- Journal of Semiconductors
- Vol. 41, Issue 12, 122201 (2020)
Abstract
1. Introduction
Bulk heterojunction (BHJ) solar cells have attracted extensive attention due to their lightweight, flexibility and potential low cost by solution and roll-to-roll print processes[
Herein, we focus on the π bridge engineering and design three small molecular donors consisting of the benzodithiophene (BDT) core, the cyano-ester ending group with branched alkyl chains to improve the solubility[
![]()
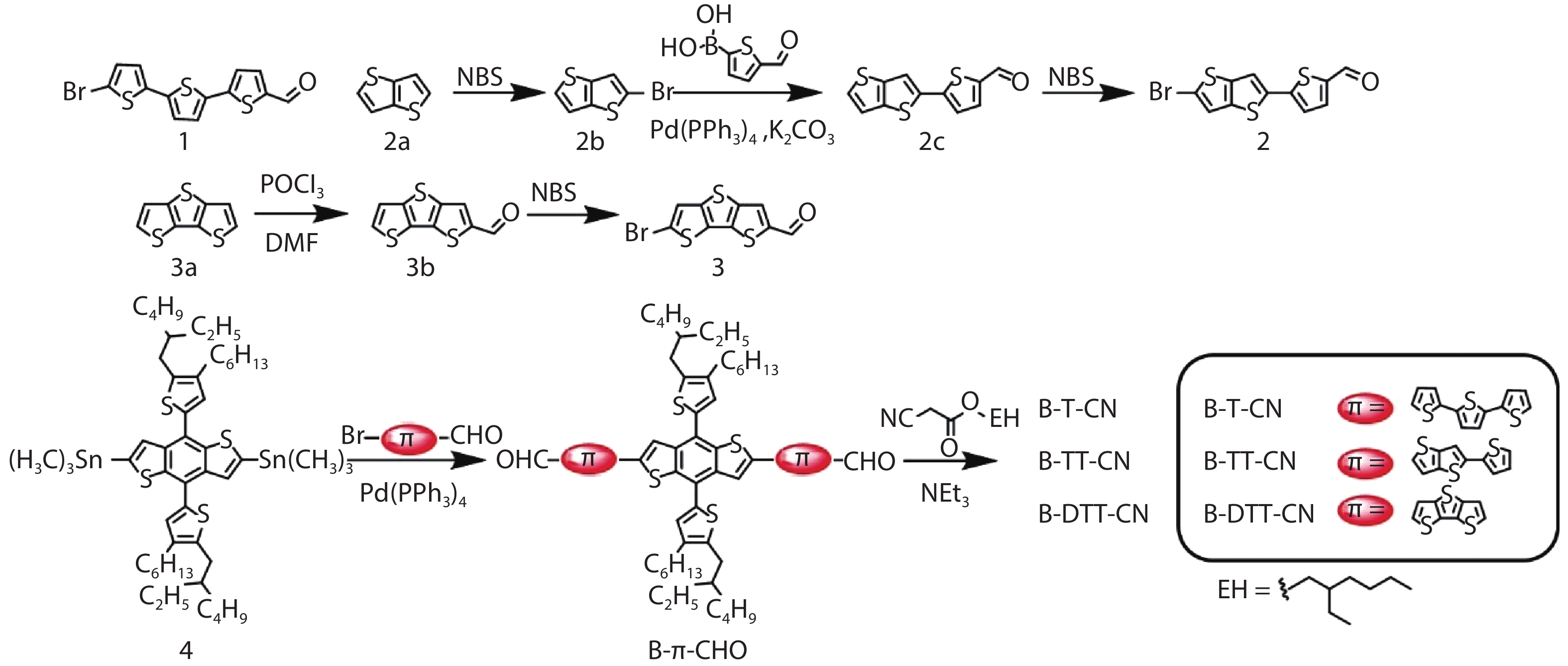
Figure 1.(Color online) Synthetic routes of B-T-CN, B-TT-CN, and B-DTT-CN.
