Search by keywords or author
Journals > > Topics > Microscopy
Microscopy|5 Article(s)
A Fluorescence Lifetime Retrieval Algorithm Based on LSTM Neural Network
Biyu YANG, and Yue XU
Fluorescence lifetime imaging technology utilizes the decay difference of the emitted fluorescences to distinguish different fluorescent molecules, which is widely used in biomedicine, chemical analysis, and life science. The quality of fluorescence lifetime imaging depends on fluorescence lifetime measurement techniques and retrieval algorithms. The measurement method based on time-correlated single-photon counting technology has become the main stream fluorescence lifetime measurement method in the field of biological research because of its high accuracy and easy low-light detection. Traditional fluorescence lifetime retrieval algorithms based on time-correlated single-photon counting technology are not suitable for the extraction of fast, high-precision, and long fluorescence lifetime. Most of the long fluorescence lifetime substances are quantum dots. In recent years, emerging deep learning techniques have also been gradually used for fluorescence lifetime retrieval, mainly realizing fluorescence lifetime imaging with fluorescence lifetimes in the range of 10 ns. Therefore, it is urgent to develop a new fluorescence lifetime retrieval algorithm to solve the constraints of retrieval accuracy and speed in a wide fluorescence lifetime range.To solve the problem of low accuracy of fluorescence lifetime retrieval in a large dynamic range, a fluorescence lifetime retrieval algorithm based on long short-term memory neural network is proposed in this paper. The algorithm uses a multi-layer long-short-term memory neural network with a time-series memory function to realize the feature extraction of the fluorescence lifetime decay histogram data which is based on time-correlated single photon counting. The unique gate structure of long-short-term memory neural network can realize the protection and control of time series information. What's more, deep learning technology is used to learn a large number of various fluorescence lifetime decay information, establish a corresponding relationship between histogram and fluorescence lifetime, and then the weight value and bias coefficients of the network are updated to make the training model more suitable for fluorescence lifetime retrieval. To train the model, the grid search method is used to select the hyperparameters of the neural network model, including a number of neurons and network layers. To make the simulated data closer to the real experimental data, the data set for model training is a time series generated by a computer simulation of the time-correlated single-photon counting process in the presence of Poisson noise. The generated time series is the series corresponding to 20 000 fluorescence lifetime decay histograms uniformly distributed in the range of 100 ns. The data were normalized to eliminate the order-of-magnitude differences, and to avoid large order-of-magnitude differences which would reduce the accuracy of the predictions. The prediction accuracy of the randomly generated 1~100 ns fluorescence lifetime outside the training data set is supposed as the evaluation standard, and the optimal model including 3 layers of LSTM network is selected for the subsequent fluorescence lifetime retrieval. Monte Carlo simulation results indicate that the proposed retrieval algorithm achieves a retrieval accuracy of 95% in the fluorescence lifetime range of 1~90 ns even when the number of photons is 5 000 which is conducive to the fluorescence lifetime imaging. In the case of the same number of photons, the retrieval range is increased by 4.5 times in comparison with the center-of-mass method. Moreover, the proposed method achieves higher retrieval accuracy of the long lifetimes than the traditional least squares method. For the imaging of 32×32 arrays, after several experimental calculations, it is shown that the center-of-mass method can complete the computing in 0.07 s, the least-squares method takes about 77 s, and the proposed algorithm takes about 9.7 s under the conditions of Windows11 (64-bit) operating system, 16 GB memory, and Intel(R) Core(TM) i5-1157G7 processor. The results reveal that neural network not only provides comparable or even better performances but also offers much faster high-throughput data analysis. A shorter time will be used to complete the array imaging when the hardware conditions are improved which provides the possibility for real-time imaging. The proposed algorithm can significantly broaden the fluorescence lifetime reduction range with high retrieval accuracy, thus, it is suitable for accurate fluorescence lifetime retrieval imaging with a single exponential large dynamic range. Fluorescence lifetime imaging technology utilizes the decay difference of the emitted fluorescences to distinguish different fluorescent molecules, which is widely used in biomedicine, chemical analysis, and life science. The quality of fluorescence lifetime imaging depends on fluorescence lifetime measurement techniques and retrieval algorithms. The measurement method based on time-correlated single-photon counting technology has become the main stream fluorescence lifetime measurement method in the field of biological research because of its high accuracy and easy low-light detection. Traditional fluorescence lifetime retrieval algorithms based on time-correlated single-photon counting technology