Yongyi Chen, Lirong Bao, Hui Wang, Zheng Ning, Xiandong Zhong, Jinle Cao, Ruiqi Shen, Wei Zhang. Research Progress in Preparation of Nanoparticles by Laser Ablation in Liquid[J]. Chinese Journal of Lasers, 2021, 48(6): 0600002
Search by keywords or author
- Chinese Journal of Lasers
- Vol. 48, Issue 6, 0600002 (2021)
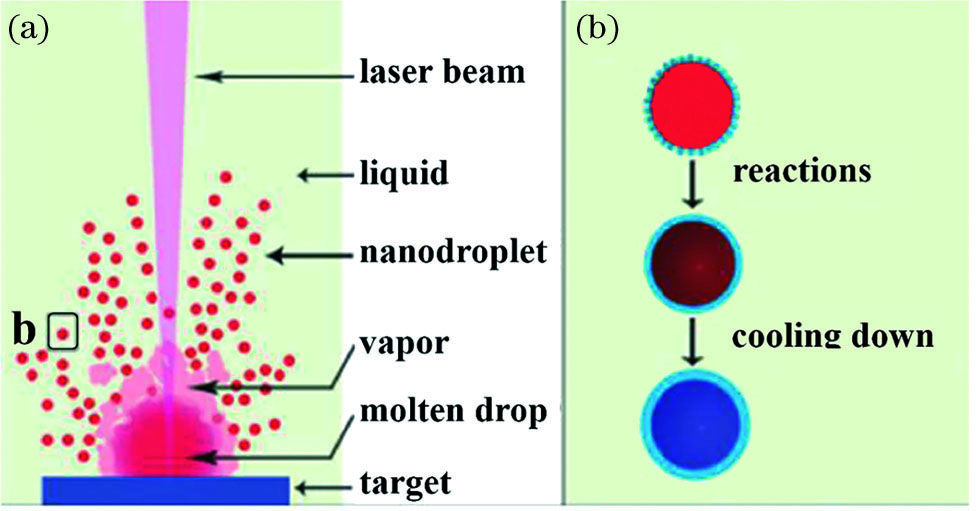
![Formation and cooling of nanodroplets[9]. (a) Formation of nanodroplets; (b) cooling of nanodroplets](/richHtml/zgjg/2021/48/6/0600002/img_1.jpg)
Fig. 1. Formation and cooling of nanodroplets[9]. (a) Formation of nanodroplets; (b) cooling of nanodroplets
![Schematic of the interaction between nanosecond pulsed laser and target[11]](/richHtml/zgjg/2021/48/6/0600002/img_2.jpg)
Fig. 2. Schematic of the interaction between nanosecond pulsed laser and target[11]
Fig. 3. Interaction between femtosecond pulsed laser and silicon wafer[11]. (a) Reaction on the surface of silicon wafer at different power densities; (b) material absorbs laser energy; (c) electrons stripped from atoms; (d) coulomb explosion on the surface of the material
Fig. 4. Formation mechanism of carbonized products with different structures in acetone[23]
Fig. 5. Formation of nanoparticles in different concentrations of SDS solutions[24]
Fig. 6. FE-SEM images of nanoparticles prepared in different concentrations of CTAB solutions[25]
Fig. 7. Molecular structure of PVP, PVA, and PEG and protection for Al nanoparticles[26]
Fig. 8. Optical microscopy and scanning electron microscopy images and corresponding particle size distribution histograms of NiO particles prepared in different solutions under the same condition[34]. (a) Distilled water; (b) ethanol solution
Fig. 9. Schematic of preparation of nanoparticles by laser ablation in dynamic microfluidics[37]. (a) Experimental setup; (b) confined mode of operation; (c) continuous mode of operation
Fig. 10. Scanning electron microscopy images of Cu nanoparticles prepared with different pulse widths[38]. (a)(b) 5 ns; (c)(d) 200 ps; (e)(f) 30 fs
Fig. 11. Formation of nanoparticle in different solutions[39]. (a) Deionized water; (b) sodium hydroxide solution; (c) hydrogen peroxide solution; (d) anhydrous ethanol
Fig. 12. Three methods for laser ablation in liquid[53]
Fig. 13. Histogram of particle size distribution of alloy nanoparticles prepared in different solutions and corresponding hydrodynamic diameter, Zeta potential, and Ferret diameter[56]. (a) Histogram of particle size distribution of alloy nanoparticles prepared in acetone; (b) histogram of particle size distribution of alloy nanoparticles prepared in MMA; (c) histogram of particle size distribution of alloy nanoparticles prepared in deionized water; (d) h
Fig. 14. TEM images of nanoparticles prepared by ablating alloy targets with different molar ratios of Pb to Zn[57]. (a)(c)(e) Lowly enlarged TEM; (b)(d)(f) highly enlarged TEM
Fig. 15. TEM images of the samples prepared in four solutions[66]
Fig. 16. Ablated silicon mass as function of the number of laser pulses in different wavelengths[74]. (a) 1064 nm; (b) 355 nm
|
Table 1. Preparation of metal nanoparticles by laser ablation in liquid
|
Table 2. Preparation of metal oxide nanoparticles by laser ablation in liquid
|
Table 3. Preparation of alloy nanoparticles by laser ablation in liquid
|
Table 4. Preparation of non-metallic nanoparticles by laser ablation in liquid
Set citation alerts for the article
Please enter your email address



