- Opto-Electronic Advances
- Vol. 6, Issue 6, 220072 (2023)
Abstract
Keywords
Introduction
Fiber optic sensing technologies can realize the detection of nearly 100 types of parameters (e.g., displacement, acceleration, rotation, strain, temperature, refractive index (RI), current, magnetic field) based on the relationship between the characteristics of the transmitted light in the optical fiber and the surrounding parameters
The first branch is to reasonably and flexibly design fiber substrates. Substrates can employ commercial fibers, e.g., multi-mode fiber
In the field of life sciences, the research on the highly sensitive detection of biomarkers has great significance for the early screening of major diseases such as cancer. Fiber optic plasmonic resonance sensors are appropriate candidates for immunosensing based on antibody-antigen interactions. When the sensor is used to detect a type of antigen, the antibody coupling agent needs to be modified on the surface of the sensor to immobilize the corresponding antibody. The high RI sensitivity of the sensor can be converted into a signal response against the microscopic interaction between the antigen and the antibody via the coupling agent with excellent biocompatibility. However, the broadening and the redshift of the resonance dip is a general phenomenon for the antibody-antigen modification
Based on above discussions, a dispersion model that can provide a generic tool for configuring plasmonic resonance spectral characteristics is proposed and developed. According to the model, a reflective nano-modified fiber optic configurable plasmonic resonance (NMF-CPR) sensor combined with the tip hot spot effect is implemented for the measurement of the RI in the range of 1.3332–1.3432 with an interval of 0.0025. Coating the plasmon-active nanomaterials under the Au layer effectively avoids the large redshift of the resonance dip and the excessive decrease in the detection accuracy caused by the broadening of the FWHM, while significantly enhances the sensitivity of the sensor. Additionally, the dense Au layer prevents the oxidation and the shedding of nanomaterials. This allows the sensor to detect liquid samples for a long time while to still keep good stability. The reflective sensing structure can be directly inserted into samples to achieve the rapid and convenient detection. By establishing the quantitative relationship between the plasmonic resonance signal and the RI of the sample, the concentration of the solution that causes the change of the RI and the biochemical interaction between the sensitive layer and the specific substance in the solution can be quantitatively analyzed.
Results and discussions
Theoretical analyses
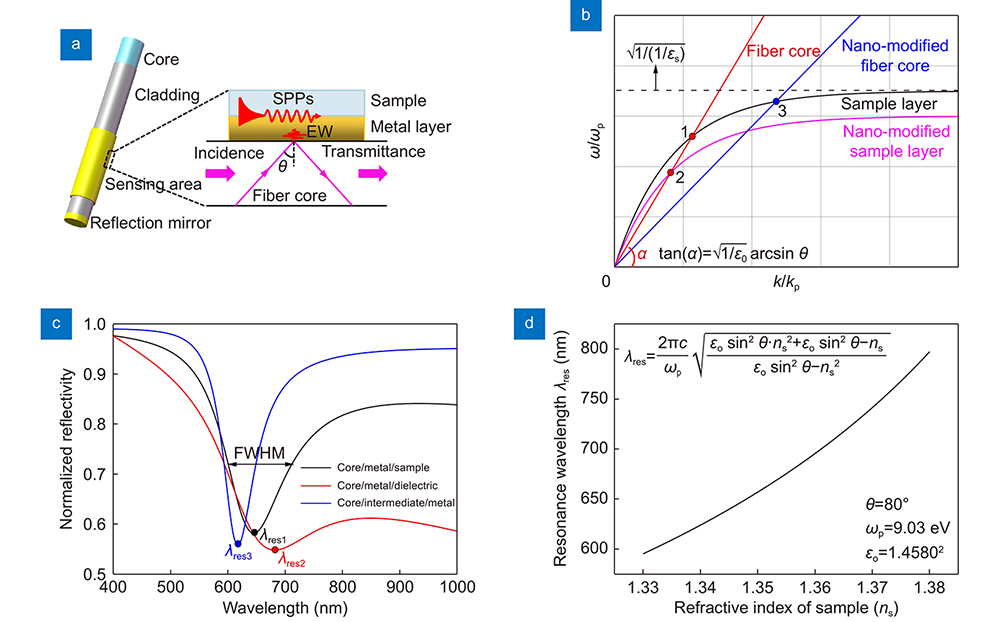
Principle of configuring plasmonic resonance spectral characteristics
The disassembly of the cylindrical surface in the optical fiber allows the plasmonic resonance at each point to be explained by a three-layer structure, as shown in
![]()
Figure 1.(
where
where
where
The intersection Point 1 of the two wavevector components in
When the fiber optic plasmonic resonance sensor is used for the multi-parameter detection, the transmission spectrum contains multiple resonance dips
Coating a dielectric layer with a high/complex dielectric constant on the upper surface of the metal layer in the conventional fiber plasmonic resonance sensor is equivalent to increasing the dielectric constant of the sample
NMF-CPR sensor/Tip hot spot enhanced NMF-CPR sensor
According to above discussions, an NMF-CPR sensor with a new-type sensing structure of fiber core/intermediate layer/metal layer/sample layer is designed, as shown in
![]()
Figure 2.Schematic of the sensing structure of (
We can achieve a higher sensitivity by doping a certain proportion of Au nanostars (AuNSs) into the WSe2 layer to construct a type of tip hot spot enhanced NMF-CPR sensor, as shown in
Numerical simulations
NMF-CPR sensor
The coupled-mode theory based finite element analysis (FEA)
![]()
Figure 3.(