are not suitable for the extraction of fast, high-precision, and long fluorescence lifetime. Most of the long fluorescence lifetime substances are quantum dots. In recent years, emerging deep learning techniques have also been gradually used for fluorescence lifetime retrieval, mainly realizing fluorescence lifetime imaging with fluorescence lifetimes in the range of 10 ns. Therefore, it is urgent to develop a new fluorescence lifetime retrieval algorithm to solve the constraints of retrieval accuracy and speed in a wide fluorescence lifetime range.To solve the problem of low accuracy of fluorescence lifetime retrieval in a large dynamic range, a fluorescence lifetime retrieval algorithm based on long short-term memory neural network is proposed in this paper. The algorithm uses a multi-layer long-short-term memory neural network with a time-series memory function to realize the feature extraction of the fluorescence lifetime decay histogram data which is based on time-correlated single photon counting. The unique gate structure of long-short-term memory neural network can realize the protection and control of time series information. What's more, deep learning technology is used to learn a large number of various fluorescence lifetime decay information, establish a corresponding relationship between histogram and fluorescence lifetime, and then the weight value and bias coefficients of the network are updated to make the training model more suitable for fluorescence lifetime retrieval. To train the model, the grid search method is used to select the hyperparameters of the neural network model, including a number of neurons and network layers. To make the simulated data closer to the real experimental data, the data set for model training is a time series generated by a computer simulation of the time-correlated single-photon counting process in the presence of Poisson noise. The generated time series is the series corresponding to 20 000 fluorescence lifetime decay histograms uniformly distributed in the range of 100 ns. The data were normalized to eliminate the order-of-magnitude differences, and to avoid large order-of-magnitude differences which would reduce the accuracy of the predictions. The prediction accuracy of the randomly generated 1~100 ns fluorescence lifetime outside the training data set is supposed as the evaluation standard, and the optimal model including 3 layers of LSTM network is selected for the subsequent fluorescence lifetime retrieval. Monte Carlo simulation results indicate that the proposed retrieval algorithm achieves a retrieval accuracy of 95% in the fluorescence lifetime range of 1~90 ns even when the number of photons is 5 000 which is conducive to the fluorescence lifetime imaging. In the case of the same number of photons, the retrieval range is increased by 4.5 times in comparison with the center-of-mass method. Moreover, the proposed method achieves higher retrieval accuracy of the long lifetimes than the traditional least squares method. For the imaging of 32×32 arrays, after several experimental calculations, it is shown that the center-of-mass method can complete the computing in 0.07 s, the least-squares method takes about 77 s, and the proposed algorithm takes about 9.7 s under the conditions of Windows11 (64-bit) operating system, 16 GB memory, and Intel(R) Core(TM) i5-1157G7 processor. The results reveal that neural network not only provides comparable or even better performances but also offers much faster high-throughput data analysis. A shorter time will be used to complete the array imaging when the hardware conditions are improved which provides the possibility for real-time imaging. The proposed algorithm can significantly broaden the fluorescence lifetime reduction range with high retrieval accuracy, thus, it is suitable for accurate fluorescence lifetime retrieval imaging with a single exponential large dynamic range.
Acta Photonica Sinica
- Publication Date: Jun. 25, 2022
- Vol. 51, Issue 6, 0618002 (2022)
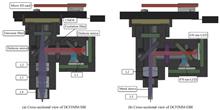
In Vivo,Dual-color Fluorescent Imaging Miniature Microscope
Kaiqiu LAN, Xibin YANG, Baoteng XU, Jialin LIU, Wei ZHOU, and Daxi XIONG
In recent years, neuroscientists have become more and more interested in brain imaging of conscious and free-behaving animals, hoping to obtain nerve impulse signals in the brains of free-behaving experimental animals, especially certain types of cellular activity may be inhibited by anesthesia. Combined with the Genetically Encoded Calcium Indicator (GECI), the miniature microscope has the ability to image the brain of free-behaving animals and obtain the signal of nerve impulse. The miniature microscope is then widely used in the study of brain science. Currently, most optical systems of miniature microscopes are limited by chromatic aberration due to the use of Gradient Index Lenses (GRIN), which does not meet the experimental requirement of the two-color fluorescent imaging effect. Dual-color fluorescence imaging miniature microscopes have a number of advantages, such as the ability to compare the activities of two different cell populations in the same brain region of a free-behaving animal combined with GECIs which have distinguishable color spectrums, or it can be used for the motion correction. Therefore, a Dual-color Fluorescent Imaging Miniature Microscope (DCFIMM) is developed. Firstly, in order to enhance the dual-color fluorescent imaging capability of the miniature microscope, a micro achromatic