2. Results and discussion
2.1. Synthesis and characterizations
The B-T-CN, B-TT-CN and B-DTT-CN were synthesized by n-bromosuccinimide bromination, Vilsmerier-Haack formylation, Suzuki and Stille coupling and Knoevenagel condensation, and the detailed synthetic routes are shown as Scheme 1 and in the Supporting Information. All of the intermediates and final products were fully characterized (Supporting Information). The thermal stabilities of three donors were measured by thermal gravimetric analysis (TGA) and the 5% weight loss decomposition temperatures are 341 °C for B-T-CN, 353 °C for B-TT-CN and 316 °C for B-DTT-CN, respectively (Fig. S2).
![]()
Figure 2.(Color online) (a) Film absorption of donor and acceptor materials. (b) The energy levels diagram of B-T-CN, B-TT-CN, B-DTT-CN, and PC71BM.
2.2. Optical and electrochemical properties
To obtain potential high Jsc values, the absorption of materials should cover from visible light to near infrared region[
![]()
Figure 3.(Color online) (a) The device structure of the all small molecule OSCs. (b) Representative current density versus applied voltage curves. (c) EQE spectra of the optimized devices.
2.3. Photovoltaic performance
To explore the potential photovoltaic properties of three small molecular donors, thin film BHJ SM OSCs were fabricated using a conventional device structure of indium tin oxide (ITO)/poly(3,4-ethylenedioxythiopene):poly(styrene sulfonate) (PEDOT:PSS)/B-T-CN or B-TT-CN or B-DTT-CN:PC71BM/Phen-NaDPO/Ag (Fig. 3(a), device area = 0.11 cm2). Three independent devices were primarily optimized through adjusting spin coating rotational speed and using solvent vapor annealing treatment (SVA). The champion devices of three materials were treated by tetrahydrofuran SVA for 10 s. The merits of B-T-CN:PC71BM, B-TT-CN:PC71BM, and B-DTT-CN:PC71BM based devices parameters are summarized in Table 2 which were tested under simulated AM 1.5G irradiation (100 mW/cm2) condition. Fig. 3(b) shows the best devices J–V curves. The Voc values of three devices (0.90 V for B-T-CN:PC71BM, 0.94 V for B-TT-CN:PC71BM, and 0.96 V for B-DTT-CN:PC71BM) are gradually increased as the fused ring expands, which demonstrates our strategy of adjusting π linking bridge to control molecular energy levels. In addition, the enhanced device Voc is reliable. The EQE spectra of three devices are shown in Fig. 3(c). The photoresponse range for three molecule-based devices is located from 350 to 670 nm which is in consistency with the film absorption. The B-T-CN:PC71BM based curve is higher than others in all EQE response region bring a higher Jsc for device (5.29 mA/cm2 for B-T-CN:PC71BM, 4.20 mA/cm2 for B-TT-CN:PC71BM, and 2.61 mA/cm2 for B-DTT-CN:PC71BM). Furthermore, The B-DTT-CN:PC71BM based device exhibits a significantly lower fill factor (FF) (39.8%) than others (55.7% for B-T-CN:PC71BM and 54.8% for B-TT-CN:PC71BM), which could be caused by the vast recombination of these devices[
![]()
Figure 4.(Color online) (a)
2.4. Charge recombination
Based on the optimal devices, we explored the charge recombination of each material based system to explain the difference of device properties. Firstly, we studied Jsc under different incident light intensities to evaluate the degree suffering from bimolecular recombination of devices. As previous studies, the dependence of current density on incident light intensity (I) obey to the power law equation as J ∝ Iα, where α represents the power factor. Briefly, fitting α value is between 0 to 1, the value more closes to 1 means the devices are less influenced by bimolecular recombination[
![]()
Figure 5.(Color online) Hole/electron mobility of optimized (a) B-T-CN:PC71BM film, (b) B-TT-CN:PC71BM film, and (c) B-DTT-CN:PC71BM film.