It is found that the electric field intensity in the sample on the surface of the NMF-CPR sensor can reach up to 55.22 V/m, which is 1.45 times higher than that (38.17 V/m) of the conventional fiber plasmonic resonance sensor and this indicates an obvious enhancement of the surface field. Compared to the conventional fiber plasmonic resonance sensor, the initial loss peak (the loss peak corresponding to the RI of the pure water i.e., 1.3332) of the NMF-CPR sensor exhibits blueshift and narrowing. As the thickness of the Au layer increases, the initial loss peak of the NMF-CPR sensor exhibits redshift, the FWHM becomes larger, and the average sensitivity (the average of the sensitivity corresponding to five RI points of each sensor) of the sensor increases and is higher than that of the conventional fiber plasmonic resonance sensor. As the thickness of WSe2 layer increases, the average sensitivity of the sensor is improved. This demonstrates a greater contribution of WSe2 layer to the sensitivity enhancement. Above results agree well with theoretical analyses. The NMF-CPR sensor with a WSe2 layer of 10 nm and an Au layer of 50 nm shows the highest figure of merit (FOM, calculated via dividing the average sensitivity by the FWHM
Tip hot spot enhanced NMF-CPR sensor
As shown in
![]()
Figure 4.(
It can also be found from
Experimental results
NMF-CPR sensor
Experimental results are shown in
![]()
Figure 5.(
It can also be found that as the external RI increases, the sensitivity of the sensor is improved. This is because the RI matching degree between the nano-modified fiber core and the sample layer is enhanced due to the increment of the RI of the sample layer. When the RI of the sample layer is close to that of the modified fiber core, a symmetric plasmonic resonance excitation configuration is generated on both sides of the Au layer
Tip hot spot enhanced NMF-CPR sensor
The scattering of the light by AuNSs will increase the FWHM of the sensor and will reduce the detection accuracy
![]()
Figure 6.(
Additionally, the stability of the tip hot spot enhanced NMF-CPR sensor is investigated. Coating the dielectric layer on the upper surface of the plasmonic resonance sensor will cause the dielectric nanomaterials to easily fall off during the detection of liquid samples, which will lead to the change of the resonance wavelength and will reduce the stability of the sensor. The intermediate layer is under the Au layer in the tip hot spot enhanced NMF-CPR sensor in this work. The dense Au layer can avoid the shedding and the denaturation of the intermediate layer. The sensor has been used to detect pure water for 15 minutes, and the resonance wavelength is kept within in a stable range, as shown in
Conclusions
A dispersion model is developed to provide a general principle for the customized design of the plasmonic resonance curve according to specific detection requirements. According to the model, a tip hot spot enhanced reflective NMF-CPR sensor is proposed and applied to the measurement of the RI in a low range. Firstly, spectral characteristics of the NMF-CPR sensors are simulated based on the FEA. It is shown that the sensitivity and the FOM of the tip hot spot enhanced NMF-CPR sensor reach 3200.11 nm/RIU and 36.14 RIU–1, respectively, which are increased by 77.78% and 46.26% compared to those of the conventional fiber plasmonic resonance sensor. Secondly, the tip hot spot enhanced NMF-CPR sensor is practically implemented. Experimental results show that the sensitivity and the FOM of the sensor reach 2995.70 nm/RIU and 25.04 RIU–1, respectively, which are increased by 67.55% and 29.47% compared to those of the conventional fiber plasmonic resonance sensor. Thirdly, the wavelength resolution in the demodulation system and the detection resolution of the tip hot spot enhanced NMF-CPR sensor are up to 0.02 nm and 2.00×10–5 RIU, respectively. This suggests that the sensor still has a high detection accuracy while the sensitivity is significantly improved. Furthermore, the dense and uniform Au layer directly detects the sample, which avoids the drift of the resonance dip caused by the shedding of the modified nanomaterials. This thereby improves the stability of the sensor. The proposed sensor also provides better performance and easier operation compared to the reported works. The developed sensor in this work can be used as a highly sensitive refractometer and shows promising prospects for future applications in the scenarios of disease early screening, food safety detection and marine environmental monitoring.
