Ya-Nan Sun, Yan-Hui Wang, Le-Le Song, Hai-Bin Du, Xiao-Chun Wang, Lan-Lai He, Si-Zuo Luo, Qin Yang, Jing Leng, Fu-Chun Liu. Photoelectron imaging on vibrational excitation and Rydberg intermediate states in multi-photon ionization process of NH3 molecule[J]. Chinese Physics B, 2020, 29(9):
Search by keywords or author
- Chinese Physics B
- Vol. 29, Issue 9, (2020)
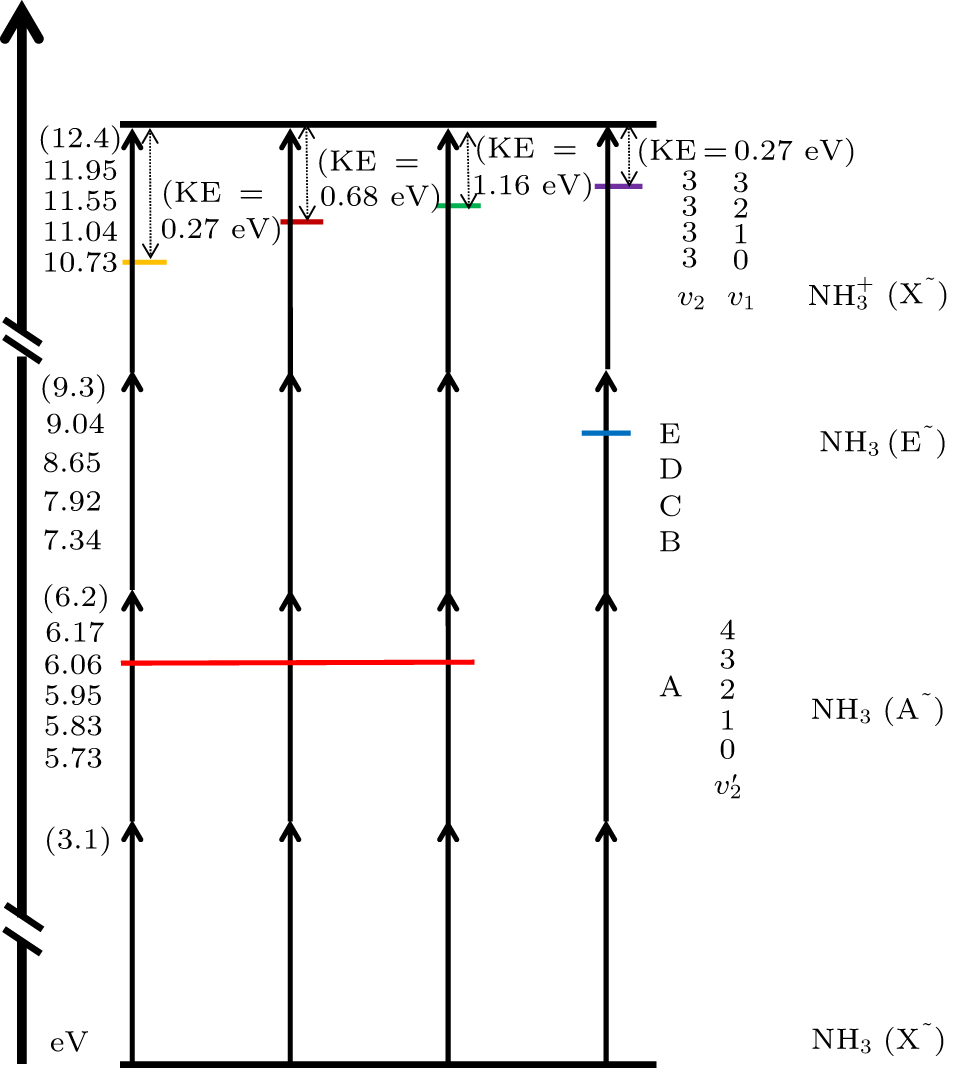
Fig. 1. Vibrational levels in different excited states of NH3 and NH 3 +
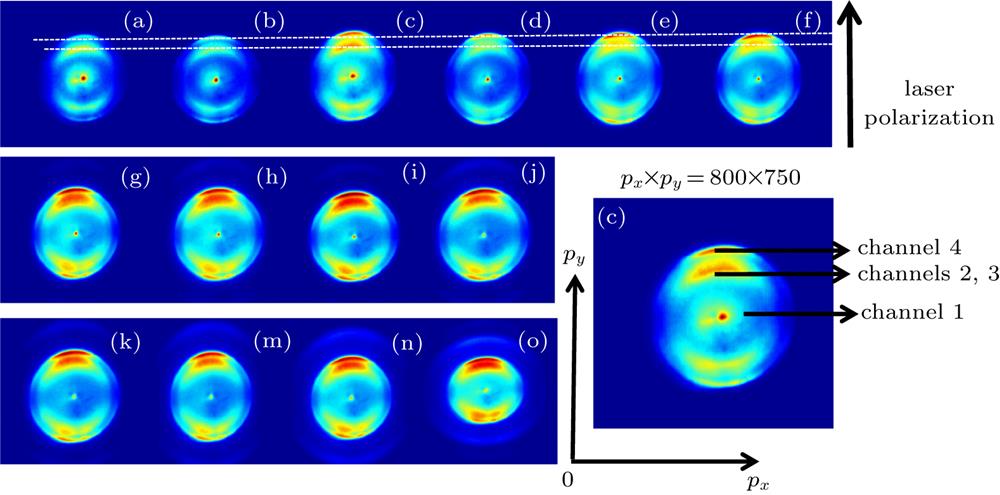
Fig. 2. Photoelectron images of NH3 molecules. The laser wavelength is 400 nm, and the intensity range is 1.01 × 1013 W/cm2–1.01 × 1014 W/cm2. (a)–(n) Corresponding photoelectron distribution images with the laser intensity of 1.01, 1.27, 1.52, 1.90, 2.53, 3.17, 3.80, 4.43, 5.07, 5.70, 6.30, 7.60, 8.87, 10.10 × (1013 W/cm2). Horizontal and vertical coordinates are x and y pixels of CCD, respectively, and the direction of laser polarization is represented by arrows.
Fig. 3. Kinetic energy distributions from slow electrons within the range of 0 eV–3.1 eV with laser intensities being 1.0, 1.27, 1.52, 1.90, 2.53, 3.17, 3.80, 4.43, 5.07, 5.70, 6.30, 7.60, 8.87, and 10.1 × (1013 W/cm2).
Fig. 4. Kinetic energy distributions of fast electrons within the range of 3.1 eV–10 eV with laser intensities being 1.01, 1.27, 1.52, 1.90, 2.53, 3.17, 3.80, 4.43, 5.07, 5.70, 6.30, 7.60, 8.87, and 10.1 × (1013 W/cm2).
Fig. 5. Angular distributions of peaks 1, 2, 3, and 4, corresponding to 1.01 × 1013 W/cm2 laser intensity.
Fig. 6. Angular distributions of peak 4 at different laser intensities.
Fig. 7. Angular distribution of peak 5 at laser intensity 5.07 × 1013 W/cm2.
Fig. 8. Angular distribution of peaks 4, 5, and 6 at the 5.07 × 1013-W/cm2 laser intensity.
Fig. 9. Results and identification for multi-photon processes of ammonia in the 400-nm wavelength laser field. Colored arrows in this picture illustrate different multi-photon processes; all photons have the 400-nm wavelength.
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 1. In non-ponderomotive force case, values of energy needed for ammonia molecule to transform from the X∼ state to different vibration levels of (upper) A state and (lower) ammonia ion.
Set citation alerts for the article
Please enter your email address