Furthermore, we measured the hole and electron carrier mobilities by space charge limited current (SCLC) method (Fig. 5). For hole-only device, the device structure is ITO/MoO3/active layer/MoO3/Ag, and electron-only cell was fabricated with the device architectures as ITO/ZnO/Phen-NaDPO/active layer/Phen-NaDPO/Ag. After blending with PC71BM, the B-T-CN:PC71BM exhibited both higher hole mobility of 2.55 × 10–5 cm2/(V·s) and electron mobility of 3.37 × 10–5 cm2/(V·s) than B-TT-CN:PC71BM (hole mobility of 1.25 × 10–5 cm2/(V·s) and electron mobility of 1.87 × 10–5 cm2/(V·s)) and B-DTT-CN:PC71BM (hole mobility of 9.14 × 10–6 cm2/(V·s) and electron mobility of 1.58 × 10–5 cm2/(V·s)). Even though the three small molecule donors have a similar chemical backbone, the blend mobilities are mainly affected by the film morphology which will be discussed in the next section. The ratio of μe and μh can evaluate the recombination of devices. As the conjugated fused rings of π bridges increase, the devices present a bigger μe/μh ratio as 1.73 for B-DTT-CN:PC71BM, 1.50 for B-TT-CN:PC71BM, and 1.32 for B-T-CN:PC71BM respectively. The more balanced mobility of B-T-CN:PC71BM could bring a lower recombination device, which is consistent with the fill factor result.
![]()
Figure 6.(Color online) Surface morphology of blend films. AFM height images of (a) B-T-CN:PC71BM blend film, (b) B-TT-CN:PC71BM blend film, and (c) B-DTT-CN:PC71BM blend film.
2.5. Blend morphology
As mentioned earlier, three devices are mainly affected by trap-assistant recombination. To better understand the charge recombination, we employed AFM and GIWAXS measurements to find the structure–properties relationship, and to analyze the active layers morphology and molecular stacking[
![]()
Figure 7.(Color online) GIWAXS two-dimensional diffraction patterns of (a) B-T-CN:PC71BM blend film, (b) B-TT-CN:PC71BM blend film, and (c) B-DTT-CN:PC71BM blend film. (d)The azimuthal angle distribution of π–π stacking.
We also employed GIWAXS to study the insight molecular stacking of blend films[
3. Conclusion
In summary, we have designed and synthesized three different type π linking bridge small molecular donors. By changing conjugated length, the small molecules achieved a deeper HOMO level, which result in a higher Voc for all SM OSCs. The Voc increased from 0.90 V enhance to 0.96 V and an overall PCE of 2.65% for B-T-CN:PC71BM based, 2.16% for B-TT-CN:PC71BM based and 1.00% for B-DTT-CN:PC71BM based devices was achieved. However, the blend morphology and molecular stacking are also changed by adjusting π linking bridges. When the π linking bridge from dithieno[2,3-b:2’,3’-D]thiophene (B-DTT-CN) change to alpha-terthiophene (B-T-CN), the blend morphology tends to present more clear interpenetrating network structures and the molecular packing becomes more uniform on the OOP detraction which could provide more moderate D/A interfaces for carrier desolation and more effective intermolecular charge transport tubes to ensure less trap recombination and increasedJsc. Our research provides a method to enhance OSC Voc, which could promote the development of high efficiency OSCs.
Acknowledgements
This work was supported by National Natural Science Foundation of China (21801238), National Youth Thousand Program Project (R52A199Z11), CAS Pioneer Hundred Talents Program B (Y92A010Q10) and Organic Semiconductor Center of Chongqing Institute of Green and Intelligent Technology, Chinese Academy of Sciences.
References
[1] L Y Lu, T Y Zheng, Q H Wu et al. Recent advances in bulk heterojunction polymer solar cells. Chem Rev, 115, 12666(2015).
[2] F W Zhao, S X Dai, Y Wu et al. Single-junction binary-blend nonfullerene polymer solar cells with 12.1% efficiency. Adv Mater, 29, 1700144(2017).
