- Journal of Inorganic Materials
- Vol. 35, Issue 3, 293 (2020)
Abstract
Cadmium (Cd(II)) is one of the toxic heavy metals from industrial activity such as nickel-cadmium batteries, mining, paint pigmentations, alloy, and electroplating[
Molybdenum disulfide (MoS2), a “rising star” material, has attracted tremendous interest in remediation of heavy metals from water over past decades[
Many mechanisms have been proposed for the interaction of metals with MoS2, including ion exchange, inner layer metal-S complexation, outer layer electrostatic attraction, surface precipitation[
In present work, both batch experiment and EXAFS technique were employed to analyze the sorption behavior and mechanism of Cd(II) on MoS2. Firstly, MoS2 were characterized by SEM, TEM, XRD, FT-IR, etc. Secondly, effects of solution pH, ionic strength, and temperature, contact time on the sorption of Cd(II) and the adsorption kinetics, isotherms, thermodynamics in solutions with various pH were evaluated. Finally, EXAFS technique was used to analyze the sorption mechanisms of Cd(II) on MoS2 as a function of pH.
1 Materials and methods
1.1 Materials
Overall chemical reagents including cadmium nitrate (Cd(NO3)2·2H2O), sodium nitrate (NaNO3), sodium hydroxide (NaOH), nitrite acid (HNO3), and molybdenum disulfide (MoS2) were analytical grade purchased from Nanjing XFNANO Materials Tech. Co. Ltd. (China) without further purification. Cd(II) stock solution was obtained by dissolving Cd(NO3)2·2H2O in distilled water.
1.2 Characterization
The intrinsic properties of MoS2 greatly determinate its sorption capacity, thereby it is helpful to investigate its characterization for revealing the sorption mechanism. Herein, TEM, SEM, EDX analysis and elemental distribution mappings of the MoS2 sample were carried out by using a transmission electron microscope (JEM-1011, Japan) instrument and a field emission scanning electron microscope (JSM-6360LV, Japan). XRD pattern of the sample was tested by the D8 Discover X-ray diffractometer (Bruker, Germany) with Cu Kα radiation (λ=0.1541 nm) and distinguished according to the JCPD standards. FT-IR spectrometer (NEXUS, America) was employed to evaluate surface functional groups of MoS2 in the wavelength range of 400-4000 cm-1. Zeta potential analyzer (Zetasizer Nano ZS, Malvern Co., UK) was used to locate the pHpzc of the adsorbent, i.e., the suspension of MoS2 and NaNO3 adjusted to an appropriate pH was test.
1.3 Batch experiment
A set of sorption experiments of Cd(II) onto MoS2 were conducted under N2 in polyethylene tubes. The stock suspensions of Cd(II), MoS2, NaNO3, and distilled water were mixed in polyethylene tubes in order to gain the desired concentrations. The pH was adjusted by adding 0.1 or 0.01 mol/L HNO3 or NaOH solution with negligible volumes. The tubes containing above mentioned mixtures were shaken for more than 12 h to achieve sorption equilibrium, and the the solid was separated by centrifugation method. Finally, the Cd concentration in the supernatant was tested. The Cd(II) sorption percentage, distribution coefficient (Kd), and sorption amount (qe) on MoS2 were calculated based on the following equations:
where C0 and Ce represent the initial and equilibrium concentrations of Cd(II) (mg/L), respectively. V is the suspension volume (L) and m is the MoS2 mass (g).
1.4 EXAFS data collection and analysis
EXAFS data were collected at room temperature on BL14W at Shanghai Synchrotron Radiation Facility (SSRF, China). The obtained EXAFS data was analyzed by using Athnea software. The raw, averaged data were processed to isolate the EXAFS oscillations by removal of the pre-edge background. The k3-weighted EXAFS spectra of Cd(II) were Fourier transformed (FT). The code FEEF7 and the as-known crystal structure of Cd(NO3)2, Cd(OH)2, CdS were used to calculate the theoretical scattering phases and amplitudes. The bond distance (R) and coordination number (CN), and the Debye-Waller factor of sample were optimized for each single peak by performing curve fitting with nonlinear least-squares.
