- Photonics Research
- Vol. 12, Issue 5, 1024 (2024)
Abstract
1. INTRODUCTION
Photodynamic therapy (PDT) is an emerging therapeutic modality that harnesses the interaction between photosensitizers, light, and molecular oxygen to selectively destroy cancer cells [1,2]. However, the current application of PDT is primarily focused on treating superficial or shallow tissue tumors. This restriction stems from the fact that the photosensitizers of traditional PDT predominantly respond to visible light, which has poor penetration into deep tissues due to the absorption of most biomolecules, thereby limiting its efficacy in treating deep-seated tumors [3]. To achieve greater light penetration depth, near-infrared (NIR) light has emerged as an ideal option due to its lower absorption by most biological tissues within the NIR window (700–1700 nm) [4–7]. NIR light possesses enhanced tissue-penetrating capabilities, allowing for more efficient penetration of deep-seated tumors and activation of photosensitizers, thus improving the effectiveness of PDT [6]. The combination of NIR light with suited photosensitizers enables deeper eradication of cancer cells and reduces damage to surrounding normal tissues [3,8,9].
To overcome the limitations of light penetration in PDT, upconversion nanoparticles (UCNPs) have been extensively investigated as promising candidates [10–16]. Researchers have synthesized UCNPs with various properties by controlling their composition, structure, and surface modifications to further optimize their performance [12,17–21]. For instance, doping with rare-earth ions such as erbium, gallium, and yttrium enables different upconversion spectra ranging from near-infrared to visible light [19,22,23]. Additionally, factors such as the size, shape, and coating materials of the nanocrystals can influence the performance of UCNPs, including upconversion efficiency, photostability, and biocompatibility [18,22,24–26]. Besides, UCNPs have demonstrated broad application prospects not only as light sources in PDT but also in areas such as biological imaging [11,27–31], drug release [11,32], and photothermal therapy [11,27,33]. In PDT treatment, UCNPs convert near-infrared (NIR) light into higher-energy visible or ultraviolet light, enabling deeper tissue penetration and activation of photosensitizers at their optimal wavelengths for therapeutic effects [2,7,30,34]. Additionally, UCNPs synthesized with precision can attain energy transfer efficiencies near 60%, ensuring effective energy conversion even in deep tissues [35]. This makes UCNPs an ideal platform for enhancing the efficacy of PDT. Furthermore, the integration of UCNPs with photosensitizers is crucial as it directly influences the efficiency of reactive oxygen species (ROS) generation. Among various photosensitizers, protoporphyrin (PPIX) has garnered significant attention due to its favorable photophysical properties, biocompatibility, and clinical applicability [36–41]. However, existing studies faced challenges such as limited and unstable photosensitizer loading of UCNPs, making it difficult to achieve sufficient ROS concentrations for effective tumor destruction, despite achieving enhanced light penetration depth [10]. Meanwhile, UCNPs cannot be actively enriched in the tumor site due to the lack of specific recognition ability. The primary challenge in cancer therapy is the disruption of the balance between cancerous and normal tissues during treatment. Due to physiological and microenvironmental differences between tumor and normal tissues, an increasing number of materials are being developed for targeted tumor therapy. Folic acid is widely used by researchers due to its affordability, accessibility, non-toxicity, non-immunogenicity, and high affinity to folate receptors [42]. Based on this, we are seeking a more efficient method in PDT, by which UCNPs with abundant photosensitizers can specifically gather into tumor tissues, to generate a lot of ROS and then improve tumor clearance rates in deep-seated tumors.
