Xiaojing Sun, Ding Yuan, Chao Wei, Xiao Yang, Lin Li. Advances in the Study of Interfaces in Laser Additive Manufacturing of Multi‐Materials with Significant Differences in Physical Properties (Invited)[J]. Chinese Journal of Lasers, 2024, 51(1): 0102003
Search by keywords or author
- Chinese Journal of Lasers
- Vol. 51, Issue 1, 0102003 (2024)
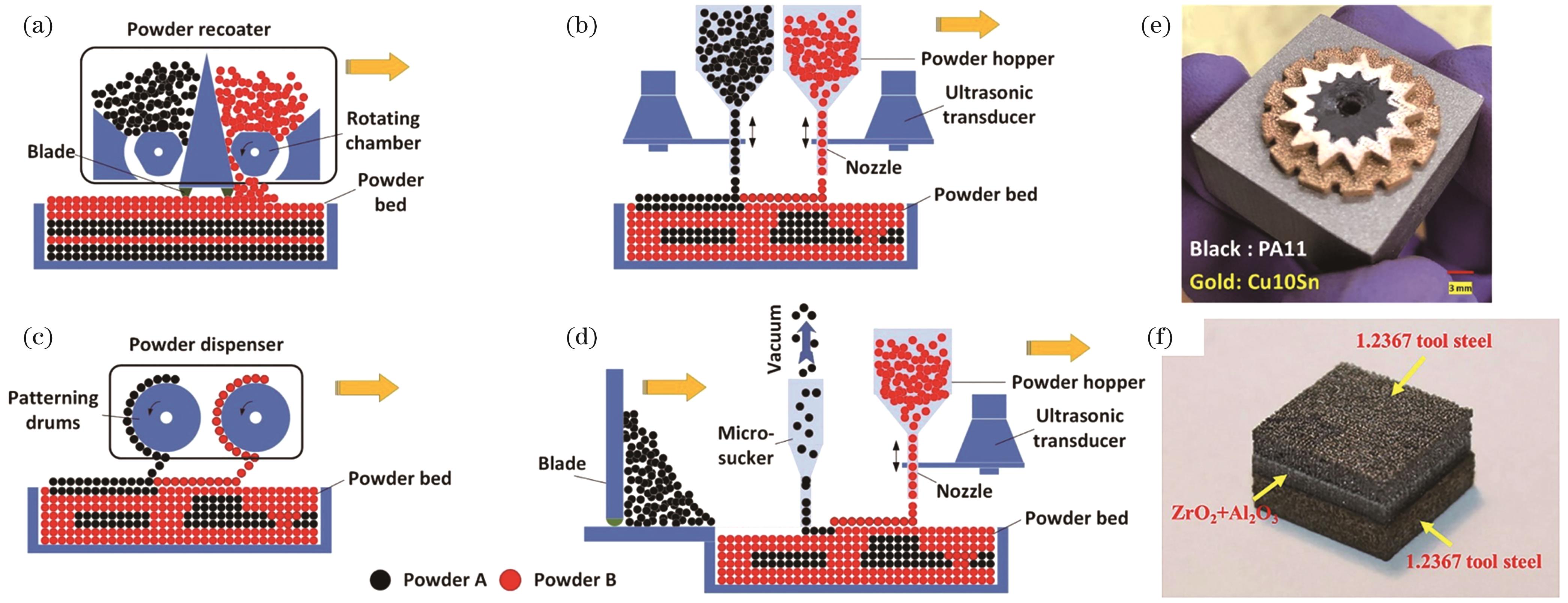
![Schematic diagrams of multi-material powder dropping/spreading methods[5,7,12] and material parts with significant differences in physical properties fabricated by L-PBF[5,13]. (a) Blade-based dual powder spreading; (b) ultrasonic-based dual powder dropping; (c) electrophotographic-based dual powder dropping; (d) “blade + ultrasonic” hybrid powder spreading; (e) polymer PA11-Cu10Sn heterostructure; (f) steel-ceramic heterostructure](/richHtml/zgjg/2024/51/1/0102003/img_01.jpg)
Fig. 1. Schematic diagrams of multi-material powder dropping/spreading methods[5,7,12] and material parts with significant differences in physical properties fabricated by L-PBF[5,13]. (a) Blade-based dual powder spreading; (b) ultrasonic-based dual powder dropping; (c) electrophotographic-based dual powder dropping; (d) “blade + ultrasonic” hybrid powder spreading; (e) polymer PA11-Cu10Sn heterostructure; (f) steel-ceramic heterostructure
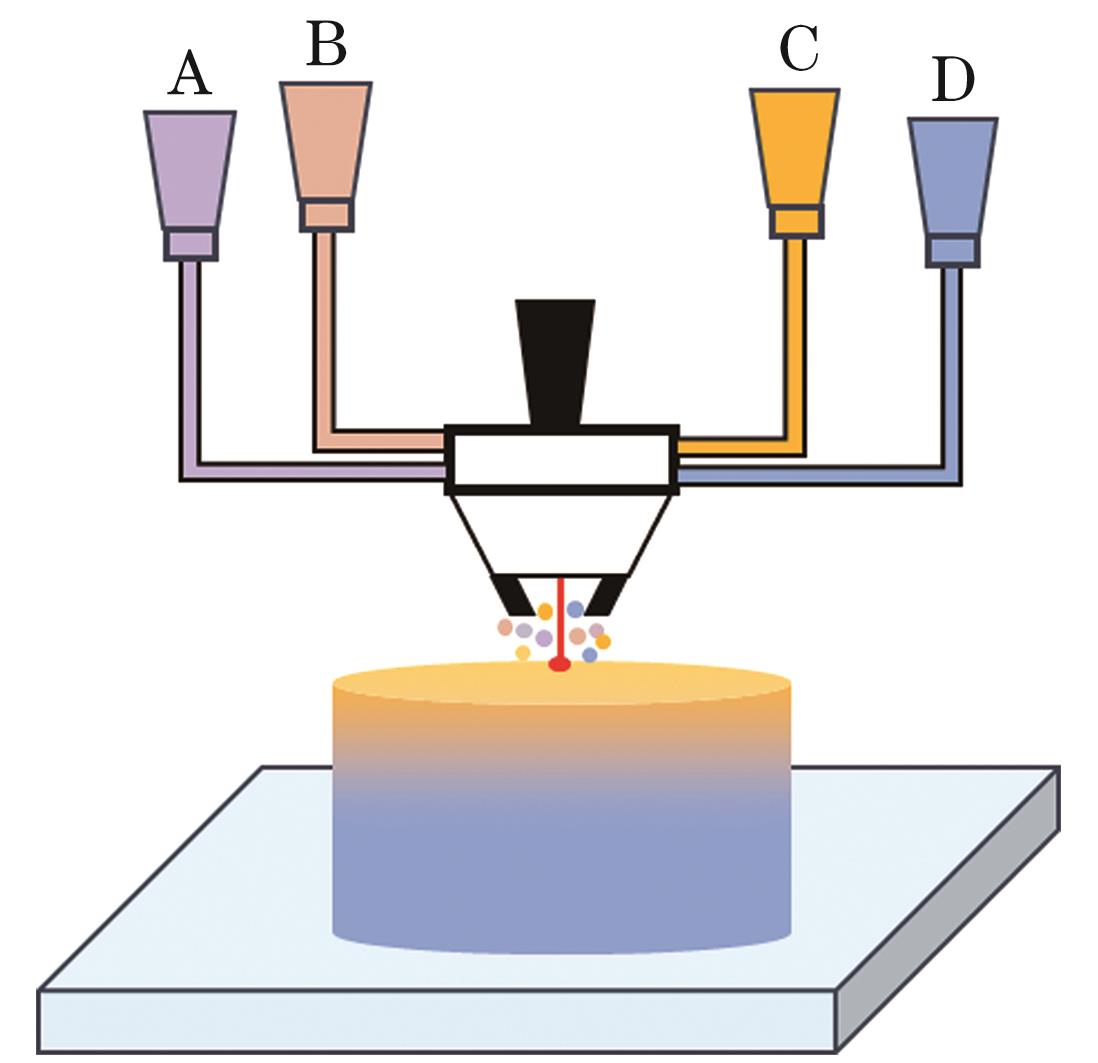
Fig. 2. Schematic diagram of a material formation system with laser powder-directed energy deposition
Fig. 3. Schematic diagram of laser induced forward transfer (LIFT) and the formed materials with significant differences in physical properties. (a) Schematic diagram of LIFT[20]; (b) graphene-nickel electrode structures[25]; (c) microstructure of Cu-MCMB-GPE-LiCoO2-Al[26]
Fig. 4. Schematic diagram of multiphoton fabrication process and formed metal-polymer material[27]. (a) Schematic diagram of multiphoton fabrication process; (b) polyimide resin substrate trenches; (c) silver-polyimide resin circuit components with significant differences in physical properties