lens is designed to replace the gradient index lens. The miniature achromatic lens is composed of double cemented lenses, which forms an infinity correction optical system with the imaging lens. And according to the application direction of cerebral cortex imaging and deep brain imaging, DCFIMM-SBI (superficial brain imaging) and DCFIMM-DBI (deep brain imaging) are designed, both have a larger imaging field of view than the monochromatic fluorescence imaging miniature microscope with grin lens, which are 1.10 mm×1.10 mm and 0.77 mm×0.77 mm respectively. Meanwhile, the dual-band filter for green and near-infrared is used to reduce fluorescent crosstalk. Secondly, a data acquisition circuit is designed to alternately trigger two LEDs with different wavelengths with the frame rate of the CMOS camera. Therefore, the green fluorescent information and near-infrared fluorescent information can be obtained in odd-numbered frames and even-numbered frames, respectively. Our system can realize the imaging speed of 10 fps with the ability of dual-color fluorescence imaging. Thirdly, the video data is stored in a micro SD card. DCFIMM is not limited by the wire transmission. Finally, the structure design of our DCFIMM is optimized. The whole weight of our DCFIMM is 4.8 g (6.2 g with a battery). The experimental results of the USAF 1951 high-resolution target show that the achievable resolution of our DCFIMM is 3.47 μm, which is comparable with monochromatic fluorescent imaging using a miniature microscope with GRIN lens. In the dual-color fluorescent imaging experiment for the hybrid microsphere, DCFIMM can distinguish the fluorescent microspheres of different colors. Compared with the experimental results of monochromatic fluorescence imaging miniature microscope with grin lens, it is found that the chromatic aberration of the DCFIMM optical system has also been well corrected, which demonstrates that our DCFIMM has the ability to distinguish fluorescence of different wavelengths. The proposed DCFIMM in this paper shows promising and wide applications for brain science research. In recent years, neuroscientists have become more and more interested in brain imaging of conscious and free-behaving animals, hoping to obtain nerve impulse signals in the brains of free-behaving experimental animals, especially certain types of cellular activity may be inhibited by anesthesia. Combined with the Genetically Encoded Calcium Indicator (GECI), the miniature microscope has the ability to image the brain of free-behaving animals and obtain the signal of nerve impulse. The miniature microscope is then widely used in the study of brain science. Currently, most optical systems of miniature microscopes are limited by chromatic aberration due to the use of Gradient Index Lenses (GRIN), which does not meet the experimental requirement of the two-color fluorescent imaging effect. Dual-color fluorescence imaging miniature microscopes have a number of advantages, such as the ability to compare the activities of two different cell populations in the same brain region of a free-behaving animal combined with GECIs which have distinguishable color spectrums, or it can be used for the motion correction. Therefore, a Dual-color Fluorescent Imaging Miniature Microscope (DCFIMM) is developed. Firstly, in order to enhance the dual-color fluorescent imaging capability of the miniature microscope, a micro achromatic lens is designed to replace the gradient index lens. The miniature achromatic lens is composed of double cemented lenses, which forms an infinity correction optical system with the imaging lens. And according to the application direction of cerebral cortex imaging and deep brain imaging, DCFIMM-SBI (superficial brain imaging) and DCFIMM-DBI (deep brain imaging) are designed, both have a larger imaging field of view than the monochromatic fluorescence imaging miniature microscope with grin lens, which are 1.10 mm×1.10 mm and 0.77 mm×0.77 mm respectively. Meanwhile, the dual-band filter for green and near-infrared is used to reduce fluorescent crosstalk. Secondly, a data acquisition circuit is designed to alternately trigger two LEDs with different wavelengths with the frame rate of the CMOS camera. Therefore, the green fluorescent information and near-infrared fluorescent information can be obtained in odd-numbered frames and even-numbered frames, respectively. Our system can realize the imaging speed of 10 fps with the ability of dual-color fluorescence imaging. Thirdly, the video data is stored in a micro SD card. DCFIMM is not limited by the wire transmission. Finally, the structure design of our DCFIMM is optimized. The whole weight of our DCFIMM is 4.8 g (6.2 g with a battery). The experimental results of the USAF 1951 high-resolution target show that the achievable resolution of our DCFIMM is 3.47 μm, which is comparable with monochromatic fluorescent imaging using a miniature microscope with GRIN lens. In the dual-color fluorescent imaging experiment for the hybrid microsphere, DCFIMM can distinguish the fluorescent microspheres of different colors. Compared with the experimental results of monochromatic fluorescence imaging miniature microscope with grin lens, it is found that the chromatic aberration of the DCFIMM optical system has also been well corrected, which demonstrates that our DCFIMM has the ability to distinguish fluorescence of different wavelengths. The proposed DCFIMM in this paper shows promising and wide applications for brain science research.