In this study, a novel composite material, PPIX-PEI-UCNP@FA NPs, was synthesized [Fig. 1(a)], comprising PPIX molecules, UCNPs doped with ytterbium (Yb) and erbium (Er), PEI, and FA. Current modifications of UCNPs with PEI typically involve a surface coating layer of PEI, which does not exploit the potential spatial structure provided by PEI to achieve higher drug loading and enhanced light absorption efficiency [43]. However, clinically, the high cytotoxicity of PEI is the main barrier to the safe use of PEI-coated nanomaterials. PEI has significant toxicity due to high polycationic charge density generated by a large number of amino groups. It can cause severe membrane damage inducing necrotic or apoptotic processes. In our work, the highly branched PEI rich in amino groups was used to link surface bifunctionalized UCNPs, forming a 3D structure with a high specific surface area, centered around PPIX-loaded UCNP nodes. This architecture achieved a porphyrin loading rate of up to 0.69% (mass fraction). PPIX was loaded onto the UCNPs and crosslinked with the 3D network formed by PEI as well, resulting in a higher loading capacity compared to previous strategies. Moreover, being crosslinked with PPIX blocked the amino groups in PEI, thus reducing its toxicity by diminishing the polycationic charge (because of less cationic ligands interacting with cells) through triple PPIX conjunction, while effectively reducing PEI toxicity by utilizing amine groups. The UCNPs extended the excitation wavelength to 980 nm, allowing therapeutic penetration beyond 10 mm. The branched PEI enhanced the loading efficiency of PPIX, leading to effective singlet oxygen generation and high tumor cell kill rates. The functionalization with folic acid (FA) enabled targeted drug delivery to cancer cells. The PPIX-PEI-UCNP@FA nanoparticles demonstrated properties of near-infrared excitation, deep tissue penetration, high ROS production, and tumor-specific targeting. Compared to PPIX loaded PLGA-PEG NPs [44], our material achieves a 3-fold increase in drug loading and lower toxicity, and nearly triples ROS production in deep tissue. Benefiting from the high loading capacity offered by the spatial structure of PEI, our approach has doubled the targeting efficiency compared to systems without PEI.
Sign up for Photonics Research TOC. Get the latest issue of Photonics Research delivered right to you!Sign up now
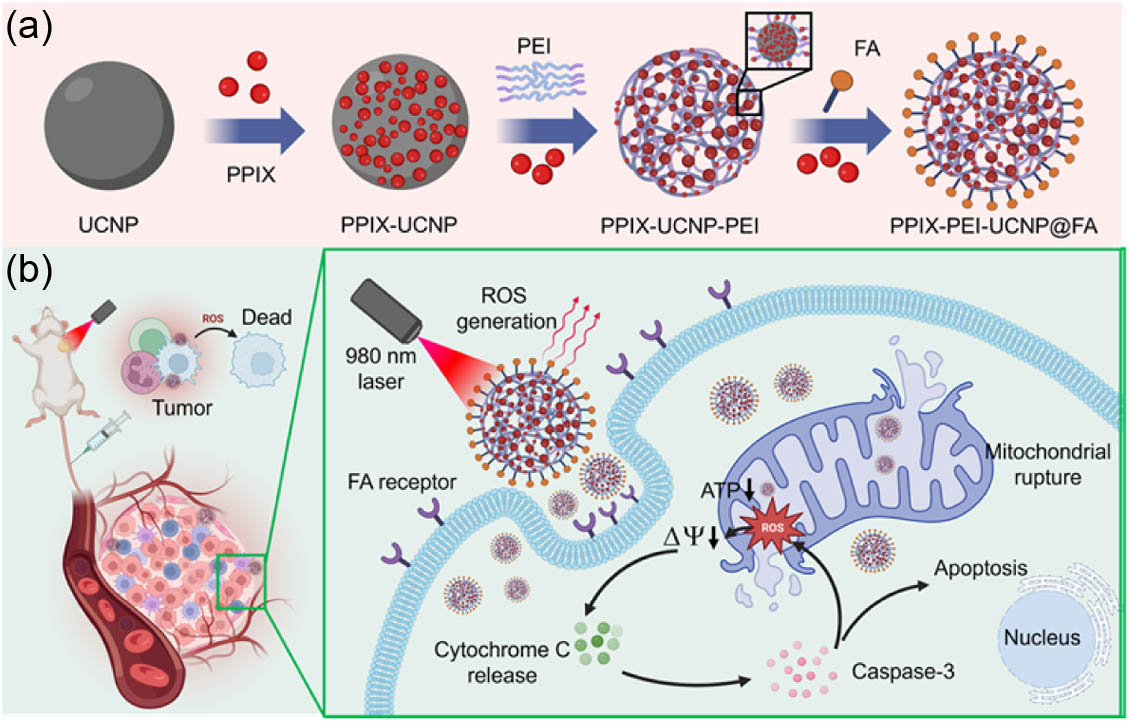
Figure 1.(a) Scheme of synthesis of PPIX-PEI-UCNP@FA NPs. (b) Mechanism of PPIX-PEI-UCNP@FA NPs as the PDT agent for tumor therapy via the 980 nm laser.