2 Results and discussion
2.1 Characterization of MoS2 nanosheets
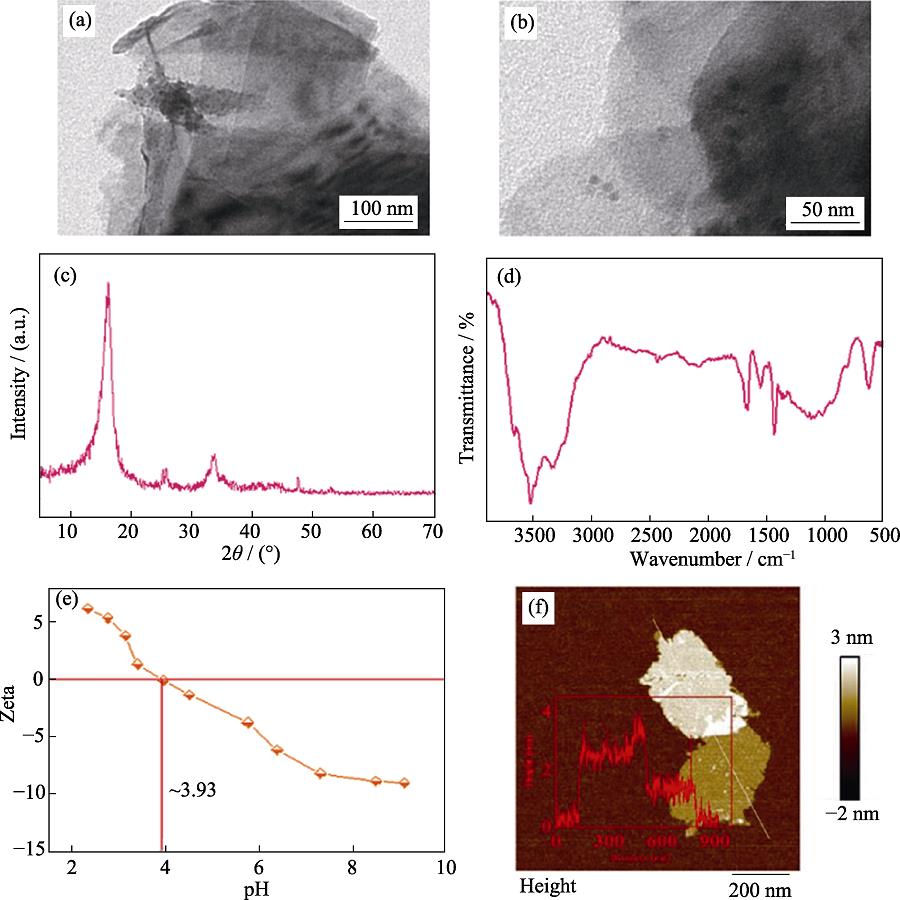
The morphologies and microstructures of MoS2 were characterized by SEM, TEM, etc., and the results are shown in Fig. S1 in detail. SEM images, EDX spectra, and elemental distribution mapping of MoS2 are shown in Fig. 1. It is evident that two non-target elements are present in MoS2 nanosheets, i.e., Cu and Zn. However, the elemental distribution percentages of Mo and S atoms are much higher than those of Cu and Zn, indicating that MoS2 contains negligible impurities. In other words, the effects of Cu and Zn in MoS2 materials on sorption can be ignored.