Materials and methods
Materials and reagents
The multimode fiber with a 600-µm silica core and 630-µm plastic cladding, and a numerical aperture of 0.37 is purchased from Yangtze Optical Fibre and Cable Joint Stock Corp., Ltd. Wuhan, China. Hydrogen peroxide (H2O2, 30% wt.%), concentrated sulfuric acid (H2SO4, 98% wt.%) and Poly(diallyldimethylammonium chloride) (PDDA, 20 wt.%) are purchased from Aladdin Corp., Ltd. Shanghai, China. WSe2 nanosheets dispersion (1 layer–10 layers, lateral size: 50 nm–1000 nm, 0.5 mg/mL) and AuNSs dispersion (diameter: 40 nm–45 nm, 0.1 mg/mL) are purchased from Nanjing 2DNANO Tech. Corp., Ltd. Nanjing, China.
Fabrication of the NMF-CPR sensor
An endpoint-reflective fiber structure, that allows dual stimulation of the plasmonic resonance (stimulated by the incident light and the reflected light, respectively) and convenient detection, is used to construct sensors. The cladding at the ends of the fiber is removed as shown in Fig. S8(a-i), and the reflection area is separated from the sensing area to avoid the interference. A grinder has been utilized to grind the end face of the fiber into a plane, as shown in Fig. S9(a). An Ag layer of 200 nm is used as the reflector due to its higher reflectance (in Fig. S9(b)), and is sputtered on the reflection area followed by sputtering an Au layer of 5 nm to avoid the oxidation of the Ag layer, as shown in Fig. S8(a-ii). The sputtering of metal layers is achieved by a magnetron sputtering apparatus (MSP-3200, Shenyang LiNing Institute of Vacuum Technology, Shenyang, China). The thicknesses of metal layers are controlled by the crystal-calibrated sputtering rate and the sputtering time. A single Au layer is sputtered on the sensing area to complete the fabrication of the conventional fiber plasmonic resonance sensor. Figure S9(c) and S9(d) indicate that the Au layer is dense, uniform and smooth.
The sensing area is immersed in the piranha solution (H2O2∶H2SO4= 1∶3, v/v) for 30 minutes to complete surface hydroxylation and then immersed in 1 mg/mL PDDA solution for 30 minutes to make the area positively charged. A flat, dense and uniform WSe2 layer is the key factor to obtain the high-quality plasmonic resonance. Otherwise, the non-uniform WSe2 flakes will increase the radiation damping of SPPs and will thereby broaden the FWHM of the resonance curve. Therefore, the ultrasonic treatment of WSe2 nanosheets is required to guarantee the nanosheets to have a smaller and uniform size for better film-forming quality. As shown in Fig. S10(a) and S10(b), sizes of ultrasonic-dispersed WSe2 nanosheets are in the order of hundreds of nanometers, and nanosheets are negatively charged. The sensing area is immersed in WSe2 dispersion for 60 minutes, and the WSe2 nanosheets can be evenly attached to the surface of the area under the adsorption of positive and negative charges, as shown in Fig. S8(a-iii). The sensing area is rinsed with deionized water to remove the unattached WSe2 and then dried. Due to the saturation of the electrostatic adsorption, the thickness of the WSe2 layer can only be roughly adjusted in a small range around 10 nm by controlling the immersion time. Therefore, NMF-CPR sensors with WSe2 layers of approximately 10 nm have been developed in this work. An Au layer is sputtered on the surface of the WSe2-coated sensing area to complete the fabrication of the NMF-CPR sensor, as shown in Fig. S8(a-iv). The realistic image of the NMF-CPR sensor is shown in Fig. S8(b-i). The scanning electron microscope (SEM) images of the surface and the cross section of the sensing area of the NMF-CPR sensor with a WSe2 layer of approximately 10 nm and an Au layer of approximately 50 nm are shown in Fig. S10(c) and S10(d).