2. RESULTS AND DISCUSSION
![]()
Figure 2.Synthesis and characterization of PPIX-PEI-UCNP@FA NPs as an agent of PDT. (a) The transmission electron microscopy (TEM) image of UCNPs (scale bar: 100 μm). (b) The Fourier-transform infrared spectroscopy (FTIR) analysis of the UCNPs and of PPIX-PEI-UCNP@FA NPs. (c) UV-visible absorption spectrum of PPIX and photoluminescence spectrum of UCNPs and PPIX-UCNPs with the 980 nm laser. (d) High angle annular dark field (HAADF) image of PPIX-UCNPs from TEM (scale bar: 100 μm). (e) An overlap of scanning TEM (STEM) mapping image of
Subsequently, we employed a one-step self-assembly and chemical synthesis approach to encapsulate the photosensitizer PPIX onto the surface of the core-shell structured upconversion nanomaterial. To enhance the loading capacity of the final product for PPIX, we utilized polyethyleneimine (PEI), rich in abundant amino groups, as the framework of the end product. Through amidation with the carboxyl groups in PPIX, we obtained PPIX-PEI-UCNPs with an average particle size of approximately 387 nm. The nanomaterials were also analyzed using scanning TEM (STEM) coupled with energy dispersive spectroscopy (EDS) to confirm the composition of nanoparticles. The high-angle annular dark-field (HAADF) STEM image in Fig. 2(d) also shows a clear structure of PPIX-PEI-UCNPs. An overlay of and element EDS mapping image is shown in Fig. 2(e), illustrating the uniform distribution of element from the original PPIX throughout the entire nanoparticle, forming the outer layer that encases the inner core containing element from the UCNPs. This indicates the successful composite formation of PPIX-UCNPs, facilitated by PEI assistance.
To extend the activation wavelength of PPIX to the near-infrared range (980 nm), fluorescence resonance energy transfer (FRET) between UCNPs and PPIX is required. For FRET to occur, the donor’s emission peak must overlap to some extent with the acceptor’s absorption peak. As shown in Fig. 2(c) (blue line), PPIX exhibits several gradually decreasing absorption peaks between 500 nm to 650 nm, while the main absorption peak of the UCNPs is around 980 nm, with distinct upconversion emission peaks at approximately 540 nm and weaker emission peaks at 525 nm and 660 nm. It is evident that the emission peak of UCNPs at around 540 nm significantly overlaps with the absorption peak of PPIX, fulfilling the basic condition for fluorescence resonance energy transfer. Additionally, the core-shell structure of the UCNPs allows for a certain distance between the donor and acceptor, which further supports the occurrence of FRET. By measuring the fluorescence spectrum of PPIX-UCNPs, a significant decrease in the fluorescence intensity at 540 nm compared to UCNPs’ fluorescence spectrum confirms that PPIX absorbs high-energy photons emitted by UCNPs [Fig. 2(c), green line]. Besides, the emission peak of porphyrin appeared at 660 nm. Thus, we successfully utilized UCNPs to extend the excitation wavelength of the photosensitizer PPIX to the near-infrared range through efficient FRET.
