Tingting WANG, Shumei SHI, Chenyuan LIU, Wancheng ZHU, Heng ZHANG. Synthesis of Hierarchical Porous Nickel Phyllosilicate Microspheres as Efficient Adsorbents for Removal of Basic Fuchsin[J]. Journal of Inorganic Materials, 2021, 36(12): 1330
Search by keywords or author
- Journal of Inorganic Materials
- Vol. 36, Issue 12, 1330 (2021)
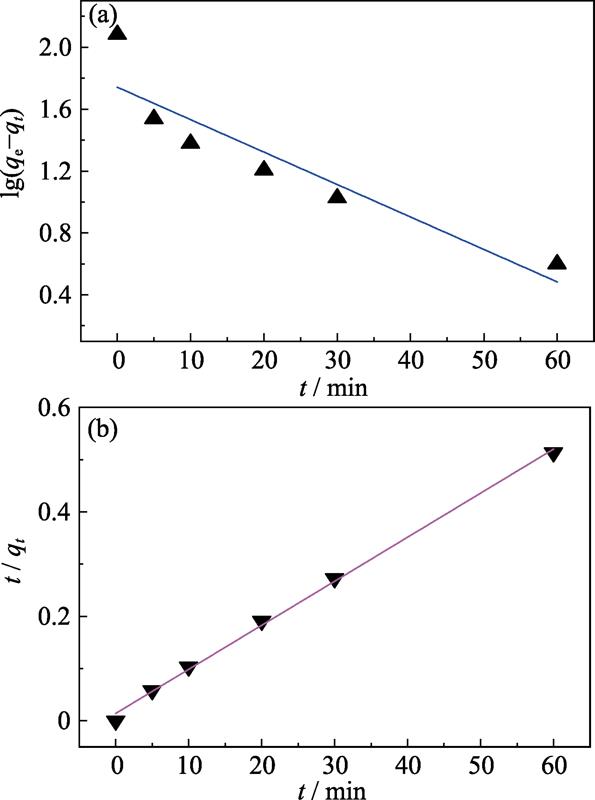
. Linear regression with pseudo-first-order (PFO) (a) and pseudo-second-order (PSO) (b) kinetic models
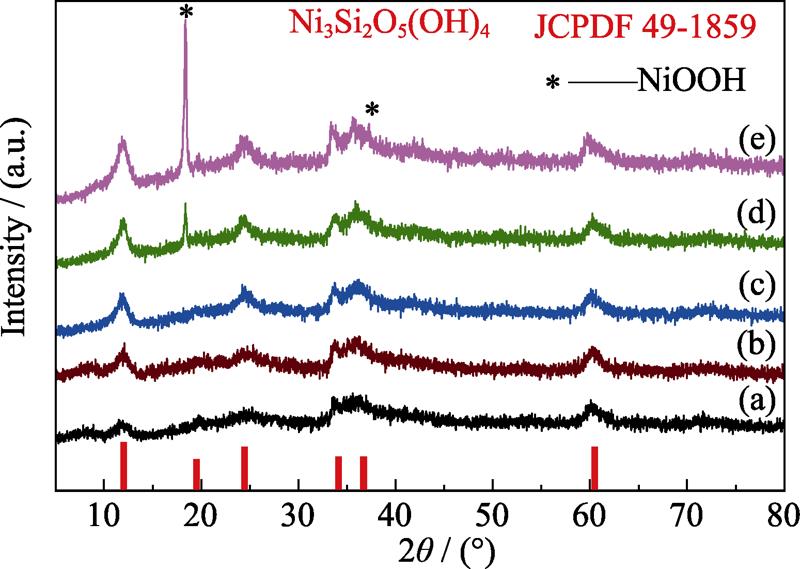
. Effect of Ni/Si molar ratio on the phase composition of the products hydrothermally synthesized at 210 ℃ for 12 h with different Ni/Si molar ratios
. SEM and TEM images of the Ni3Si2O5(OH)4 microspheres hydrothermally synthesized at 210 ℃ for 12 h with Ni/Si molar ratio of 1 : 1
. Adsorption isotherms of BF on the Ni3Si2O5(OH)4 microspheres fitted with the Langmuir (a) and Freundlich (b) isotherm models
. N2 adsorption-desorption isotherms of the products hydrothermally synthesized at 210 ℃ for 12 h with different Ni/Si molar ratios
. Pore size distribution derived from desorption (a) and adsorption (b) branch of the isotherm of the products hydrothermally synthesized with different Ni/Si molar ratios
. Effect of alkali source on composition and morphology of the products
. Schematic illustration for the formation of Ni3Si2O5(OH)4 microspheres
. Molecular structure of BF (a), Zeta potential of Ni3Si2O5(OH)4 microspheres (Ni/Si molar ratio of 1 : 1) (b), variation of the adsorption rate and capacity with adsorption time at different pH (Ni/Si molar ratio of 1 : 1) (c), and effect of Ni/Si molar ratio on the adsorption performance (d)
|
Table 0. Comparison of the adsorption capacities for BF on various adsorbents
|
Table 0. Textural properties of the products
| ||||||||||||||||||
Table 0. Corresponding fitting parameters originated from the non-linear regression by using Langmuir and Freundlich isotherm models
| ||||||||||||||||||||
Table 0. Adsorption kinetic model parameters for the adsorption of BF on the Ni3Si2O5(OH)4 microspheres

Set citation alerts for the article
Please enter your email address



