Peihui Li, Yijian Chen, Boyu Wang, Mengmeng Li, Dong Xiang, Chuancheng Jia, Xuefeng Guo. Single-molecule optoelectronic devices: physical mechanism and beyond[J]. Opto-Electronic Advances, 2022, 5(5): 210094
Search by keywords or author
- Opto-Electronic Advances
- Vol. 5, Issue 5, 210094 (2022)
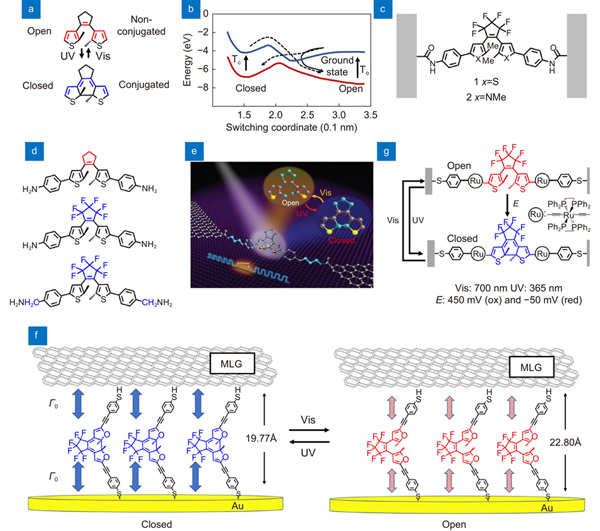
Fig. 1. Diarylethene units used in single-molecule switches. (a ) Diarylethene photoisomerization mechanism. (b ) Potential energy curves of the molecular switching. The switching process is initiated by an excitation to the first excited state. (c ) Diarylethene bridged between the electrode ends. (d ) Molecular structures with special design for single-molecule switches. (e ) A graphene-diarylethene-graphene single-molecule switch. (f ) Self-assembled monolayer devices with diarylethene units. (g ) Molecular isomerization under external controls of electrochemical potential and light irradiation. Figure reproduced with permission from: (a) ref.24, American Chemical Society; (b) ref.26, American Physical Society; (c) ref.27, American Chemical Society; (d) ref.28, John Wiley and Sones; (e) ref.7, American Association for the Advancement of Science; (f) ref.30, American Chemical Society; (g) ref.32, under a Creative Commons Attribution 3.0 Unported Licence.
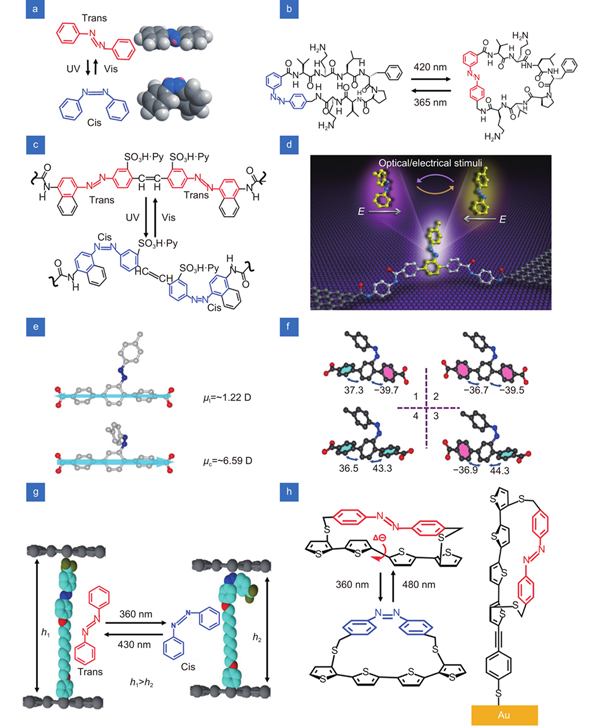
Fig. 2. Azobenzene units used in single-molecule switches. (a ) Structures of trans and cis isomers of azobenzene. (b ) Azobenzene as the core unit in the macromolecule. (c ) Chemical structures of molecules whose configurational change of the azobenzene unit occurs directly in the molecular backbone. (d ) Schematic of azobenzene as the side group of bridging molecule in the junction. (e ) Dipole projection on the molecular backbone. The arrow denotes the direction of the dipole projection. (f ) Four different conformational states due to the asymmetry caused by the introduction of additional azobenzene side group in cis form in the terphenyl backbone. (g ) Vertical distance between two graphene electrodes is regulated by the conformational changes in aryl azobenzene molecules with light irradiation. (h ) Vertical tunneling self-assembled monolayer device with an azobenzene derivative on Au electrode. Figure reproduced with permission from: (a, c) ref.33, (b) ref.34, (f) ref.36, John Wiley and Sons; (d, e) ref.35, under a Creative Commons Attribution 4.0 International License; (g) ref.37, Springer Nature; (h) ref.38, American Chemical Society.
Fig. 3. Other units used in single-molecule switches. (a ) DHP/CPD single-molecule junction. (b ) DHA/VHF single-molecule junction. (c ) SP/MC single-molecule junction. (d ) Schematic diagram of a vertical tunneling molecular switch with rGO thin films as the transparent top contact and the molecular structures of DHA and VHF. (e ) Single-molecule switches with chirality molecules. The spin-polarization direction of the current switches with the chirality inversion. (f ) Schematic diagram of SP SAMs in EGaIn/Ga2O3//SAM/AuTS junctions in their open and closed forms. Figure reproduced with permission from: (a) ref.39, American Chemical Society; (b) ref.40, under a Creative Commons Attribution 4.0 International License; (c) ref.41, American Chemical Society; (d) ref.43, John Wiley and Sons; (e) refs.19, 44, under a Creative Commons Attribution 4.0 International License; (f) ref.42, American Chemical Society.