Fabrication of the tip hot spot enhanced NMF-CPR sensor
The fabrication of the tip hot spot enhanced NMF-CPR sensor is similar to that of the NMF-CPR sensor, as shown in Fig. S8(a-iii) and S8(a-iv). Figure S11(a) and S11(b) indicate that sizes of AuNSs are in the order of tens of nanometers which is smaller than those of WSe2 nanosheets, and AuNSs are positively charged. AuNSs can be evenly attached to the surface of negatively charged WSe2 nanosheets, and there are still a large number of hydroxyl groups in the mixture of WSe2 nanosheets and AuNSs (WSe2@AuNSs), as shown in Fig. S11(c) and S11(d). Therefore, the WSe2@AuNSs can also be attached to the PDDA functionalized fiber surface. The realistic image of the tip hot spot enhanced NMF-CPR sensor is shown in Fig. S8(b-ii). Specific fabrication processes of sensors are shown in Fig. S12.
Demodulation system
The demodulation system for the RI measurement is shown in Fig. S13. The light emitted from the light source (DH-2000, Ocean Insight, Inc. Florida, USA) enters the fiber optic sensor through a Y-type fiber patch cord. Then the light is reflected by the Ag reflector, and is collected by the spectrometer (Maya2000 Pro, Ocean Insight, Inc. Florida, USA). The insertion of the sensor into RI solutions prepared by glycerol and the withdrawal are controlled by a dip coater (SYDC-100, Shanghai SanYan Technology Corp., Ltd. Shanghai, China). The spectral signal is processed by the all-phase low-pass filtering algorithm
References
[1] T Liu, H Li, T He, CZ Fan, ZJ Yan et al. Ultra-high resolution strain sensor network assisted with an LS-SVM based hysteresis model. Opto-Electron Adv, 4, 200037(2021).
[2] BO Guan, L Jin, J Ma, YZ Liang, X Bai. Flexible fiber-laser ultrasound sensor for multiscale photoacoustic imaging. Opto-Electron Adv, 4, 200081(2021).
[3] RL Wang, HZ Zhang, QY Liu, F Liu, XL Han et al. Operando monitoring of ion activities in aqueous batteries with plasmonic fiber-optic sensors. Nat Commun, 13, 547(2022).
[4] C Caucheteur, T Guo, F Liu, BO Guan, J Albert. Ultrasensitive plasmonic sensing in air using optical fibre spectral combs. Nat Commun, 7, 13371(2016).
[5] C Lee, B Lawrie, R Pooser, KG Lee, C Rockstuhl et al. Quantum plasmonic sensors. Chem Rev, 121, 4743-4804(2021).
[6] Y Liu, W Peng. Fiber-optic surface Plasmon resonance sensors and biochemical applications: a review. J Lightwave Technol, 39, 3781-3791(2021).
[7] Y Zhao, RJ Tong, F Xia, Y Peng. Current status of optical fiber biosensor based on surface Plasmon resonance. Biosens Bioelectron, 142, 111505(2019).
[8] JY Jing, K Liu, JF Jiang, TH Xu, S Wang et al. Performance improvement approaches for optical fiber SPR sensors and their sensing applications. Photonics Res, 10, 126-147(2022).
[9] Y Singh, SK Raghuwanshi. Titanium dioxide (TiO2) coated optical fiber-based SPR sensor in near-infrared region with bimetallic structure for enhanced sensitivity. Optik, 226, 165842(2021).
[10] LK Li, YN Zhang, YF Zhou, WL Zheng, YT Sun et al. Optical fiber optofluidic bio-chemical sensors: a review. Laser Photonics Rev, 15, 2000526(2021).
[11] R Singh, S Kumar, FZ Liu, C Shuang, BY Zhang et al. Etched multicore fiber sensor using copper oxide and gold nanoparticles decorated graphene oxide structure for cancer cells detection. Biosens Bioelectron, 168, 112557(2020).
[12] GY Wang, Y Lu, LC Duan, JQ Yao. A refractive index sensor based on PCF with ultra-wide detection range. IEEE J Sel Top Quant, 27, 5600108(2021).