In order to achieve specific targeting of tumor cells, increase the drug concentration inside the cells, and reduce the damage to normal cells, we introduced folate (FA) to PPIX-PEI-UCNPs. Studies have confirmed that folate receptors (FRs) are widely expressed on the surface of various tumor cells, while only a small amount is expressed in normal cells. Additionally, folate receptors have high affinity for folate or folate derivatives. Thus, we successfully obtained FA-modified nanomaterials, PPIX-PEI-UCNP@FA NPs, with FR as the target. Based on the infrared spectrum of the composite material [Fig. 2(b), red line], notable changes are observed in the UCNPs upon coupling with PPIX and FA. The disappearance of the N-H deformation vibration absorption peak of amino group at and the appearance of a new N-H bending vibration strong absorption peak at indicate the formation of amide bonds between the amino groups of the UCNPs and the FA, confirming their successful coupling. In the range of , characteristic absorption peaks are present in the spectrum of the new composite, which verifies the presence of UCNPs. The N-H stretching vibration peak at and the porphyrin skeleton vibration peak at around confirm the presence of PPIX. The Zetasizer Nano analyzer measurement revealed that the materials carry a negative charge (), which facilitates their accumulation in tumor sites through the bloodstream for an extended period. Furthermore, these nanomaterials are internalized by tumor cells with high expression of folate receptors through receptor-specific interactions. Under light activation, targeted therapy for tumors is achieved, thanks to the specific binding of FA to the folate receptors on tumor cells. Finally, to validate the photodynamic therapy efficacy of PPIX-PEI-UCNP@FA NPs, we used 9,10-anthracenediyl-bis(methylene)dimalonic acid (ABDA) as an active oxygen probe to test its ability to generate singlet oxygen under near-infrared laser irradiation in an aqueous environment. ABDA can undergo an oxidation-reduction reaction with active oxygen, leading to a reduction in the probe’s fluorescence intensity. Figure 2(f) displays the time-dependent UV–visible absorption spectra of the PPIX-PEI-UCNP@FA NPs and ABDA mixture solution upon 980 nm laser irradiation at . After 30 min of irradiation, the fluorescence intensity decreased to one-sixth of its original value, and ROS was significantly enhanced, demonstrating the material’s excellent capability to generate active oxygen. To assess the effectiveness of the composite material in deep tissue, we initially evaluated the tissue penetration depths of both 980 nm and 635 nm lasers. Two lasers were directed through varying thicknesses of pork tissue, and the transmitted light spots were measured. The results indicated a tissue penetration depth of approximately 10 mm for the 980 nm laser, whereas the 635 nm laser exhibited a penetration depth of less than 5 mm [Fig. 2(g)]. Figure 2(h) displays the spot profiles of the two beams after passing through 10 mm of tissue. For a specific confirmation of the therapeutic effect within deep tissue, separate experiments with the two lasers penetrating different tissue thicknesses were conducted. We measured the amount of generated ROS in the material under 30-min irradiation. As shown in Fig. 2(i), when passing through 10 mm of tissue, the 980 nm laser generates ROS at approximately 10 times the level of the 635 nm laser, which shows an excellent deep tissue killing effect.
![]()
Figure 3.Cell viability of (a) 4T1 cells and (b) L929 cells after incubation 48 h with different concentrations of PPIX-PEI-UCNP@FA NPs in the presence or absence of laser (980 nm,
It has previously been confirmed that PPIX-PEI-UCNP@FA NPs could generate excessive ROS when exposed to the laser, which caused cell damage. Therefore, -dichlorofluorescin diacetate (DCFH-DA) as an ROS probe was used to detect the production efficiency of ROS at the cellular level, in which 4T1 cells treated with were used as the positive control. According to the fluorescence staining images, 4T1 cells treated with showed substantial amounts of green fluorescence, which indicated that treatment activated the internal oxidative stress reaction of the cells, prompting them to produce an enormous quantity of ROS [Fig. 3(h)]. Interestingly, a similar increasing phenomenon of strong green intensity was observed after PPIX-PEI-UCNP@FA NPs in NIR-triggered treatment but was negligible in the other groups [Fig. 3(g)]. The quantitative analysis of different treated cells on flow cytometry also showed that PPIX-PEI-UCNP@FA NPs combined with laser irradiation revealed higher levels of intracellular ROS [Fig. 3(f)]. However, in L929 cells, except for the positive control group, there was no excessive production of ROS after different treatments. The apoptotic cells were then accessible via terminal-deoxynucleotidyl transferase-mediated dUTP nick-end-labeling (TUNEL) following different treatments. Representative images of TUNEL staining for the various treatments are shown in Fig. 3(i). The PPIX-PEI-UCNP@FA NPs group displayed significantly more green fluorescence in the presence of the laser than the negative control group, which was comparable to cells treated with and indicated a significant quantity of 4T1 cells going through apoptosis. These results suggested that the PPIX-PEI-UCNP@FA NPs could be stimulated by laser irradiation to produce excess ROS and hence induce cell apoptosis.
