Zemei PAN, Qiuping ZHANG, Man SONG, Huan YUAN, Yutong LIU, Yuanjie SU, Ming XU. Facile Synthesis of ZnO/TiO2 Nanocomposite Photocatalysts and Study of Their Photocatalytic Performance[J]. Acta Photonica Sinica, 2022, 51(4): 0416001
Search by keywords or author
- Acta Photonica Sinica
- Vol. 51, Issue 4, 0416001 (2022)
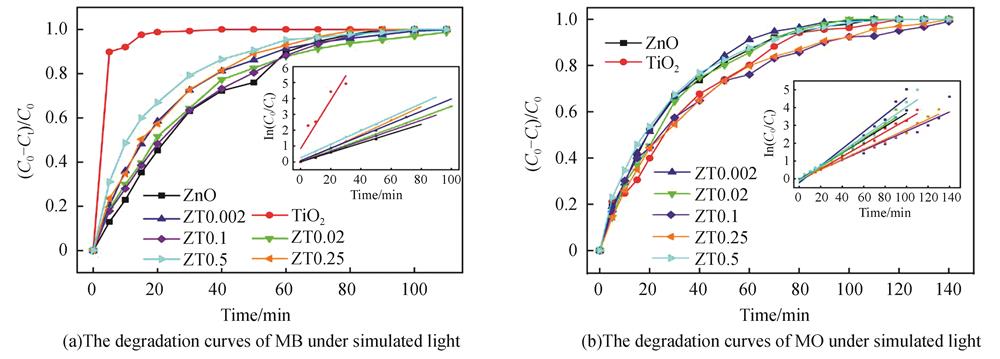
Fig. 1. Photodegradation curves of MB and MO of pure ZnO,commercial TiO2 and ZnO/TiO2 nanocomposites under simulated light(The inset is the fitting of the pseudo-first-order kinetic function)
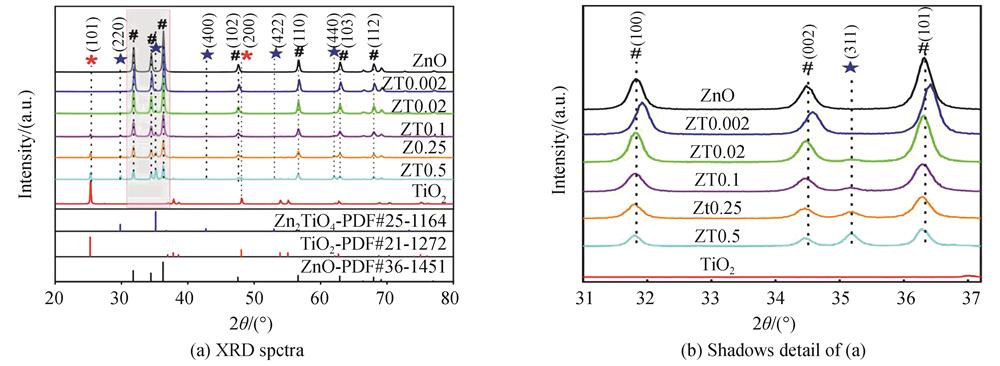
Fig. 2. XRD spectra of pure ZnO,commercial TiO2 and ZnO/TiO2 nanocomposites samples
Fig. 3. High-resolution TEM images and SAED images of sample ZT0.002 and ZT0.5
Fig. 4. SEM images of samples
Fig. 5. The particle size distribution
Fig. 6. XPS spectra of samples
Fig. 7. The oxygen content ratio diagram of samples
Fig. 8. Energy paramagnetic resonance spectra of the samples at room temperature
Fig. 9. UV-vis absorption spectra of samples
Fig. 10. The surface photovoltage spectra of samples
Fig. 11. Effect of different scavengers on the photocatalytic degradation of MB and MO over ZT0.002
Fig. 12. Photocatalytic schematic
Fig. 13. The efficiency curves of MB,MO degradation of ZT0.002 sample under simulated sunlight in different
|
Table 1. The weight percentages of elements Ti and Zn and mole percentages of Ti/(Ti+Zn)in the samples ZT0.002,ZT0.02,ZT0.1,ZT0.25,ZT0.5
|
Table 2. The performance parameters of pure ZnO,commercial TiO2 and ZnO/TiO2 nanocomposites degradation of MB and MO under simulated light
|
Table 3. Photodegradation of different semiconductor composite photocatalysts on organic pollutants
|
Table 4. The parameters of pure ZnO,commercial TiO2 and ZnO/TiO2 nanocomposites

Set citation alerts for the article
Please enter your email address