[13] A Urrutia, Villar I Del, P Zubiate, CR Zamarreño. A comprehensive review of optical fiber refractometers: toward a standard comparative criterion. Laser Photonics Rev, 13, 1900094(2019).
[14] LH Liu, XJ Zhang, Q Zhu, KW Li, Y Lu et al. Ultrasensitive detection of endocrine disruptors via superfine plasmonic spectral combs. Light Sci Appl, 10, 181(2021).
[15] SJ Weng, L Pei, JS Wang, TG Ning, J Li. High sensitivity D-shaped hole fiber temperature sensor based on surface Plasmon resonance with liquid filling. Photonics Res, 5, 103-107(2017).
[16] D Wang, W Li, QR Zhang, BQ Liang, ZK Peng et al. High-performance tapered fiber surface Plasmon resonance sensor based on the graphene/Ag/TiO2 layer. Plasmonics, 16, 2291-2303(2021).
[17] H Song, Q Wang, WM Zhao. A novel SPR sensor sensitivity-enhancing method for immunoassay by inserting MoS2 nanosheets between metal film and fiber. Opt Laser Eng, 132, 106135(2020).
[18] JY Jing, K Liu, JF Jiang, TH Xu, S Wang et al. Double-antibody sandwich immunoassay and plasmonic coupling synergistically improved long-range SPR biosensor with low detection limit. Nanomaterials, 11, 2137(2021).
[19] FC Chien, SJ Chen. A sensitivity comparison of optical biosensors based on four different surface Plasmon resonance modes. Biosens Bioelectron, 20, 633-642(2004).
[20] S Jain, A Paliwal, V Gupta, M Tomar. Smartphone integrated handheld Long Range Surface Plasmon Resonance based fiber-optic biosensor with tunable SiO2 sensing matrix. Biosens Bioelectron, 201, 113919(2022).
[21] AM Shrivastav, L Satish, A Kushmaro, V Shvalya, U Cvelbar et al. Engineering the penetration depth of nearly guided wave surface Plasmon resonance towards application in bacterial cells monitoring. Sensor Actuators B:Chem, 345, 130338(2021).
[22] XD Jiang, WR Xu, N Ilyas, MC Li, RK Guo et al. High-Performance coupled Plasmon waveguide resonance optical sensor based on SiO2: Ag film. Results Phys, 26, 104308(2021).
[23] JY Ma, K Liu, JF Jiang, TH Xu, S Wang et al. All optic-fiber coupled Plasmon waveguide resonance sensor using ZrS2 based dielectric layer. Opt Express, 28, 11280-11289(2020).
[24] LT Ji, SQ Yang, RN Shi, YJ Fu, J Su et al. Polymer waveguide coupled surface Plasmon refractive index sensor: a theoretical study. Photonic Sens, 10, 353-363(2020).
[25] MB Ross, JC Ku, B Lee, CA Mirkin, GC Schatz. Plasmonic metallurgy enabled by DNA. Adv Mater, 28, 2790-2794(2016).
[26] WJ Zhang, XL Zeng, A Yang, LP Teng, Y Zhu. Research on evanescent field ammonia detection with gold-nanosphere coated microfibers. Opto-Electron Eng, 48, 200451(2021).
[27] JY Jing, Q Zhu, ZX Dai, SY Li, Q Wang et al. Sensing self-referenced fiber optic long-range surface Plasmon resonance sensor based on electronic coupling between surface Plasmon polaritons. Appl Opt, 58, 6329-6334(2019).
[28] YL Yang, HJ Chen, XX Zou, XL Shi, WD Liu et al. Flexible carbon-fiber/semimetal Bi nanosheet arrays as separable and recyclable plasmonic photocatalysts and photoelectrocatalysts. ACS Appl Mater Interfaces, 12, 24845-24854(2020).
[29] AV Kabashin, P Evans, S Pastkovsky, W Hendren, GA Wurtz et al. Plasmonic nanorod metamaterials for biosensing. Nat Mater, 8, 867-871(2009).
[30] R Kant, R Tabassum, BD Gupta. Xanthine oxidase functionalized Ta2O5 nanostructures as a novel scaffold for highly sensitive SPR based fiber optic xanthine sensor. Biosens Bioelectron, 99, 637-645(2018).