![]()
Figure 4.Cellular uptake of PPIX-PEI-UCNP@FA NPs in 4T1 cells determined by (a) confocal laser scanning microscopy (CLSM, scale bar: 25 μm) and (b) flow cytometry at different times. (c) Colocalization analysis of PPIX-PEI-UCNP@FA NPs and mitochondria using CLSM and the corresponding fluorescent line profile (red line representing PPIX-UCNP NPs and green line representing Mito-Tracker, scale bar: 25 μm). (d) Fluorescent images of mitochondrial ROS production after various treatments (scale bar: 25 μm). (e) Western blot (WB) results of caspase-3, cytochrome c, Bcl-2, and Bax after different treatments. (f) The relative protein expression levels of WB in (e). (g) Fluorescence images of mitochondrial membrane potential in 4T1 cells after different treatments using JC-1 (scale bar: 100 μm). (h) Flow cytometry of mitochondrial membrane potential (MMP) of 4T1 cells after different treatments using JC-1 and (i) the corresponding statistics of MMP loss. (j) BioTEM images of 4T1 cells after incubation with PPIX-PEI-UCNP@FA NPs with or without the laser (980 nm,
Mitochondria play an important role in regulating cell function, and redox imbalances in mitochondria could lead to mitochondrial dysfunction, which may eventually lead to cell death. In order to explore the mechanism of apoptosis induced by PPIX-PEI-UCNP@FA NPs in NIR-triggered PDT, the co-localization of NPs and mitochondria was initially analyzed. With the assistance of CLSM, it could be seen that the green fluorescence of mitochondria overlapped well with the red fluorescence of PPIX-PEI-UCNP@FA NPs after 12 h of incubation, demonstrating that the majority of NPs reached the mitochondria and were co-located with the mitochondria. By analyzing the correlation between mitochondria and PPIX-PEI-UCNP@FA NPs, it could be shown that there was a strong link between mitochondria and PPIX-PEI-UCNP@FA NPs, which suggested that the NPs had a special capacity for targeting mitochondria [Fig. 4(c)]. Then, corresponding fluorescent probes were used to detect ROS production in 4T1 cells after different treatments. The results showed that abundant ROS was induced following incubation of 4T1 cells with PPIX-PEI-UCNP@FA NPs and laser irradiation [Fig. 4(d)]. These excessive ROS may lead to impairment of mitochondrial function and cell death. Compared with the negative control group, the PPIX-PEI-UCNP@FA NPs and laser combination group resulted in a 58.52% decrease in ATP production, which was lower than that of the -treated group (44.1%), presumably due to the mitochondrial dysfunction induced by the NPs under the laser. Additionally, ROS could cause changes in MMP, which was associated with cell apoptosis. Thus, the change in MMP was determined by a JC-1 probe, which existed in its aggregate state and generated red fluorescence at high MMP, while it appeared in monomer form and emitted green fluorescence under low MMP. It was clearly demonstrated that the majority of JC-1-labeled mitochondria displayed red fluorescence in the absence of lasers or PPIX-PEI-UCNP@FA NPs. In contrast, when the NPs and the laser were incubated with cells, the fluorescence color of the mitochondria dramatically changed from red to green [Fig. 4(g)]. Flow cytometry also revealed a statistically significant reduction (36.2%) in the number of 4T1 cells in the left upper corner corresponding to the JC-1 aggregate form in mitochondria after receiving PPIX-PEI-UCNP@FA NPs and laser treatment, indicating that NPs could damage mitochondrial function under laser excitation [Figs. 4(h) and 4(i)]. Changes in mitochondrial morphology may potentially be indicative of changes in mitochondrial function. In order to better comprehend the alterations in mitochondria, the ultrastructure of different post-treated cells was observed using biological transmission electron microscopy (BioTEM). As shown in Fig. 4(j), the morphology of 4T1 cells was essentially unchanged in the absence of laser or PPIX-PEI-UCNP@FA NPs treatment, while the laser-induced NP group caused severe mitochondrial structural damage, revealing extensive mitochondrial swelling with destruction of mitochondrial cristae.
Western blot (WB) assay was further performed to elucidate the mechanism of cell apoptosis induced by PPIX-PEI-UCNP@FA NPs in NIR-triggered PDT [Fig. 4(e)]. Bcl-2 and Bax proteins in the Bcl-2 family played an important regulatory role in cell apoptosis and could mediate the release of cytochrome c (Cyto c) through mitochondria-dependent pathway. The results showed that the increasing intracellular ROS levels induced by laser irradiation significantly stimulated the up-regulation of apoptosis-associated proteins Bax/Bcl-2 and Cyto c, which were 2.1 times and 3.0 times higher than those in the negative control group, respectively, indicating PPIX-PEI-UCNP@FA NPs in NIR-triggered PDT activated subsequent apoptosis [Fig. 4(f)]. For caspase-3, as one of the most critical apoptotic “executors” downstream of mitochondria-related pathways, its activation was largely dependent on the release of Cyto c. After both NPs and laser irradiation treatment, the relative protein expression of caspase-3 increased. Overall, all these findings demonstrated that PPIX-PEI-UCNP@FA NPs could significantly induce cell apoptosis via caspase-3-mediated mitochondria-dependent pathway in NIR-triggered PDT.