Acta Photonica Sinica
- Publication Date: Jun. 25, 2022
- Vol. 51, Issue 6, 0618001 (2022)
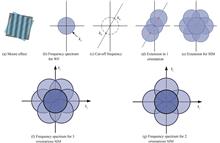
Investigation on Super-resolution Image Reconstruction for Structured Illumination Microscopy in the Spatial Domain
Caihong HUANG, Xingxing ZHU, Wei GUO, Dingrong YI, Fujiang JIN, and Lei HUANG
The spatial resolution of traditional optical microscopy imaging technology is limited by the optical diffraction limit, which can only reach the order of half wavelength of the incident wavelength, which greatly limits the application scope of optical microscopy technology. Among the mainstream optical super-resolution microscopy techniques, Structured Illumination Microscopy (SIM) is an attractive choice for fast super-resolution microscopy due to its fast imaging speed, low phototoxicity, and no additional complex sample preparation process. SIM technology applies periodic sinusoidal fringe structured light with a spatial frequency close to the diffraction limit to the sample. The high-frequency information that could not pass through the sample is now converted into a low-frequency "Moire fringe", which couples the high-frequency information of the sample beyond the diffraction limit to the imaging. The frequency region detectable by the system can theoretically double the lateral resolution of the microscope. The SIM image reconstruction algorithm is the key to determine the final super-resolution image quality. Traditional frequency-domain image reconstruction algorithms need to estimate the initial phase, spatial frequency and other parameters of the structured light field. It is also necessary to perform multiple Fourier transforms between the spatial domain and the frequency domain. The operation speed is slow affecting its application in real-time dynamic imaging and other fields. The proposed spatial-domain super-resolution imaging algorithms are currently limited to solving the 2π/3 phase shift for structured light super-resolution reconstruction. In the traditional structured illumination microscopy based on the projection of the digital micromirror device, the structured fringes with a period of 4 pixels need to adopt a phase shift interval of π/2. The spatial domain reconstruction algorithm under this application has not been reported yet. Here, a super-resolution image reconstruction for SIM in the Spatial domain is proposed. The algorithm is called differential SIM, or DIFF-SIM for short. First, three structured light illumination images with phases 0, π/2, and π are obtained. The two adjacent original images are subtracted to achieve the elimination of background interference, at which point we obtain two new expressions. Simplify the subtracted expression using the two-angle sum-difference formula of trigonometric functions. Then, we construct a new complex function, and take the simplified two expressions as the complex real part and the complex imaginary part respectively. Next, Euler's formula is used to simplify the complex number to the e-exponential function. Using the convolution formula, the function is transformed, and then the Fourier transform frequency shift characteristics are used to further simplify the expression. Finally, by taking the modulo of the result, the frequency domain spectrum of the system is expanded to obtain a super-resolution reconstructed image. Theoretically, when the structured light frequency is equal to the cutoff frequency of the system, the algorithm can double the lateral resolution of conventional microscopy systems. It is verified by simulation that the resolution of DIFF-SIM is nearly 1 time higher than that of wide-field imaging. Meanwhile, the super-resolution reconstruction effect of DIFF-SIM and FairSIM, the frequency domain reconstruction method of SIM, is the same. In a projected structured illumination microscope based on digital micromirror devices, spatial domain super-resolution reconstruction experiments were performed on fluorescent microspheres and bovine pulmonary artery endothelial cells. The system employs computer-controlled digital micromirror devices for fast fringe generation and uses multicolor light-emitting diodes for illumination. Firstly, the system resolution analysis experiment was carried out, and the system Point Spread Function (PSF) obtained by the fluorescent microspheres proved that the algorithm could expand the system resolution. The experimental results of the resolution expansion are close to the theoretical value. Then, super-resolution reconstruction of bovine pulmonary artery endothelial cells was performed, and the experimental results were compared using the frequency domain method FairSIM. The experimental results show that the DIFF-SIM algorithm can obtain super-resolution image reconstruction effects similar to FairSIM. In the defocus experimental verification of the groove of the coin, it is proven that the algorithm can eliminate the interference of the defocused background focal plane information similar to laser scanning confocal microscopy (LSCM). To evaluate the efficiency of the DIFF-SIM algorithm, a comparison of the FairSIM and DIFF-SIM algorithms is performed on the same computer, and the execution speed of DIFF-SIM is approximately 5 times faster than that of FairSIM. This study is