Xinxin FENG, Xuanzhi MAO, Mingxing ZHANG, Lijun HU, Hongwei HAN, Zhe XING, Rong LI, Guozhong WU. Synthesis of functional nonwoven fabric using radiation grafting for the selective adsorption of Au(Ⅲ) in an acidic solution[J]. Journal of Radiation Research and Radiation Processing, 2023, 41(6): 060201
Search by keywords or author
Journals >Journal of Radiation Research and Radiation Processing >Volume 41 >Issue 6 >Page 060201 > Article
- Journal of Radiation Research and Radiation Processing
- Vol. 41, Issue 6, 060201 (2023)
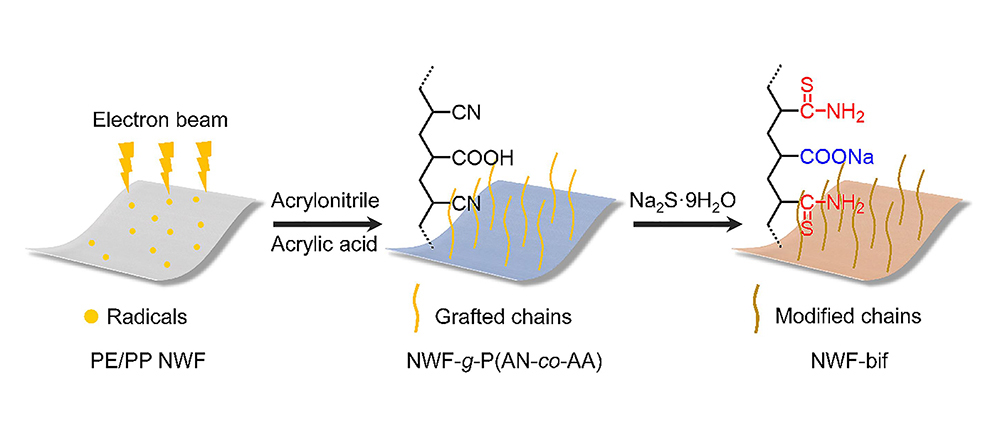
Fig. 1. Synthetic route for NWF-bif

Fig. 2. FTIR spectra of NWF samples before and after modification
Fig. 3. TG and DTG curves of NWF samples before and after modification (color online)
Fig. 4. Water contact angles of NWF samples before and after modification
Fig. 5. SEM images of (a) PE/PP NWF, (b) NWF-g-P(AN-co-AA), (c) NWF-bif, and (d) Au@NWF-bif
Fig. 6. Effect of pH on the Au(Ⅲ) adsorption (initial concentration, 25 mg/L; sorbent dosage, 0.1 g/L; time, 240 h )(color online)
Fig. 7. (a) Effect of adsorption time on Au(Ⅲ) adsorption capacity; fitting results of (b) pseudo-first-order and (c) pseudo-second-order models (initial concentration, 25 mg/L; sorbent dosage, 0.1 g/L; pH, 2)
Fig. 8. (a) Adsorption isotherm curve of NWF-bif to Au(III); Fitting results of (b) Langmuir and (c) Freundlich isotherm models (sorbent dosage, 0.1 g/L; time, 240 h; pH, 2)
Fig. 9. Selectivity of NWF-bif in mixed metal ion solution (initial concentration of each ion, 10 mg/L; sorbent dosage, 1 g/L; time, 120 h; pH, 2)
Fig. 10. Optical photos of Au@NWF-bif before and after high-temperature treatment
Fig. 11. Elements distribution of NWF-bif before and after Au(III) adsorption
Fig. 12. (a) XPS spectra of NWF-bif before and after adsorption; (b) Au 4f fine spectra of Au@NWF-bif; (c) XRD spectra of pristine PE/PP NWF, NWF-bif, and Au@NWF-bif; (d) O 1s, (e) N 1s, and (f) S 2p XPS fine spectra of NWF-bif before and after adsorption
| |||||||||||||||||||||
Table 1. Fitting parameters of kinetic models
| ||||||||||||||||||||||||
Table 2. Fitting parameters of isotherm models
|
Table 3. Distribution coefficient (K) of tested metal ions and selectivity coefficient (β) of Au(III)

Set citation alerts for the article
Please enter your email address