![]()
Figure 5.(a) Treatment protocol for normal BALB/c mice intravenously injected with PBS and PPIX-PEI-UCNP@FA NPs. (b) Representing H&E staining images of major organs (heart, liver, spleen, lung, and kidney) treated with PPIX-PEI-UCNP@FA NPs in the presence of the laser (980 nm,
![]()
Figure 6.(a) Schematic diagram of the establishment of a 4T1 tumor model and treatment process. (b) Distribution of PPIX-PEI-UCNP@FA NPs
Then, the 20 subcutaneously injected mice were randomly separated into four groups. In accordance with the in vitro experimental groups, G1: PBS (without laser), G1: PBS (with laser), G1: PPIX-PEI-UCNP@FA NPs (without laser), and G1: PPIX-PEI-UCNP@FA NPs (with laser) were selected for the
Histological analysis of tumor tissues and major organs was performed after undergoing various therapies. In all treatment groups, no significant pathological morphological changes were discovered in the major organs (liver, heart, kidney, spleen, and lung), indicating that the treatment group had good biosafety. Representative H&E tumor staining images showed that the cell structure was regularly arranged in the normal saline group (with or without laser) and the PPIX-PEI-UCNP@FA NPs group (without laser), and the nucleus shape was complete and clear. In contrast, in the PPIX-PEI-UCNP@FA NPs plus light treatment group, the tumor tissue was severely damaged, and the nucleus was widely fractured or even disappeared [Fig. 6(h)]. At the same time, TUNEL staining images also demonstrated that the combination treatment group led to the highest levels of apoptosis, as evidenced by the wide dispersion of green regions in Fig. 6(i). In the other three groups, only scattered green fluorescence was present. These results indicated the effective anticancer activity of PPIX-PEI-UCNP@FA NPs
3. CONCLUSION
Traditional PDT faces challenges in treating large or deep-seated tumors due to the limited penetration of visible light through thick tissues. To overcome this limitation, near-infrared light has been introduced into the field of PDT, as its absorption within the biological body is relatively low, thus enabling greater depth of penetration. However, the advancement of two-photon PDT is constrained by the requirement for the simultaneous absorption of two coherent near-infrared photons, leading to lower efficiency and the need for costly pulse lasers. To address this issue, upconversion fluorescence technology has been introduced. It has the capability to convert low-energy near-infrared light into high-energy visible light, matching the activation absorption wavelength of photosensitizers deep in the tissue, while maintaining efficient ROS generation. Compared to conventional two-photon excitation, upconversion fluorescence demonstrates higher efficiency and does not necessitate complex and expensive equipment, making it achievable through cost-effective continuous-wave laser diodes. This approach enables PDT to achieve greater penetration depths while reducing costs, thus becoming a viable option for practical treatment. In this study, we used UCNPs as carriers for PDT drugs, incorporating branched PEI to create a spatial structure centered on UCNP nodes, achieving a PPIX drug loading capacity of 0.69% (mass fraction). This facilitates the efficient generation of ROS in skin tumors (10 mm depth). Both in vitro and
APPENDIX A: MATERIALS AND METHODS
A total of 1 mmol of rare earth chlorides (0.80 mmol yttrium chloride, 0.18 mmol erbium chloride, 0.02 mmol erbium chloride) was placed in a three-neck flask with a capacity of 100 mL. Subsequently, 6 mL of oleic acid and 15 mL of octadecene were added. The mixture was heated under an argon atmosphere to 160°C and allowed to react for 60 min. Then, it was cooled to room temperature. A solution containing 4 mmol of ammonium fluoride and 2.5 mmol of sodium hydroxide in 10 mL of methanol was prepared and added dropwise to the reaction system. The mixture was stirred at room temperature for 60 min before raising the temperature to 60°C and maintaining the reaction for another 60 min. Following that, the temperature of the system was rapidly increased to 300°C and kept at that temperature for 60 min under an argon atmosphere. Finally, the temperature of the system was allowed to cool naturally to room temperature. Excess anhydrous ethanol was added to the reaction system to precipitate the product, which was then centrifuged three times at a speed of 8000 r/min. The resulting product was dispersed in n-hexane for subsequent use.
