Yu GUO, Xiaoqing JIANG, Hongmei WU, Yu XIAO, Dafu WU, Xin LIU. Preparation of 2-hydroxy-1-naphthalene Functionalized SBA-15 Adsorbent for the Adsorption of Chromium(III) Ions from Aqueous Solution [J]. Journal of Inorganic Materials, 2021, 36(11): 1163
Search by keywords or author
- Journal of Inorganic Materials
- Vol. 36, Issue 11, 1163 (2021)
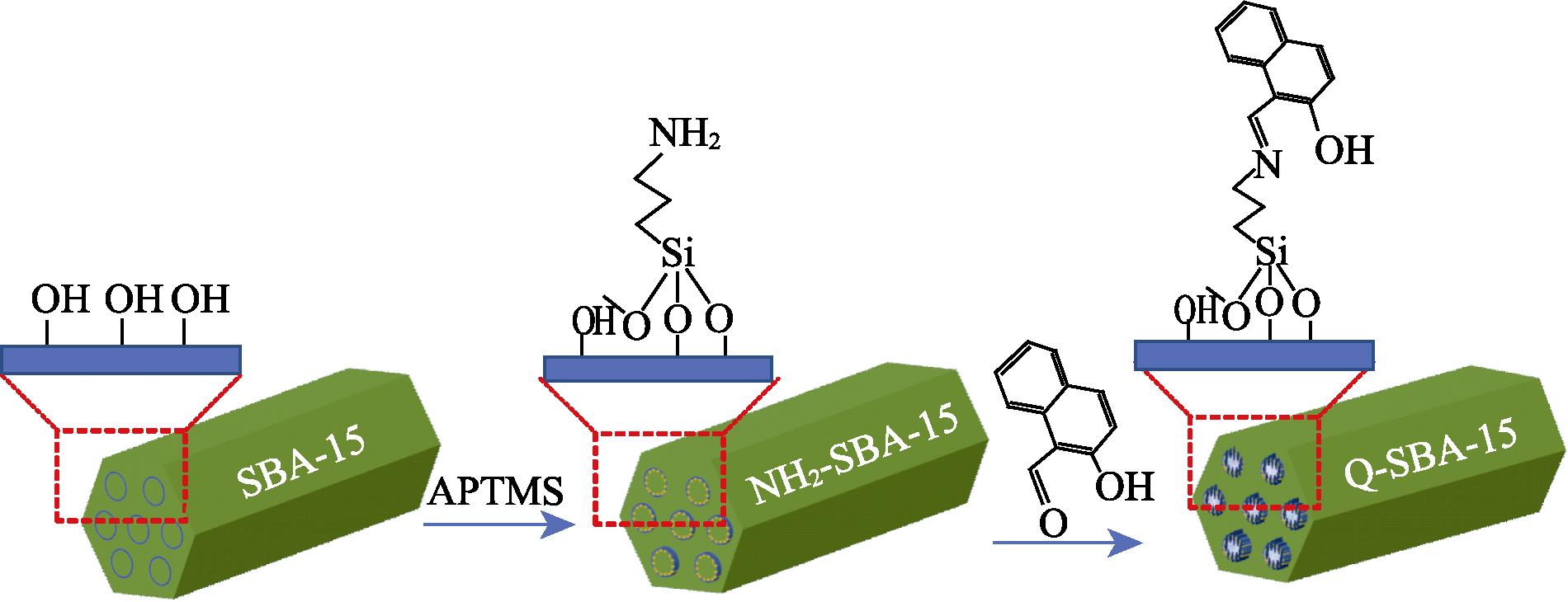
1. Schematic diagram of the synthetic process for Q-SBA-15
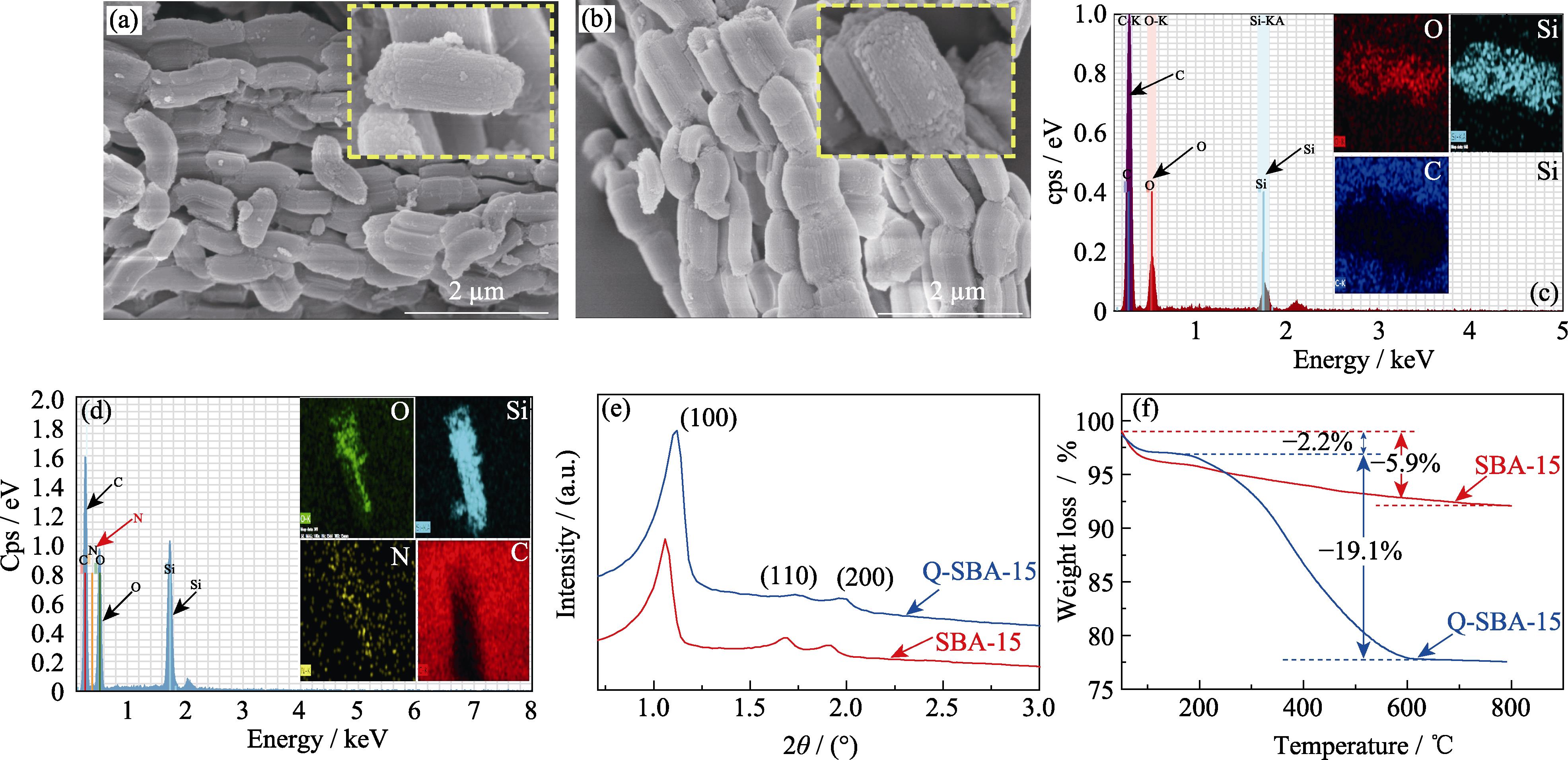
2. (a, b) SEM images, (c, d) EDX elemental analyses, (e) XRD patterns and (f) TG curves of (a, c) SBA-15 and (b, d) Q-SBA-15 samples
3. (a) N2 adsorption-desorption isotherms, (b) pore size distributions of SBA-15 and Q-SBA-15, (c, d) TEM images of Q-SBA-15
4. (a) FT-IR spectra of Q-SBA-15 and SBA-15, and (b) XPS survey spectrum of Q-SBA-15
5. Effect of solution pH on the adsorption of Cr(III) with Q-SBA-15
6. Effects of ionic strength on Cr(III) adsorption
7. Linearized fitting curves of (a) Langmuir model and (b) Freundlich model for adsorption of Cr(III) on Q-SBA-15
8. XPS spectra of Q-SBA-15
9. Regeneration property of Q-SBA-15
S1. (a) Effect of contacting time on the adsorption capacity of Cr(III) by Q-SBA-15 and (b) EDX analyses of Q-SBA-15 after adsorption of Cr(III)
S2. Fitting curves for adsorption of Cr(III) by (a) pseudo-first-order model and (b) pseudo-first-order model
S3. Plots of lnK cversus 1/T for the adsorption of Cr(III) by Q-SBA-15
|
Table 1. Textural properties of the synthesized samples
| ||||||||||||||||||||||
Table 1. Kinetic parameters for Cr(III) adsorption onto Q-SBA-15
|
Table 2. Comparison of Cr(III) adsorption performance with different materials selected from literature
| ||||||||||||||||||||||||||||||||||||||
Table 2. Different model parameters for adsorption of Cr(III) by Q-SBA-15 at different temperatures
|
Table 3. Thermodynamic parameters of Cr(III) adsorption on Q-SBA-15

Set citation alerts for the article
Please enter your email address