Fig. 5. Schematic diagram of hybrid multi-material laser additive manufacturing (HMM-LAM) and the fabricated multiple materials with significant differences in physical properties[37]. (a) HMM-LAM system integrating fused filament fabrication (FFF) and L-PBF; (b)‒(c) metal-polymer components
Fig. 6. Interface cracking and defect issues of the materials with significant differences in physical properties. (a) 316L stainless steel-polymer PET heterostructure[37]; (b) Invar 36-V component gradient heterostructure[40]; (c) NiCr-YSZ heterostructure interface[41]; (d) Al-316L stainless steel heterostructure interface[42]; (e) Ti6Al4V-Al12Si component gradient heterostructure[17]
Fig. 7. Laser absorption rate of various materials. (a) Laser absorption rates of common materials at different wavelengths[43]; (b) laser absorption rate of PC and Cu at different wavelengths[44]
Fig. 8. Residual stress and strain due to local molten pool contraction
Fig. 11. Process optimization. (a) Process optimization of 316L SS-CuSn10 heterostructure interface[86]; (b) process optimization of Ti-Al materials with significant differences in physical properties fabricated by L-PBF, formation parameters optimization of AlSi10Mg[89]
Fig. 12. 1.2367 tool steel- ZrO2 and Al2O3 ceramic materials with significant differences in physical properties[95]. (a) Interface microstructure before laser re-melting; (b) interface microstructure after laser re-melting
Fig. 12. Interface optimization methods for materials with significant differences in physical properties
Fig. 13. Common functional graded design methods for heterogeneous materials
Fig. 14. Transition bonding. (a)‒(h) Macroscopic and microscopic morphology of direct bonding, mixture bonding, and transition bonding of Cu10Sn-W heterostructure formed by L-PBF[96]; (i)‒(l) microscopic morphology and deformation under compression load of Ti-Al gyroscope heterostructure with significant differences in physical properties formed by L-PBF[97]
