Yuanfei Hu, Junrong Zheng, Enming You, Songyuan Ding. Sub‑10 nm Ultrathin Metal Films: Preparation, Optical Properties, and Applications[J]. Chinese Journal of Lasers, 2023, 50(1): 0113010
Search by keywords or author
- Chinese Journal of Lasers
- Vol. 50, Issue 1, 0113010 (2023)
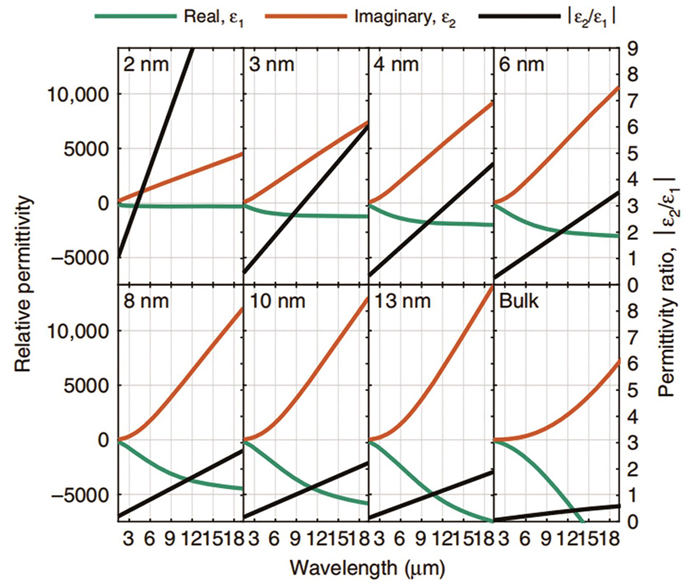
![Relative permittivities of ultrathin gold films with different thicknesses[14]](/richHtml/zgjg/2023/50/1/0113010/img_01.jpg)
Fig. 1. Relative permittivities of ultrathin gold films with different thicknesses[14]
![Wettability of metal on substrate surface is described by Young's equation[21]](/richHtml/zgjg/2023/50/1/0113010/img_02.jpg)
Fig. 2. Wettability of metal on substrate surface is described by Young's equation[21]
Fig. 3. Surface morphologies of 6-nm-thick silver films prepared with different seed layers[18]. (a) No seed layer; (b) seed layer is 1-nm-thick Cu; (c) seed layer is 1-nm-thick Si; (d) seed layer is 1-nm-thick Ti
Fig. 4. Characterization results of thickness and morphology of 3 nm ultrathin gold films[30]. (a) Height profile and (b) STEM image for 3-nm-thick gold film grown on 1-nm-thick copper seed layer; SEM images of (c) 3-nm-thick gold film without seed layer and (d) 3-nm-thick gold film with seed layer
Fig. 5. Principle of organic modification method and surface morphologies of prepared ultrathin gold film[12,41-42]. (a) Principle of preparing ultrathin gold films by MPTMS; (b)principle of preparing ultrathin gold film by APTMS ; (c) SEM image of surface morphology of ultrathin gold film without adhesion layer ; (d) SEM image of surface morphology of ultrathin gold film prepared by MPTMS adhesion layer; (e) SEM image of surface morphology of ultrathin gold film prepared by APTMS adhesion layer
Fig. 6. Diagram of co-deposition and SEM images and surface morphologies of silver films[19]. (a) Diagram of co-deposition; (b) SEM image of 9 nm pure Ag film with surface morphology shown in inset; (c) SEM image of 9 nm Al-doped Ag film with surface morphology shown in inset
Fig. 7. Results of low temperature deposition[20]. (a) Diagram of low temperature deposition; (b)-(e) SEM images of 5-nm-thick gold films deposited at different temperatures; (f) block resistance of ultra-thin gold films as a function of deposition temperature
Fig. 8. SEM and TEM images of 8-nm-thick gold nanoshells[64]. (a) SEM image; (b) TEM image
Fig. 9. Red-shifting of plasmon of UTMF. (a) Dipole resonance frequencies of 1-5 layer metal disks as a function of diameter [72]; (b) schematic and SEM images of ultrathin Ag film ribbon [73]; (c) dispersion curves of Au film ribbons with different thicknesses[30];(d) normalized extinction spectra of Ag film ribbons with width of 70 nm and different film thicknesses[73]; (e) extinction spectra of SiO2 nanoparticles containing 8-32 nm thick gold shells[62]
Fig. 10. Gate-tunable plasmons in UTMF. (a) Extinction spectra of single-atom layer gold disk and gold sphere with diameter of 20 nm as a function of doping charge density[8] ; (b) ratio of resonance energy offset to full width at half-maximum as a function of doping concentration for gold disk and gold sphere[8]; (c) measured offsets of plasmonic resonant wavelength in whole voltage cycle for UTMF with different thicknesses [30]; (d) simulated shift of plasmonic resonant wavelength by varying effective surface carrier density with conceptual view of dynamic tuning of UTMF nanoribbons by ion-gel gating shown in inset[30]
Fig. 11. Electrical and optical properties of ultrathin gold films[15]. (a) Schematics of prepared structure and incident direction of detection light; (b) resistivity and sheet resistance of Au films as a function of deposited thickness; (c) transmission and reflection spectra of Au films with different thicknesses; (d) experimental (left) and simulated (right) absorption spectra of ultrathin gold films with different thicknesses
Fig. 12. Nonlinear response of ultrathin metal films. (a) AFM images of prepared gold films with different thicknesses[81] ; (b) measured third-order polarizability of gold films[81]; (c) STEM image of metal quantum well cross section[82] ; (d) closed-loop z-scan curves for gold films with thicknesses of 3 nm and 15 nm[82]; (e) variation of calculated second harmonic, third harmonic and Kerr nonlinear polarizability with thickness and wave vector[9]
Fig. 13. Properties of transparent electrode. (a) Schematic of flexible polymer light-emitting diodes (PLEDs) using ultrathin Ni-doped Ag-based flexible transparent electrode[86] ; (b) external quantum efficiencies of Ni-doped Ag-based and ITO-based PLEDs[86]; (c) calculated and measured transmittance curves of transparent electrode with dielectric-metal-dielectric structure[87]; (d) photograph of fabricated flexible electrode[87]
Fig. 14. Development history of ultrathin metal film
|
Table 1. Summary of relevant work for each method
Set citation alerts for the article
Please enter your email address