![]()
Figure .SEM image (a), TEM image (b), XRD pattern (c), FT-IR spectrum (d), Zeta potentials (e), and height cross-section profile (inset) and corresponding AFM image (f) of MoS2 samples
![]()
Figure .SEM image (a), EDX spectra, and corresponding elemental maps (b) for MoS2 nanosheets
2.2 Batch sorption
Fig. 2 shows the pH dependence of Cd(II) sorption on MoS2 in 0.1, 0.01 and 0.001 mol/L NaNO3 solutions, respectively. It is observed that the sorption is strongly dependent on pH. The percentage of Cd(II) sorption sharply increased at pH 3.3-6.5 and then creeped until a plateau level at pH>6.5. At pH 3.3-6.5, owing to protonation reaction, the number of protonated sites decreased with the increase of pH, leading to stronger affinity between the ions and MoS2. However, the saturated surface of MoS2 immobilized more cadmium ions at pH>6.5. It is noted that the species of Cd(II) are highly dependent on the solution pH. As provided by Wang et al.[
![]()
Figure .Effects of pH and ionic strength on Cd(II) adsoprtion onto MoS2 nanosheets
To understand the sorption kinetics and to determine their phenomenological coefficients, the sorption capacity as a function of contact time was shown in Fig. 3(a). The sorption rates at different pH were similar and the contact time needed for complete adsorption of Cd(II) on MoS2 was 2 h. The final sorption capacities at pH 4.55, 5.34, and 6.12 reached 28.4, 37.2 and 49.3 mg/g, respectively. These results illustrate that pH only enhances the sorption capacity but does not promote the sorption rate. Besides, 3 kinetic models were employed to analyze the kinetic data, including pseudo-first- kinetic model[
where qt (mg/g) represents the sorption capacity of Cd(II) at time t (h); k1 (h-1), k2 (g/(mg·h)), ki (g/(mg·h1/2)) are the rate constants of 3 models, respectively; C (mg/L) indicates the thickness of boundary layer. The linear plots of 3 models went with Fig. 3(b-d), respectively. The corresponding parameters can be found in Table 1. We can see that the sorption kinetics could be fitted better by the pseudo-second-order kinetic model than pseudo-first-order kinetic model due to the higher correlation coefficients (R2). The result implied the chemical sorption process[
![]()
Figure .Cd(II) adsorption on MoS2 nanosheets as a function of contact time (a) and the fitting of pseudo-first-order kinetic model (b), pseudo-second-order kinetic model (c) and intra-particle diffusion model (d) at different pH
![]()
Figure .Adsorption isotherms (a) and fitting results of Langmuir (b) and Freundlich (c) sorption isotherms of Cd(II) sorption on MoS2 at different temperatures and different pH
![]()
Figure .Linear plots of ln
| Sample conditions | shells | CN | ||
|---|---|---|---|---|
| Cd(NO3)2(aq) | Cd-O | 0.233(4) | 6.2(3) | 0.0010(1) |
| Cd(OH)2 | Cd-O | 0.238(2) | 6.1(4) | 0.0014(5) |
| Cd-Cd | 0.359(3) | 5.9(4) | 0.0032(5) | |
| CdS | Cd-S | 0.259(1) | 4.1(3) | 0.0024(2) |
| pH 3.56, sorption | Cd-S | 0.255(2) | 3.9(5) | 0.0027(3) |
| pH 6.48, sorption | Cd-S | 0.257(1) | 3.8(4) | 0.0023(5) |
| pH 9.57, sorption | Cd-O | 0..34(5) | 5.9(6) | 0.0016(3) |
| Cd-Cd | 0.357(3) | 5.6(4) | 0.0037(2) |
Table 3.
Structural parameters of Cd(II) reference and sorption samples
| pH | ||||
|---|---|---|---|---|
| 4.55 | 33.023 | 0.059 | 0.9896 | |
| Pseudo-first- | 5.34 | 23.903 | 0.053 | 0.9069 |
| 6.12 | 41.777 | 0.074 | 0.9835 | |
| pH | ||||
| 4.55 | 35.638 | 0.038 | 0.9869 | |
| Pseudo-second- | 5.34 | 42.230 | 0.078 | 0.9978 |
| 6.12 | 52.659 | 0.077 | 0.9986 | |
| pH | ||||
| 4.55 | 19.985 | 2.628 | 0.942 | |
| Intra-particle diffusion model | 5.34 | 32.500 | 1.877 | 0.987 |
| 6.12 | 39.759 | 3.004 | 0.980 |
Table 1.
