Kun Wang, Yi-Zhi Qu, Chun-Hua Liu, Ling Liu, Yong Wu, H P Liebermann, Robert J. Buenker. Charge transfer in low-energy collisions of Be3+ and B4+ ions with He[J]. Chinese Physics B, 2020, 29(9):
Search by keywords or author
- Chinese Physics B
- Vol. 29, Issue 9, (2020)
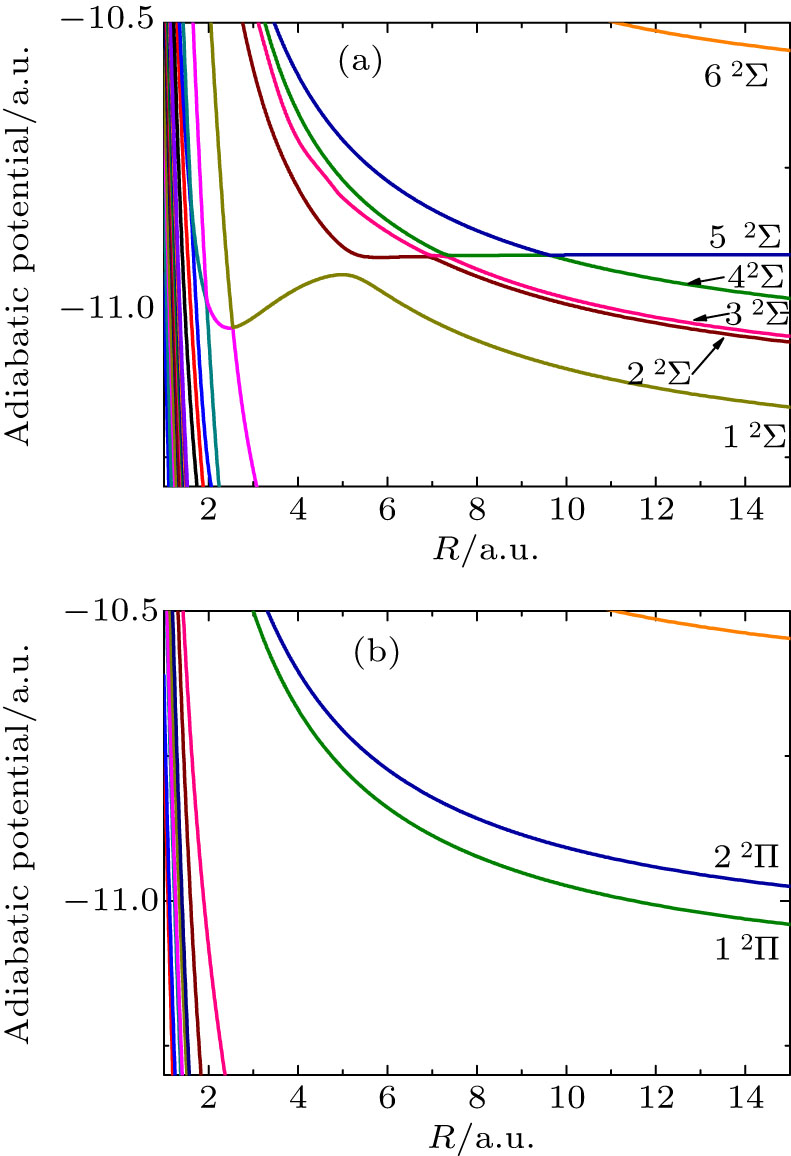
Fig. 1. Potential curves of BeHe3+ molecular ions refer to Table 1 .