[31] JN Chen, M Badioli, P Alonso-González, S Thongrattanasiri, F Huth et al. Optical nano-imaging of gate-tunable graphene plasmons. Nature, 487, 77-81(2012).
[32] Y Singh, MK Paswan, SK Raghuwanshi. Sensitivity enhancement of SPR sensor with the black phosphorus and graphene with Bi-layer of gold for chemical sensing. Plasmonics, 16, 1781-1790(2021).
[33] LX Liu, K Ye, ZY Jia, TY Xue, AM Nie et al. High-sensitivity and versatile plasmonic biosensor based on grain boundaries in polycrystalline 1L WS2 films. Biosens Bioelectron, 194, 113596(2021).
[34] Q Wu, NB Li, Y Wang, YC Xu, JD Wu et al. Ultrasensitive and selective determination of carcinoembryonic antigen using multifunctional ultrathin amino-functionalized Ti3C2-MXene nanosheets. Anal Chem, 92, 3354-3360(2020).
[35] S Yang, W Bao, XZ Liu, J Kim, RK Zhao et al. Subwavelength-scale lasing perovskite with ultrahigh Purcell enhancement. Matter, 4, 4042-4050(2021).
[36] A Shalabney, I Abdulhalim. Sensitivity-enhancement methods for surface Plasmon sensors. Laser Photonics Rev, 5, 571-606(2011).
[37] Y Zhao, M Lei, SX Liu, Q Zhao. Smart hydrogel-based optical fiber SPR sensor for pH measurements. Sensor Actuators B:Chem, 261, 226-232(2018).
[38] ZH Zhao, Q Wang. Gold nanoparticles (AuNPs) and graphene oxide heterostructures with gold film coupling for an enhanced sensitivity surface Plasmon resonance (SPR) fiber sensor. Instrum Sci Technol, 50, 530-542(2022).
[39] VG Kravets, AV Kabashin, WL Barnes, AN Grigorenko. Plasmonic surface lattice resonances: a review of properties and applications. Chem Rev, 118, 5912-5951(2018).
[40] S Shi, LB Wang, RX Su, BS Liu, RL Huang et al. A polydopamine-modified optical fiber SPR biosensor using electroless-plated gold films for immunoassays. Biosens Bioelectron, 74, 454-460(2015).
[41] F Chiavaioli, CAJ Gouveia, PAS Jorge, F Baldini. Towards a uniform metrological assessment of grating-based optical fiber sensors: from refractometers to biosensors. Biosensors, 7, 23(2017).
[42] XZ Zhang, BY Yang, JF Jiang, K Liu, XJ Fan et al. Side-polished SMS based RI sensor employing macro-bending perfluorinated POF. Opto-Electron Adv, 4, 200041(2021).
[43] B Dastmalchi, P Tassin, T Koschny, CM Soukoulis. A new perspective on plasmonics: confinement and propagation length of surface plasmons for different materials and geometries. Adv Opt Mater, 4, 177-184(2016).
[44] WL Zheng, YN Zhang, LK Li, XG Li, Y Zhao. A plug-and-play optical fiber SPR sensor for simultaneous measurement of glucose and cholesterol concentrations. Biosens Bioelectron, 198, 113798(2022).
[45] C Ciracì, RT Hill, JJ Mock, Y Urzhumov, AI Fernández-Domínguez et al. Probing the ultimate limits of plasmonic enhancement. Science, 337, 1072-1074(2012).
[46] Y Vasimalla, HS Pradhan, RJ Pandya. SPR performance enhancement for DNA hybridization employing black phosphorus, silver, and silicon. Appl Opt, 59, 7299-7307(2020).
[47] HG Gu, BK Song, MS Fang, YL Hong, XG Chen et al. Layer-dependent dielectric and optical properties of centimeter-scale 2D WSe2: evolution from a single layer to few layers. Nanoscale, 11, 22762-22771(2019).
[48] IH Malitson. Interspecimen comparison of the refractive index of fused silica. J Opt Soc Am, 55, 1205-1209(1965).
[49] M Krüger, M Schenk, P Hommelhoff. Attosecond control of electrons emitted from a nanoscale metal tip. Nature, 475, 78-81(2011).
[50] Q Chen, L Liang, QL Zheng, YX Zhang, L Wen. On-chip readout plasmonic mid-IR gas sensor. Opto-Electron Adv, 3, 190040(2020).