Parameters of kinetic models for the adsorption of Cd(II) on MoS2 as a function of pH
2.3 EXAFS analysis
To reveal the interaction mechanisms between Cd(II) and MoS2 at microscopic level, local structures of Cd(II) adsorbed on MoS2 were investigated at various pH by EXAFS. The k3-weighted, normalized, background-subtracted EXAFS spectra $\sqrt{\chi -function}$and the corresponding Fourier transformed radial structure functions (RSFs) magnitudes and imaginary parts of Cd(II) reference samples at 3 pH are shown in Fig. S4. The EXAFS oscillation of the Cd(NO3)2 (aq) was a single sinusoidal waveform arising from the backscattering of oxygen atoms in the first shell. The RSFs present similar characteristic and possess only one peak at ~0.2 nm. The existence of Cd-O bond and absence of direct binding for Cd(II) to MoS2 surface imply a typical outer-sphere complexation of Cd(II)[
![]()
Figure .Normalized, background-subtracted and k3-weighted EXAFS spectra (a) and corresponding RSF magnitudes and imaginary parts (b) of Cd reference samples
![]()
Figure .EXAFS spectra (a) and the corresponding RSF magnitudes and imaginary parts (b) of Cd reference samples
![]()
Figure .Schematic representation of possible sorption sites on the surface of MoS2
3 Conclusions
In this work, the interaction mechanisms and microscopic structure of Cd(II) and MoS2 nanosheets were investigated by batch experiments and EXAFS technology. In batch experiments, the pH-dependent and ionic strength- independent sorption of Cd(II) onto MoS2 implied an inner-sphere complexation in the range of pH 3.3-9.6. The better fitted pseudo-second-order kinetics confirmed the chemical nature of the sorption and the intra-particle diffusion model reflected the sorption process from the surface to intra-particle diffusion to final equilibrium. The isotherms could be simulated better by Freundlich isotherms than by Langmuir isotherms model, indicating the heterogeneity of active sites on MoS2. The thermodynamics implied the sorption at each pH was a spontaneous, endothermic, and irreversible process. The EXAFS spectra revealed the coexistence of two sorption types. The inner-sphere complexation was formed in the form of Cd-S bond similar to CdS complex at pH 3.56 and 6.48, while the precipitation occurred in the form of Cd-O and Cd-Cd bonds similar to Cd(OH)2 at pH 9.57. In summary, the interaction mechanisms and local structure of Cd(II) and MoS2 are strongly affected by the solution pH.
Supporting materials
Supporting materials related to this article can be found at https://doi.org/10.15541/jim20190381.
