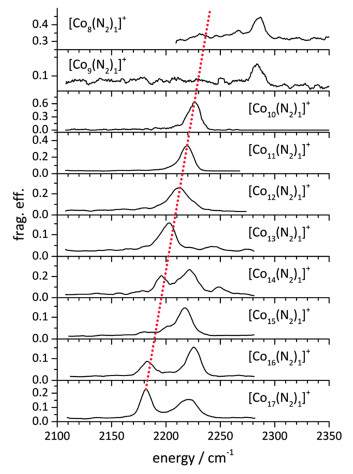
Li-hui Mou, Gui-duo Jiang, Zi-yu Li, Sheng-gui He. Activation of Dinitrogen by Gas-Phase Species†[J]. Chinese Journal of Chemical Physics, 2020, 33(5): 507
Search by keywords or author
- Chinese Journal of Chemical Physics
- Vol. 33, Issue 5, 507 (2020)
Abstract
Set citation alerts for the article
Please enter your email address