[3] Y Z Lin, X W Zhan. Oligomer molecules for efficient organic photovoltaics. Acc Chem Res, 49, 175(2016).
[4] J Hou, O Inganäs, R H Friend et al. Organic solar cells based on non-fullerene acceptors. Nat Mater, 17, 119(2018).
[5] Q S Liu, Y F Jiang, K Jin et al. 18% efficiency organic solar cells. Sci Bull, 65, 272(2020).
[6] Y Cui, H F Yao, J Q Zhang et al. Single-junction organic photovoltaic cells with approaching 18% efficiency. Adv Mater, 32, 1908205(2020).
[7] Y S Chen, X J Wan, G K Long. High performance photovoltaic applications using solution-processed small molecules. Acc Chem Res, 46, 2645(2013).
[8] S D Collins, N A Ran, M C Heiber et al. Small is powerful: Recent progress in solution-processed small molecule solar cells. Adv Energy Mater, 7, 1602242(2017).
[9] Y Huo, H L Zhang, X W Zhan. Nonfullerene all-small-molecule organic solar cells. ACS Energy Lett, 4, 1241(2019).
[10] Z C Zhou, S J Xu, J N Song et al. High-efficiency small-molecule ternary solar cells with a hierarchical morphology enabled by synergizing fullerene and non-fullerene acceptors. Nat Energy, 3, 952(2018).
[11] J Yuan, Y Q Zhang, L Y Zhou et al. Single-junction organic solar cell with over 15% efficiency using fused-ring acceptor with electron-deficient core. Joule, 3, 1140(2019).
[12] X Y Dong, K Yang, H Tang et al. Improving molecular planarity by changing alky chain position enables 12.3% efficiency all-small-molecule organic solar cells with enhanced carrier lifetime and reduced recombination. Sol RRL, 4, 1900326(2020).
[13] Q H Yue, H Wu, Z C Zhou et al. 13.7% efficiency small-molecule solar cells enabled by a combination of material and morphology optimization. Adv Mater, 31, 1904283(2019).
[14] J F Ge, L C Xie, R X Peng et al. 13.34% efficiency non-fullerene all-small-molecule organic solar cells enabled by modulating the crystallinity of donors via a fluorination strategy. Angew Chem Int Ed, 59, 2808(2020).
[15] J Gao, J F Ge, R X Peng et al. Over 14% efficiency nonfullerene all-small-molecule organic solar cells enabled by improving the ordering of molecular donors via side-chain engineering. J Mater Chem A, 8, 7405(2020).
[16] H Y Chen, D Q Hu, Q G Yang et al. All-small-molecule organic solar cells with an ordered liquid crystalline donor. Joule, 3, 3034(2019).
[17] Y S Liu, X J Wan, F Wang et al. High-performance solar cells using a solution-processed small molecule containing benzodithiophene unit. Adv Mater, 23, 5387(2011).
[18] B Kan, Q Zhang, M M Li et al. Solution-processed organic solar cells based on dialkylthiol-substituted benzodithiophene unit with efficiency near 10%. J Am Chem Soc, 136, 15529(2014).
[19] J Y Zhou, X J Wan, Y S Liu et al. Small molecules based on benzo[1, 2-b: 4, 5-b’]dithiophene unit for high-performance solution-processed organic solar cells. J Am Chem Soc, 134, 16345(2012).
[20] W Ni, M M Li, X J Wan et al. A high-performance photovoltaic small molecule developed by modifying the chemical structure and optimizing the morphology of the active layer. RSC Adv, 4, 31977(2014).
[21] S L Shen, P Jiang, C He et al. Solution-processable organic molecule photovoltaic materials with bithienyl-benzodithiophene central unit and indenedione end groups. Chem Mater, 25, 2274(2013).
[22] K Sun, Z Xiao, S Lu et al. A molecular nematic liquid crystalline material for high-performance organic photovoltaics. Nat Commun, 6, 6013(2015).