Microscopic Insights into pH-dependent Adsorption of Cd(II) on Molybdenum Disulfide Nanosheets
DONG Lijia1, GUO Xiaojie2, LI Xue1, CHEN Chaogui1, JIN Yang1, AHMED Alsaedi3, TASAWAR Hayat3,4, ZHAO Qingzhou5, SHENG Guodong6
(1. School of Life Science, Shaoxing University, Shaoxing 312000, China; 2. College of Materials & Environmental Engineering, Hangzhou Dianzi University, Hangzhou 310018, China; 3. NAAM Research Group, Department of Mathematics, Faculty of Science, King Abdulaziz University, Jeddah 21589, Saudi Arabia; 4. Department of Mathematics, Quaid-I-Azam University, Islamabad 44000, Pakistan; 5. College of Resources and Environment, University of Chinese Academy of Sciences, Beijing 100049, China; 6. College of Chemistry and Chemical Engineering, Shaoxing University, Shaoxing 312000, China)
Characterization of MoS2 nanosheets
The morphologies and microstructures of MoS2 nanosheets were characterized by SEM, TEM, XRD, FT-IR, Zeta potentials, AFM, and EDX. Both SEM and TEM images (Fig. S1(a,b)) show the obvious 2D layered structure of MoS2 and its typical diameter ranges from hundreds of nanometers to several micrometers, implying ultrathin nature[
Sorption isotherms and thermodynamics
The sorption isotherms studies were carried out at different pH (i.e., 4.55, 5.34, 6.12) and 3 temperatures (i.e., 293, 313, 333 K) (Fig. S2). In Fig. S2(a), the sorption amount of Cd(II) on MoS2 was promoted by high temperature at each solution pH and also by high pH at each temperature, implying that pH had no impact on temperature effects. All equilibrium data were modeled using the linear forms of Langmuir and Freundlich isotherms by the following equations[
where qmax (mg/g) and KL (L/mg) are the maximum sorption capacity of Cd(II) on per weight unit of MoS2 and the Langmuir affinity parameter, respectively; n and KF (mg1-n·Ln/g) represent the exponent and Freundlich affinity-capacity parameter, respectively. Combined the linear plots of fitted isotherms (Fig. S2(b,c)) with the corresponding isotherm parameters (Table S2), it was concluded that the sorption processes at each pH were more appropriately fitted by Freundlich model than Langmuir model due to higher R2 values, which was consistent with previous studies[
where Kθ is the distribution coefficient, and the values of lnKθ at each pH are assessed by plotting lnKdvs. Ce (Fig. S3(a)). The ∆Sθ at each pH is calculated from the slope of linear plot of ∆Gθ vs. T (Fig. S3(b)). The correlated thermodynamic parameters are recorded in Table S3. It can be inferred that Cd(II) sorption on MoS2 at each solution pH was a spontaneous, endothermic, and irreversible process due to ∆Gθ<0, ∆Hθ>0, and ∆Sθ>0[
References:
[1]WANG Q, YANG L, JIA F, et al. Removal of Cd(II) from water by using nano-scale molybdenum disulphide sheets as adsorbents. J. Mol. Liq., 2018, 263: 526-533.
[2]COLEMAN J N, LOTYA M, O’NEILL A, et al. Two dimensional nanosheets produced by liquid exfoliation of layered materials. Science, 2011, 331: 568-571.
[3]SPLENDIANI A, SUN L, ZHANG Y, et al. Emerging photoluminescence in monolayer MoS2. Nano Lett., 2010, 10: 1271-1275.
[4]KUMAR A S K, JIANG S J, WARCHOL J K. Synthesis and characterization of two-dimensional transition metal dichalcogenide magnetic MoS2@Fe3O4 nanoparticles for adsorption of Cr(VI)/Cr(III). ACS Omega, 2017, 2: 6187-6200.
[5]TONG S, DENG H, WANG L, et al. Multi-functional nanohybrid of ultrathin molybdenum disulfide nanosheets decorated with cerium oxide nanoparticles for preferential uptake of lead (II) ions. Chem. Eng. J., 2018, 335: 22-31.
[6]TEMKIN M J, PYZHEV V. Recent modifications to langmuir isotherms. Acta Physchim., 1940, 12: 217-222.
[7]XUE C, QI P S, LIU Y Z. Adsorption of aquatic Cd2+ using a combination of bacteria and modified carbon fiber. Adsorpt. Sci. Technol., 2017, 36: 857-871.
[8]GU P, ZHANG S, ZHANG C, et al. Two-dimensional MAX-derived titanate nanostructures for efficient removal of Pb(II). Dalton Trans., 2019, 48(6): 2100-2107.
[9]CHEN W, LU Z, XIAO B, et al. Enhanced removal of lead ions from aqueous solution by iron oxide nanomaterials with cobalt and nickel doping. J. Clean. Prod., 2019, 211: 1250-1258.
[10]ZHANG D, NIU H Y, ZHANG X L, et al. Strong adsorption of chlorotetracycline on magnetite nanoparticles. J. Hazard. Mater., 2011, 192: 1088-1093.