A total of 0.5 mmol of yttrium chloride solid was combined with 6 mL of oleic acid and 15 mL of octadecene. The mixture was heated under an argon atmosphere to 160°C and allowed to react for 60 min. It was then cooled to room temperature. The previously prepared NaYF4:18%Yb,2%Er nanomaterial was dispersed in 8 mL of -hexane and added to the reaction system. The mixture was stirred at this temperature for 30 min. Subsequently, a solution containing 2 mmol of ammonium fluoride and 1.25 mmol of sodium hydroxide in 10 mL of methanol was added dropwise to the reaction system. The mixture was stirred at room temperature for 60 min before raising the temperature to 60°C and maintaining the reaction for 30 min. After that, the temperature of the system was rapidly increased to 300°C and kept at that temperature for 60 min under an argon atmosphere. Finally, the temperature of the system was allowed to cool naturally to room temperature. Excess anhydrous ethanol was added to the reaction system to precipitate the product, which was then centrifuged three times at a speed of 8000 r/min. The resulting product was dispersed in n-hexane for subsequent use.
Synthesized NaYF4:18%Yb,2%Er@NaYF4 nanoparticles were collected by centrifugation and dispersed in 50 mL of anhydrous methanol with 500 mg of the particles. Surface functionalization was conducted by adding 10 mmol APTES and 55 mmol MPTMS, followed by stirring under an argon atmosphere at room temperature for 24 h to ensure complete silanization. Unreacted silane agents were removed by centrifugation (10,000 r/min, 10 min). The UCNPs were redispersed in 50 mL of anhydrous methanol, followed by the addition of 10 mL of 30% hydrogen peroxide and stirring for another 12 h at room temperature to oxidize thiol groups to carboxyl groups. Afterward, centrifugation and washing with ultrapure water were performed at least three times to remove any remaining hydrogen peroxide.
To prepare the PPIX solution, 0.1 g of PPIX was dissolved in 100 mL of DCM to create a 1 mg/mL solution. A PBS solution containing double the molar amount of EDC and NHS was prepared. EDC/NHS solution was added slowly to the PPIX solution and stirred at room temperature for 1 h. This solution was then added to the above-modified UCNPs and mixed by stirring at room temperature for 24 h. Unreacted PPIX and solvent were removed by centrifugation (8500 r/min, 15 min). The UCNPs were then washed with ultrapure water, repeated more than three times.
0.1 g of PEI was dissolved in 100 mL of ultrapure water to prepare a 1 mg/mL PEI solution, to which 1.5 mg of EDC and 0.1 mg of NHS were added. The mixture was stirred at room temperature for 30 min before being added to the UCNP suspension modified with PPIX. Electron beam irradiation of the solution was performed (3 MeV voltage, total dose of 100 kGy, dose rate of 2 kGy/min) for 50 min. The reaction was further stirred for 4 h at room temperature to form the PEI-linked UCNP complex. An additional 0.05 mL of activated PPIX was introduced to the reaction, adjusting the pH to neutral to maximize the reactivity of amine groups and prevent carboxyl protonation. The mixture was stirred overnight at room temperature. Centrifugation (10,000 r/min, 10 min) was used to remove unreacted PEI and PPIX. The complex was washed with ultrapure water, repeated more than three times. The washed complex was then transferred to a dialysis bag and dialyzed against PBS or ultrapure water for 48 h to remove small molecular impurities. The dialyzed complex was dried in a vacuum oven at 40°C for 12 h.
20 mg of folic acid was ultrasonically dispersed in 10 mL of MES buffer. To this mixture, 80 mg of EDC and 120 mg of NHS were added, followed by ultrasonication for 15 s. The mixture was then sealed with a film and incubated on a shaker at 37°C for 15 min. The composite particles prepared in step (4) were introduced into the reaction system, ultrasonicated rapidly for 1 min, and then transferred to a shaker at 37°C for 12 h. An additional 0.05 mL of activated PPIX was subsequently added to the mixture, which then reacted on the shaker for another 4 h. Upon completion of the reaction, the product was washed with deionized water and centrifuged (8000 r/min), repeated three times to obtain the final product (PPIX-PEI-UCNP@FA).