Fig. 4. Photoconductance of single-molecule devices. (a ) Schematic illustration of the Au/NH-PTCDI-NH/Au junction under light irradiation. (b ) SAM-templated addressable nanogap devices comprised of AminoPyr or SC18. (c ) Schematic presentation of the charge transport of a molecular junction affected by the excitation. (d ) Bonding geometry of a porphyrin-C60 dyad molecule in the gold-ITO tunnel junction. (e ) Schematic diagram of carbon/bilayer/carbon tunneling junctions. (f ) Energy level diagram for a BTB–AQ bilayer junction. Figure reproduced with permission from: (a, c) ref.47, American Chemical Society; (b) ref.50, under a Creative Commons Attribution 4.0 International License; (d) ref.49, American Chemical Society; (e, f) ref.51, John Wiley and Sons.
Fig. 5. Photo-assisted transport mechanism in tunnelling junctions. (a ) A vacuum gold tunnelling junction. (b ) Plasmonic enhancement of the electromagnetic field in the junctions. (c ) Schematic representation of the mechanically controllable break fiber junction chip. The inset shows a zoomed view of the suspended fiber/Cr/Au bridge. (d ) Strong shift of the electrode energy level in imidazole single-molecule junctions caused by photon absorption. Figure reproduced with permission from: (a) ref.54, Springer Nature; (b) ref.55, American Chemical Society; (c, d) ref.56, The Royal Society of Chemistry.
Fig. 6. “Hot electron” effect of single-molecule devices. (a ) Schematic demonstration of an illuminated metal-molecule-metal junction with 4,4′-bipyridine (BP) molecule. (b ) Nonradiative decay of SPP with generated hot electrons and holes. (c ) Nonequilibrium distribution of hot electrons and holes in biased junctions. (d ) Experimental setup and strategy to map hot-carrier energy distributions. (e ) Schematic illustration of the real-time observation of the plasmon-induced chemical reaction. Figure reproduced with permission from: (a) ref.59, American Chemical Society; (b–d) ref.62, (e) ref.63, American Association for the Advancement of Science.
Fig. 7. Photovoltaic effect in single-molecule devices. (a ) A GaAs-molecule-Au molecular junction. (b ) Schematic band diagram for the illuminated metal-molecule-semiconductor junction under reverse biases. (c ) I–V characteristics of the Au-GaAs junction. Figure reproduced with permission from: (a, b) ref.66, American Chemical Society; (c) ref.67, The Royal Society of Chemistry.
Fig. 8. Electroluminescence of plasmon. (a ) Photon emission induced by inelastic tunneling through a nano-gap between a sharp Au tip and an Au substrate. (b ) Schematic diagram and mechanism diagram of how hot electrons excite plasmon electroluminescence. (c ) Illustration of the molecular tunneling junction with a SAM of SCn. (d ) Blinking of plasmon sources obtained from molecular tunneling junction with a SC12 SAM. (e ) Corresponding spectra of plasmon electroluminescence excited at different biases. Figure reproduced with permission from: (a) ref.71, American Chemical Society; (b) ref.72, (c–e) ref.73, Springer Nature.
Fig. 9. Electroluminescence from molecules in a STM nanocavity. (a ) A single ZnPc molecule in a STM nanocavity, where the Au (111) substrate is covered with sodium chloride. (b ) Schematic demonstration of the luminescence mechanism for a neutral ZnPc. (c ) STM-induced light emission spectrum (black line) of the ZnPc linear tetramer. (d ) Phosphorescence scanning tunnel luminescence map of the PTCDA/NaCl/Ag (111) system. (e ) Schematic images of the exciton formation mechanism; the arrows represent electrons. Arrows up and down represent the direction of electron spin. (f ) Peak characteristics of fluorescence and phosphorescence. Figure reproduced with permission from: (a, b) ref.77, American Association for the Advancement of Science; (c) ref.80, American Physical Society; (d−f) ref.82, Springer Nature.
Fig. 10. Coupling of molecular electroluminescence and plasmon nanocavity. (a ) Two different junction structures: on top of the molecule or in close proximity to the molecule. (b ) STM-induced luminescence of the situation that the tip is in close proximity to the molecule which cause Fano resonance. (c ) Excitation of molecular fluorescence through intermolecular energy transfer. (d ) Schematic illustration of the process of intermolecular energy transfer. (e ) Exciton splitting diagram for different coherent dipole–dipole coupling modes. Figure reproduced with permission from: (a, b) ref.83, under a Creative Commons Attribution 4.0 International License; (c, d) ref.86, (e) ref.87, Springer Nature.
Fig. 11. Electroluminescence in single-molecule junctions. (a ) A polythiophene molecular wire suspended between the metal substrate and the tip of STM. (b ) Fluorescent junctions with different emitting units suspending between the substrate and the tip of the STM by oligothiophene chains. (c ) A single graphene nanoribbon junction. (d ) Device structure of the nanotube–molecule–nanotube junction. (e ) Chemical structure of the molecule, which consists of a central 2,6-dibenzylamino core-substituted NDI chromophore (blue), two OPE rods (red) and phenanthrene anchor units (green). (f ) Energy-level model with the HOMO and LUMO molecular orbitals of the OPE and NDI subunits. Figure reproduced with permission from: (a) ref.90, American Physical Society; (b) ref.91, American Chemical Society; (c) ref.92, American Chemical Society; (d–f) ref.93, Springer Nature.

Set citation alerts for the article
Please enter your email address