[11]ZHANG H, YU X, CHEN L, et al. Study of 63Ni adsorption on NKF-6 zeolite. J. Environ. Radioact., 2010, 101: 1061-1069.
[12]BEKCI Z, SEKI Y, YURDAKOC M K. A study of equilibrium and FTIR, SEM/EDS analysis of trimethoprim adsorption onto K10. J. Mol. Struct., 2007, 827: 67-74.
References
[1] G ZENG, Y LIU, L TANG et al. Enhancement of Cd(II) adsorption by polyacrylic acid modified magnetic mesoporous carbon. Chem. Eng. J., 259, 153-160(2015).
[2] G YANG, L TANG, X LEI et al. Cd(II) removal from aqueous solution by adsorption on ketoglutaric acid-modified magnetic chitosan. Appl. Surf. Sci., 292, 710-716(2014).
[3] L LUO, B MA Y, Z ZHANG S et al. An inventory of trace element inputs to agricultural soils in China. J. Environ. Manage, 90, 2524-2530(2009).
[4] A KHAN T, A CHAUDHRY S, I ALI. Equilibrium uptake, isotherm and kinetic studies of Cd(II) adsorption onto iron oxide activated red mud from aqueous solution. J. Mol. Liq., 202, 165-175(2015).
[5] R AWUAL M, M KHRAISHEH, H ALHARTHI N et al. Efficient detection and adsorption of cadmium(II) ions using innovative nano-composite materials. Chem. Eng. J., 343, 118-127(2018).
[6] Q LIAO, D ZOU, W PAN et al. Highly-efficient scavenging of P(V), Cr(VI), Re(VII) anions onto g-C3N4 nanosheets from aqueous solutions as impacted
[7] L DONG, J YANG, Y MOU et al. Effect of various environmental factors on the adsorption of U(VI) onto biochar derived from rice straw. J. Radioanal. Nucl. Chem., 314, 377-386(2017).
[8] D SHENG G, Q YANG, F PENG et al. Determination of colloidal pyrolusite, Eu(III) and humic substance interaction: a combined batch and EXAFS approach. Chem. Eng. J., 245, 10-16(2014).
[9] J YU S, X WANG X, W PANG H et al. Boron nitride-based materials for the removal of pollutants from aqueous solutions: a review. Chem. Eng. J., 333, 343-360(2018).
[10] W YAO, X WANG, Y LIANG et al. Synthesis of novel flower-like layered double oxides/carbon dots nanocomposites for U(VI) and 241Am(III) efficient removal: batch and EXAFS studies. Chem. Eng. J., 332, 775-786(2018).
[11] J WANG, X WANG X, X ZHAO G et al. Polyvinylpyrrolidone and polyacrylamide intercalated molybdenum disulfide as adsorbents for enhanced removal of chromium(VI) from aqueous solutions. Chem. Eng. J., 334, 569-578(2018).
[12] Q LIAO, S ZOU D, W PAN et al. Highly efficient capture of Eu(III), La(III), Nd(III), Th(IV) from aqueous solutions using g-C3N4 nanosheets. J. Mol. Liq., 252, 351-361(2018).
[13] X WANG X, J YU S, K WANG X. Removal of radionuclides by metal-organic framework-based materials. J. Inorg. Mater., 34, 17-26(2019).
[14] N WANG, H PANG, S YU et al. Investigation of adsorption mechanism of layered double hydroxides and their composites on radioactive uranium: a review. Acta Chim. Sinica, 77, 143-152(2019).
[15] X LIU, R MA, X WANG et al. Graphene oxide-based materials for efficient removal of heavy metal ions from aqueous solution: a review. Environ. Pollut., 252, 62-73(2019).
[16] X WANG X, L CHEN, L WANG et al. Synthesis of novel nanomaterials and their application in efficient removal of radionuclides. Sci. China Chem., 62, 933-967(2019).
