(1)
(2)
![]()
Figure 4.XPS spectra for: Ce(3
O(1s) spectra [Fig.4(b)] were deconvoluted into two contributions: one with binding energy (BE) of 529.3~529.8 eV was assigned to lattice oxygen (Oα) and the other with BE of 531.0~531.4 eV (Oβ) wasassigned to surface-adsorbed oxygen, such as or O-. Meanwhile, Ce-V-Ti catalysts with a lot of Ce4+ icons possessed a considerable amount of active surface oxygen chemically adsorbed on the vacancies (O-). Ce-V-Ti catalysts show the highcatalytic activity of combustion CB.
Fig.4(c) showed XPS curves of surface vanadium species. In Ce-V-Ti samples, V(2p3/2) level near 517.2 eV was ascribed to V5+ species and at 516.1 eV was assigned to V4+ species. V acted as a co-catalytic composition in CeO2/VOx catalysts. In the oxidation process, oxygen transferred from CeO2 to VOx and promoted the rate of a cyclic catalytic reaction. Appropriate V4+ icons in low valance were beneficial to improve the activity of the catalysts and ensure the electron transfer faster in the process of the mutual transformation of V4+ and Ce4+.
(3)
The Ce-V-Ti catalysts were analyzed by FTIR spectra, and the results were shown in Fig.5. The absorption band in the 3 200~3 700 cm-1 could be attributed to O—H stretching vibration in hydroxyl, carboxylic and phenolic groups. The adsorption bands at 1 610~1 680 cm-1 could be attributed to C=C stretching vibrations in carboxylic or pyridine-like structure. The bands at 1 050~1 160 cm-1 could be attributed to the bending vibrations of C=O groups. It could be seen that the FT-IR spectra of O—H (3 417 cm-1), C—H (1 388, 1 455, 2 848 and 2 917 cm-1), C=O (1 064 cm-1) and C=C (1 631 cm-1) surface functional groups existed in Ce-V-Ti samples. The strong peaks at 3 417 cm-1 were O—H functional groups. The aliphatic C—H groups at 1 388, 1 455, 2 848 and 2 917 cm-1 were the most peaks[
![]()
Figure 5.FTIR spectra of the Ce-V/Ti catalysts
3 Conclusions
(1) The Ce-V-Ti catalysts synthesized by sol-gel method showed excellent catalytic activity in a simulating iron ore sintering flue gas (CB: 100 ppm; 16% O2 and 84% N2; GHSV: 30 000 h-1; catalyst amount: 200 mg). The catalytic conversion of CB over Ce(15)-V(2.5)-Ti catalysts can achieve 58% at 150 ℃ and 95% at 300 ℃.
(2) Ce played a major catalytic role and V acted as a co-catalytic composition. The chemical interaction between Ce and Ti can improve the number of oxygen vacancies. The solid solution reaction of Ce into Tiwas promoted by adding V. V4+ can help to promote the reduction reaction.
(3) The specific surface area of the Ce-V-Ti catalysts was increased. It was favorable to the dispersion of the active components on the surface of Ti. Most of the functional groups on the Ce-V-Ti catalysts were C—H groups and O—H, which was expedited the adsorption and desorption of CB.
References
[1] Q Cang D, L Zhang L, F Wei R. Iron and Steel, 49, 1(2014).
[2] Q Shi, L Zhang H, M Long H et al. Journal of Iron & Steel Research International, 25, 1(2018).
[3] X Qian L, M Long H, J Chun T et al. Process Safety Environmental Protection, 117, 82(2018).
[4] Wenrui Zhang, Aidong Tang, Jianliang Xue et al. Spectroscopy and Spectral Analysis, 36, 3075(2016).
[5] H Xiao Y, W Dai, H Cai G et al. Spectroscopy and Spectral Analysis, 36, 3062(2016).
[6] W Li W, F Yu M, D Li X et al. Chemosphere, 156, 383(2016).
[7] H Zhao, S Yang, F Dong et al. Molecular Catalysis, 463, 119(2019).
[8] Y Gu, H Huang, J Zhao et al. Journal of Catalysis, 326, 54(2015).
[9] Q Wu, K Liu, Y Chen. Chemosphere, 154, 472(2016).
[10] S Watanabe, C Song, X Ma. Journal of Physical Chemistry C, 113, 14249(2009).
[11] Z Wu, H Overbury S, J Rondinone A. Journal of Physical Chemistry C, 115, 25368(2011).
[12] Y Fang, H Zhang. Journal of Alloys and Compounds, 781, 201(2019).
Set citation alerts for the article
Please enter your email address