[23] B Qiu, L Xue, Y Yang et al. All-small-molecule nonfullerene organic solar cells with high fill factor and high efficiency over 10%. Chem Mater, 29, 7543(2017).
[24] H J Bin, J Yao, Y K Yang et al. High-efficiency all-small-molecule organic solar cells based on an organic molecule donor with alkylsilyl-thienyl conjugated side chains. Adv Mater, 30, 1706361(2018).
[25] Y Wang, B Liu, C W Koh et al. Facile synthesis of polycyclic aromatic hydrocarbon (PAH)-based acceptors with fine-tuned optoelectronic properties: Toward efficient additive-free nonfullerene organic solar cells. Adv Energy Mater, 9, 1803976(2019).
[26] J H Wan, X P Xu, G J Zhang et al. Highly efficient halogen-free solvent processed small-molecule organic solar cells enabled by material design and device engineering. Energy Environ Sci, 10, 1739(2017).
[27] D Deng, Y J Zhang, J Q Zhang et al. Fluorination-enabled optimal morphology leads to over 11% efficiency for inverted small-molecule organic solar cells. Nat Commun, 7, 13740(2016).
[28] T N Duan, M Babics, A Seitkhan et al. F-Substituted oligothiophenes serve as nonfullerene acceptors in polymer solar cells with open-circuit voltages >1 V. J Mater Chem A, 6, 9368(2018).
[29] D X Liu, B Kan, X Ke et al. Extended conjugation length of nonfullerene acceptors with improved planarity via noncovalent interactions for high-performance organic solar cells. Adv Energy Mater, 8, 1801618(2018).
[30] Q Xie, X F Liao, L Chen et al. Random copolymerization realized high efficient polymer solar cells with a record fill factor near 80%. Nano Energy, 61, 228(2019).
[31] C M Proctor, M Kuik, T Q Nguyen. Charge carrier recombination in organic solar cells. Prog Polym Sci, 38, 1941(2013).
[32] S R Cowan, A Roy, A J Heeger. Recombination in polymer-fullerene bulk heterojunction solar cells. Phys Rev B, 82, 245207(2010).
[33] T Kirchartz, F Deledalle, P S Tuladhar et al. On the differences between dark and light ideality factor in polymer: Fullerene solar cells. J Phys Chem Lett, 4, 2371(2013).
[34] S Wheeler, F Deledalle, N Tokmoldin et al. Influence of surface recombination on charge-carrier kinetics in organic bulk heterojunction solar cells with nickel oxide interlayers. Phys Rev Appl, 4, 024020(2015).
[35] R Z Liang, M Babics, V Savikhin et al. Carrier transport and recombination in efficient “all-small-molecule” solar cells with the nonfullerene acceptor IDTBR. Adv Energy Mater, 8, 1800264(2018).
[36] F W Zhao, C R Wang, X W Zhan. Morphology control in organic solar cells. Adv Energy Mater, 8, 1703147(2018).
[37] T N Duan, J Gao, T L Xu et al. Simple organic donors based on halogenated oligothiophenes for all small molecule solar cells with efficiency over 11%. J Mater Chem A, 8(2020).
[38] L Y Yang, S Q Zhang, C He et al. Modulating molecular orientation enables efficient non-fullerene small-molecule organic solar cells. Chem Mater, 30, 30(2018).
[39] V Vohra, K Kawashima, T Kakara et al. Efficient inverted polymer solar cells employing favourable molecular orientation. Nat Photonics, 9, 403(2015).
[40] J Jung, W Lee, C Lee et al. Controlling molecular orientation of naphthalenediimide-based polymer acceptors for high performance all-polymer solar cells. Adv Energy Mater, 6, 1600504(2016).
[41] S S Chen, H J Cho, J Lee et al. Modulating the molecular packing and nanophase blending via a random terpolymerization strategy toward 11% efficiency nonfullerene polymer solar cells. Adv Energy Mater, 7, 1701125(2017).
Set citation alerts for the article
Please enter your email address