[17] B FENG, C YAO, S CHEN et al. Highly efficient and selective recovery of Au(III) from a complex system by molybdenum disulfide nanoflakes. Chem. Eng. J., 350, 692-702(2018).
[18] J CHEN H, J HUANG, L LEI X et al. Adsorption and diffusion of lithium on MoS2 monolayer: the role of strain and concentration.. Int. J. Electrochem. Sci., 8, 2196-2203(2013).
[19] F JIA, Q WANG, J WU et al. Two-dimensional molybdenum disulfide as a superb adsorbent for removing Hg+ from water.. ACS Sustainable Chem. Eng., 5, 7410-7419(2017).
[20] F JIA, X ZHANG, S SONG. AFM study on the adsorption of Hg 2+ on natural molybdenum disulfide in aqueous solutions. Phys. Chem. Chem. Phys., 19, 3837-3844(2017).
[21] Z WANG, B MI. Environmental applications of 2D molybdenum disulfide (MoS2) nanosheet. Environ. Sci. Technol., 51, 8229-8244(2017).
[22] K AI, C RUAN, M SHEN et al. MoS2 nanosheets with widened interlayer spacing for high-efficiency removal of mercury in aquatic systems.. Adv. Funct. Mater., 26, 5542-5549(2016).
[23] S TONG, H DENG, L WANG et al. Multi-functional nanohybrid of ultrathin molybdenum disulfide nanosheets decorated with cerium oxide nanoparticles for preferential uptake of lead (II) ions. Chem. Eng. J., 335, 22-31(2018).
[24] X LI, Q LI, W LINGHU et al. Sorption properties of U(VI) and Th(IV) on two-dimensional molybdenum disulfide (MoS2) nanosheets: effects of pH, ionic strength, contact time, humic acids and temperature.. Environ. Technol. Innov., 11, 328-338(2018).
[25] Q WANG, L YANG, F JIA et al. Removal of Cd(II) from water by using nano-scale molybdenum disulphide sheets as adsorbents. J. Mol. Liq., 263, 526-533(2018).
[26] L ZHI, W ZUO, F CHEN et al. 3D MoS2 composition aerogel as chemosensors and adsorbents for colorimetric detection and high- capacity adsorption of Hg2+.. ACS Sustain. Chem. Eng., 4, 3398-3408(2016).
[27] K AI, C RUAN, M SHEN et al. MoS2 nanosheets with widened interlayer spacing for high-efficiency removal of mercury in aquatic systems.. Adv. Funct. Mater., 26, 5542-5549(2016).
[28] J AGHAGOLI M, H BEYKI M, F SHEMIRANI. Application of dahlia-like molybdenum disulfide nanosheets for solid phase extraction of Co(II) in vegetable and water samples. Food Chem., 223, 8-15(2017).
[29] X GAO, D SHENG G, Y HUANG Y. Mechanism and microstructure of Eu(III) interaction with
[30] L DONG, Q LIAO, W LINGHU et al. Application of EXAFS with a bent crystal analyzer to study the pH-dependent microstructure of Eu(III) onto birnessite. J. Environ. Chem. Eng., 6, 842-848(2018).
[31] F VASCONCELOS I, A HAACK E, A MAURICE P et al. EXAFS analysis of cadmium(II) adsorption to kaolinite.. Chem. Geol., 249, 237-249(2008).
[32] C LIU, I FRENKEL A, A VAIRAVAMURTHY et al. Sorption of cadmium on humic acid: mechanistic and kinetic studies with atomic force microscopy and X-ray absorption fine structure spectroscopy. Can. J. Soil Sci., 81, 337-348(2001).
[33] D SHENG G, T YANG S, M LI Y et al. Retention mechanisms and microstructure of Eu(III) on manganese dioxides studied by batch and high resolution EXAFS technique. Radiochim. Acta, 102, 155-167(2014).
[34] N COLEMAN J, M LOTYA, A O’NEILL et al. Two dimensional nanosheets produced by liquid exfoliation of layered materials.. Science, 331, 568-571(2011).