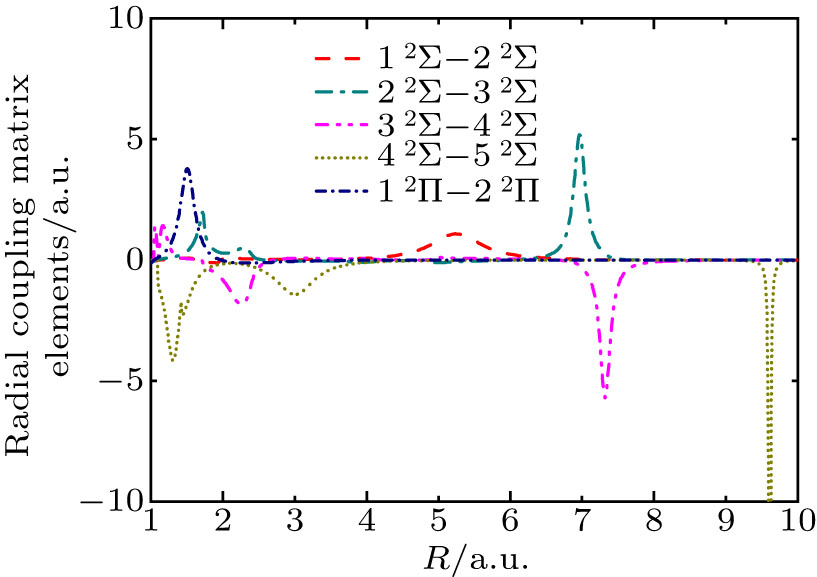
Fig. 2. Radial coupling matrix elements for BeHe3+.
Fig. 3. Rotational coupling matrix elements for BeHe3+.
Fig. 4. Potential curves of BHe4+ molecular ions refer to Table 2 .
Fig. 5. Coupling matrix elements for BHe4+: (a) and (b) radial coupling matrix element between 1Σ+ states, (c) radial coupling matrix element between 1σ states, (d) rotational coupling matrix element between 1Σ+ and 1π states, (e) and (f) some important detailed radial coupling matrix element between exit states with the initial state.
Fig. 6. Comparison between the present SEC cross sections for the Be3+(1s) + He(1s2) collision with other theoretical results. Present QMOCC calculation considering the ETF effects (solid line with filled squares); semi-classical molecular orbital close coupling (SMOCC) results of Suzuki et al. [5 ] (dashed line with filled circles).
Fig. 7. State-selective cross sections of present QMOCC results (considering the ETF effects) and the results of Suzuki et al. [5 ] for electron capture to the 1s2s and 1s2p states of Be2+ ions.
Fig. 8. Cross sections of present QMOCC results (considering the ETF effects) and the results of Suzuki et al. [5 ] for electron capture to singlet and triplet states of Be2+ ions.
Fig. 9. Comparison between the present SEC cross sections for the B4+(1s) + He(1s2) collision with other theoretical[7 ,8 ,10 ] and experimental[11 ] results. Present QMOCC calculation considering the ETF effects (solid line with open squares); AOCC results of Hansen et al. [7 ] (dashed line); semi-classical results of Fritsch et al. [8 ] (dash dotted line) and Shimakura et al. [10 ] (dotted line). Experimental results of Iwai et al. [11 ] (filled circles with error bars).
Fig. 10. Comparison between the present state-selective SEC cross sections for the B4+(1s) + He(1s2) collision with other theoretical[7 ,8 ,10 ] results. Present QMOCC calculation considering the ETF effects (solid line); AOCC results of Hansen et al. [7 ] (dotted line); semi-classical results of Fritsch et al. [8 ] (dashed line) and Shimakura et al. [10 ] (dash dotted line).
Fig. 11. Comparison between the present DEC cross sections for the B4+(1s) + He(1s2) collision with other theoretical results.[7 –9 ] Present QMOCC calculation considering the ETF effects (solid line with open circles); AOCC results of Hansen et al. [7 ] (dash dotted line); semi-classical results of Fritsch et al. [8 ] (dotted line); and full electron molecular expansion method of Bacchus-Montabonel[9 ] (dashed line).
Fig. 12. Comparison between the present state-selective DEC cross sections for the B4+(1s) + He(1s2) collision with other theoretical results.[7 –9 ] Present QMOCC calculation considering the ETF effects (solid line); AOCC results of Hansen et al. [7 ] (dash dotted line); semi-classical results of Fritsch et al. [8 ] (dotted line); and full electron molecular expansion method of Bacchus-Montabonel[9 ] (dashed line).
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 1. Asymptotic separated-atom energies for the states of BeHe3+. The bold 5 2Σ represents the initial state.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 2. Asymptotic separated-atom energies for the states of BHe4+. The bold 13 2Σ represents the initial state.
Set citation alerts for the article
Please enter your email address