A suitable quantity of PPIX-PEI-UCNP@FA NPs were subjected to ultrasonic cleaning for a duration of at least 5 min in an ultrasonic cleaner. Subsequently, 5 μL of the solution was deposited onto a thin carbon film supported on a copper grid. After drying, the morphology of the samples was observed using the Thermo Fisher TALOS F200X 200 kV transmission electron microscope, followed by energy dispersive X-ray spectroscopy (EDS) elemental analysis and mapping surface scan measurements.
A predetermined quantity of dried PPIX-PEI-UCNP@FA NPs were ground and pressed into pellets. The structural characterization of the magnetic nanoparticles was conducted using the Thermo Fisher Nicolet 6700 Fourier-transform infrared spectrometer over a scanning range of to .
The ultraviolet-visible absorption spectra of the samples were recorded using the Thermo Fisher EV300 UV-Vis spectrophotometer.
Sample testing was carried out using the FLS1000 steady-state and transient fluorescence spectrometer from Edinburgh Instruments, United Kingdom.
The particle size, distribution, and surface potential of the samples were determined using the NanoBrook Omni Nanosizer for zeta potential measurements.
Then, a live/dead test was performed. The L929 and 4T1 cells were seeded onto 24-well plates ( cells per well) and cultured with PPIX-PEI-UCNP@FA NPs (60 μg/mL) for 48 h. Every day for 10 min, the L929 and 4T1 cells were exposed to radiation. Afterwards, the cells were stained with calcein-AM and PI (2 and 4 μmol/L) for 30 min. A fluorescent microscope (Olympus IX73) was employed to view fluorescence images of live/dead staining.
For flow cytometry detection, the 4T1 cells were seeded onto 6-well plates ( cells per well) followed by incubating with PPIX-PEI-UCNP@FA NPs (60 μg/mL) for various times. Then, cells were rinsed with fresh PBS to eliminate uninternalized NPs and trypsinized to collect cells. Cell suspensions were prepared by washing the cells with PBS several times and adding new PBS for flow cytometry.
The 4T1 cells ( cells) were seeded onto 6-well plates and cultured with PPIX-PEI-UCNP@FA NPs overnight. After being fixed in PBS containing 2.5% glutaradehyde according to the standard protocol, the mitochondrial morphology of cells was observed using the biological transmission electron microscopy (TEM, Tecnai G2).
For TUNEL staining analysis, the slides were stained with TUNEL in accordance with the approved method. The stained slides were imaged using a fluorescence microscope (80i, Nikon).
Acknowledgment
Acknowledgment. The authors acknowledge support from the state Key Laboratory on Fiber Optic Local Area Communication Networks and State Key Laboratory of Metal Matrix Composites, and they also thank the Ruijin Hospital.
References
[3] C. Sisi, Y. Deyan, C. Yuqi.
[5] G. Liu, J. Sheng, Y. Zhao.
[16] L. Yanyan, M. Xianfu, W. Bu. Upconversion-based photodynamic cancer therapy. Coord. Chem. Rev., 379, 82-98(2017).
[31] C. Song, S. Zhang, Q. Zhou. Upconversion nanoparticles for bioimaging. Nanotechnol. Rev., 6, 233-242(2017).
[34] G. Arif, J. Xu, P. Yang. Upconversion processes: versatile biological applications and biosafety. Nanoscale, 9, 12248-12282(2017).
[38] E. S. Nyman, P. H. Hynninen. Research advances in the use of tetrapyrrolic photosensitizers for photodynamic therapy. J. Photochem. Photobiol. B, 73, 1-28(2004).
[39] A. F. McDonagh, L. A. Palma. Tin-protoporphyrin: a potent photosensitizer of bilirubin destruction. Photochem. Photobiol., 42, 261-264(2010).
[40] J. Kou, D. Dou, L. Yang. Porphyrin photosensitizers in photodynamic therapy and its applications. Oncotarget, 8, 81591-81603(2017).

Set citation alerts for the article
Please enter your email address