[35] A SPLENDIANI, L SUN, Y ZHANG et al. Emerging photoluminescence in monolayer MoS2.. Nano Lett., 10, 1271-1275(2010).
[36] K KUMAR A S, J JIANG S, K WARCHOL J. Synthesis and characterization of two-dimensional transition metal dichalcogenide magnetic MoS2@Fe3O4 nanoparticles for adsorption of Cr(VI)/Cr(III). ACS Omega, 2, 6187-6200(2017).
[37] R TAKAMATSU, K ASAKURA, J CHUN W et al. EXAFS studies about the sorption of cadmium ions on montmorillonite. Chem. Lett., 35, 224-225(2006).
[38] X HUANG, T CHEN, X ZOU et al. The adsorption of Cd(II) on manganese oxide investigated by batch and modeling techniques. Int. J. Environ. Res. Public Health, 14, 1145(2017).
[39] E GUECHI, D BEGGAS. Removal of cadmium (II) from water using fibre fruit lufa as biosorbent. Desalin. Water Treat., 94, 181-188(2017).
[40] A ABASIYAN S M, R MAHDANINIA G. Polyvinyl alcohol- based nanocomposite hydrogels containing magnetic laponite RD to remove cadmium. Environ. Sci. Poll. Res., 25, 14977-14988(2018).
[41] F CORBETT J. Pseudo first-order kinetics. J. Chem. Educ., 49, 663(1972).
[42] S HO Y, G MCKAY. A comparison of chemisorption kinetic models applied to pollutant removal on various sorbents. Process. Saf. Environ., 76, 332-340(1998).
[43] H GRAAF G, H SCHOLTENS, J STAMHUIS E et al. Intra-particle diffusion limitations in low-pressure methanol synthesis. Chem. Eng. Sci., 45, 773-783(1990).
[44] S HO Y, G MCKAY. The kinetics of sorption of divalent metal ions onto sphagnum moss peat. Water Rer., 34, 735-742(2000).
[45] J TEMKIN M, V PYZHEV. Recent modifications to Langmuir isotherms. Acta Physchim, 12, 217-222(1940).
[46] C XUE, S QI P, Z LIU Y. Adsorption of aquatic Cd 2+ using a combination of bacteria and modified carbon fiber. Adsorpt. Sci. Technol., 36, 857-871(2017).
[47] P GU, S ZHANG, C ZHANG et al. Two-dimensional MAX- derived titanate nanostructures for efficient removal of Pb(II). Dalton Trans., 48, 2100-2107(2019).
[48] W CHEN, Z LU, B XIAO et al. Enhanced removal of lead ions from aqueous solution by iron oxide nanomaterials with cobalt and nickel doping. J. Clean. Prod., 211, 1250-1258(2019).
[49] D ZHANG, Y NIU H, L ZHANG X et al. Strong adsorption of chlorotetracycline on magnetite nanoparticles. J. Hazard. Mater., 192, 1088-1093(2011).
[50] H ZHANG, X YU, L CHEN et al. Study of 63Ni adsorption on NKF-6 zeolite.. J. Environ. Radioact., 101, 1061-1069(2010).
[51] Z BEKCI, Y SEKI, K YURDAKOC M. A study of equilibrium and FTIR, SEM/EDS analysis of trimethoprim adsorption onto K10. J. Mol. Struct., 827, 67-74(2007).
[52] M GRÄFE, B SINGH, M BALASUBRAMANIAN. Surface speciation of Cd(II) and Pb(II) on kaolinite by EXAFS spectroscopy. J. Colloid Interf. Sci., 315, 21-32(2007).
[53] G SHENG, H DONG, R SHEN et al. Microscopic insights into the temperature dependent adsorption of Eu(III) onto titanate nanotubes studied by FTIR, XPS, XAFS and batch technique. Chem. Eng. J., 217, 486-494(2013).
Set citation alerts for the article
Please enter your email address