Fig. 15. LAMed gradient materials with significant differences in physical properties. (a) Heterostructure of Fe-Al discontinuous gradient materials[98]; (b)(c) DED formed Fe-Al discontinuous gradient samples and the microstructures of various regions of the samples[98]; (d) Ti-Al continuous gradient materials[16]; (e)‒(g) microstructures of Ti-Al continuous gradient materials formed by L-PBF[16]
Fig. 16. Al-Ti heterostructure with significant differences in physical properties fabricated by the composite system of LAM+cold spraying[38]. (a)(c) Al-Ti6Al4V heterostructure with significant differences in physical properties and its interface morphology; (b)(d) Al+Al2O3 mixture-Ti6Al4V heterostructure with significant differences in physical properties and its interface morphology
Fig. 17. Simulation results of L-PBF materials with significant differences in physical properties. (a) Thermal boundary conditions of the calculation domain of 316L stainless steel-Cu10Sn heterointerface and simulation results of temperature field distribution at laser melting heterointerface and track morphology after solidification[87]; (b) simulation results of track morphology after solidification, component distribution and molten pool morphology of laser melted mixed IN718-Cu10Sn powder beds under different hatch spacings[90]; (c) finite element model of Ti6Al4V-TiB2 heterostructure and temperature distribution of the cross-section molten pool of laser melted TiB2 layer at different laser powers[91]
|
Table 1. Advantages and limitations of multi-material laser additive manufacturing technologies
| ||||||||||||||||||||||||||||||||||||||||||||||||||
Table 2. Thermophysical properties of typical metals in additive manufacturing
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 3. Interface optimization methods and related literatures for materials with significant differences in physical properties
| ||||||||||||||||||||||||
Table 4. Spectral absorptance for copper, aluminium, stainless steel and titanium at blue, green and infrared laser wavelengths[101]
Set citation alerts for the article
Please enter your email address