[51] Q Wu, Y Sun, D Zhang, S Li, Y Zhang et al. Ultrasensitive magnetic field-assisted surface Plasmon resonance immunoassay for human cardiac troponin I. Biosens Bioelectron, 96, 288-293(2017).
[52] JS Jensen, O Sigmund. Topology optimization for Nano-photonics. Laser Photonics Rev, 5, 308-321(2011).
[53] M Xue, K Liu, T Wang, PX Chang, JF Jiang et al. Single mode fiber SPR refractive index sensor based on U-shaped structure. Acta Photonica Sin, 46, 1006004(2017).
[54] EM You, YQ Chen, J Yi, ZD Meng, Q Chen et al. Nanobridged rhombic antennas supporting both dipolar and high-order plasmonic modes with spatially superimposed hotspots in the mid-infrared. Opto-Electron Adv, 4, 210076(2021).
[55] TS Wu, Y Shao, Y Wang, SQ Cao, WP Cao et al. Surface Plasmon resonance biosensor based on gold-coated side-polished hexagonal structure photonic crystal fiber. Opt Express, 25, 20313-20322(2017).
[56] Y Shen, JH Zhou, TR Liu, YT Tao, RB Jiang et al. Plasmonic gold mushroom arrays with refractive index sensing figures of merit approaching the theoretical limit. Nat Commun, 4, 2381(2013).
[57] WQ Zhu, R Esteban, AG Borisov, JJ Baumberg, P Nordlander et al. Quantum mechanical effects in plasmonic structures with subnanometre gaps. Nat Commun, 7, 11495(2016).
[58] J Homola. Surface Plasmon resonance sensors for detection of chemical and biological species. Chem Rev, 108, 462-493(2008).
[59] XM Wang, CL Zhao, YR Wang, SZ Jin. A proposal of T-structure fiber-optic refractive index sensor based on surface Plasmon resonance. Opt Commun, 369, 189-193(2016).
[60] H Harutyunyan, ABF Martinson, D Rosenmann, LK Khorashad, LV Besteiro et al. Anomalous ultrafast dynamics of hot plasmonic electrons in nanostructures with hot spots. Nat Nanotechnol, 10, 770-774(2015).
[61] GQ Wang, KQ Wang, J Ren, S Ma, ZH Li. A novel doublet-based surface Plasmon resonance biosensor via a digital Gaussian filter method. Sensor Actuators B:Chem, 360, 131680(2022).
[62] Q Wang, JY Jing, WM Zhao, XC Fan, XZ Wang. A novel fiber-based symmetrical long-range surface Plasmon resonance biosensor with high quality factor and temperature self-reference. IEEE Trans Nanotechnol, 18, 1137-1143(2019).
[63] R Zakaria, NM Zainuddin, MASA Fahri, PM Thirunavakkarasu, SK Patel et al. High sensitivity refractive index sensor in long-range surface Plasmon resonance based on side polished optical fiber. Opt Fiber Technol, 61, 102449(2021).
[64] DL Hou, XX Ji, NN Luan, L Song, YS Hu et al. Surface Plasmon resonance sensor based on double-sided polished microstructured optical fiber with hollow core. IEEE Photonics J, 13, 6800408(2021).
[65] R Zakaria, NAM Zainuddin, SA Raya, SAK Alwi, T Anwar et al. Sensitivity comparison of refractive index transducer optical fiber based on surface Plasmon resonance using Ag, Cu, and bimetallic Ag-Cu layer. Micromachines, 11, 77(2020).
[66] NAM Zainuddin, MM Ariannejad, PT Arasu, SW Harun, R Zakaria. Investigation of cladding thicknesses on silver SPR based side-polished optical fiber refractive-index sensor. Results Phys, 13, 102255(2019).
[68] N Cennamo, D Massarotti, L Conte, L Zeni. Low cost sensors based on SPR in a plastic optical fiber for biosensor implementation. Sensors, 11, 11752-11760(2011).
[69] YZ Cao, JY Ma, K Liu, XD Huang, JF Jiang et al. Optical fiber SPR sensing demodulation algorithm based on all-phase filters. Acta Phys Sin, 66, 074202(2017).
[70] WC Jing, GH Wang, K Liu, YM Zhang, H Dong et al. Application of weighted wavelength algorithm on the demodulation of a fiber Bragg grating optical sensing system. J Optoelectron·Laser, 18, 1022-1025(2007).
Set citation alerts for the article
Please enter your email address