conducive to expanding the application range of the SIM method, helping to take advantage of the technical advantages of SIM with low light dose and low phototoxicity, and has good application potential in dynamic imaging of living cells and long-term monitoring. The spatial resolution of traditional optical microscopy imaging technology is limited by the optical diffraction limit, which can only reach the order of half wavelength of the incident wavelength, which greatly limits the application scope of optical microscopy technology. Among the mainstream optical super-resolution microscopy techniques, Structured Illumination Microscopy (SIM) is an attractive choice for fast super-resolution microscopy due to its fast imaging speed, low phototoxicity, and no additional complex sample preparation process. SIM technology applies periodic sinusoidal fringe structured light with a spatial frequency close to the diffraction limit to the sample. The high-frequency information that could not pass through the sample is now converted into a low-frequency "Moire fringe", which couples the high-frequency information of the sample beyond the diffraction limit to the imaging. The frequency region detectable by the system can theoretically double the lateral resolution of the microscope. The SIM image reconstruction algorithm is the key to determine the final super-resolution image quality. Traditional frequency-domain image reconstruction algorithms need to estimate the initial phase, spatial frequency and other parameters of the structured light field. It is also necessary to perform multiple Fourier transforms between the spatial domain and the frequency domain. The operation speed is slow affecting its application in real-time dynamic imaging and other fields. The proposed spatial-domain super-resolution imaging algorithms are currently limited to solving the 2π/3 phase shift for structured light super-resolution reconstruction. In the traditional structured illumination microscopy based on the projection of the digital micromirror device, the structured fringes with a period of 4 pixels need to adopt a phase shift interval of π/2. The spatial domain reconstruction algorithm under this application has not been reported yet. Here, a super-resolution image reconstruction for SIM in the Spatial domain is proposed. The algorithm is called differential SIM, or DIFF-SIM for short. First, three structured light illumination images with phases 0, π/2, and π are obtained. The two adjacent original images are subtracted to achieve the elimination of background interference, at which point we obtain two new expressions. Simplify the subtracted expression using the two-angle sum-difference formula of trigonometric functions. Then, we construct a new complex function, and take the simplified two expressions as the complex real part and the complex imaginary part respectively. Next, Euler's formula is used to simplify the complex number to the e-exponential function. Using the convolution formula, the function is transformed, and then the Fourier transform frequency shift characteristics are used to further simplify the expression. Finally, by taking the modulo of the result, the frequency domain spectrum of the system is expanded to obtain a super-resolution reconstructed image. Theoretically, when the structured light frequency is equal to the cutoff frequency of the system, the algorithm can double the lateral resolution of conventional microscopy systems. It is verified by simulation that the resolution of DIFF-SIM is nearly 1 time higher than that of wide-field imaging. Meanwhile, the super-resolution reconstruction effect of DIFF-SIM and FairSIM, the frequency domain reconstruction method of SIM, is the same. In a projected structured illumination microscope based on digital micromirror devices, spatial domain super-resolution reconstruction experiments were performed on fluorescent microspheres and bovine pulmonary artery endothelial cells. The system employs computer-controlled digital micromirror devices for fast fringe generation and uses multicolor light-emitting diodes for illumination. Firstly, the system resolution analysis experiment was carried out, and the system Point Spread Function (PSF) obtained by the fluorescent microspheres proved that the algorithm could expand the system resolution. The experimental results of the resolution expansion are close to the theoretical value. Then, super-resolution reconstruction of bovine pulmonary artery endothelial cells was performed, and the experimental results were compared using the frequency domain method FairSIM. The experimental results show that the DIFF-SIM algorithm can obtain super-resolution image reconstruction effects similar to FairSIM. In the defocus experimental verification of the groove of the coin, it is proven that the algorithm can eliminate the interference of the defocused background focal plane information similar to laser scanning confocal microscopy (LSCM). To evaluate the efficiency of the DIFF-SIM algorithm, a comparison of the FairSIM and DIFF-SIM algorithms is performed on the same computer, and the execution speed of DIFF-SIM is approximately 5 times faster than that of FairSIM. This study is conducive to expanding the application range of the SIM method, helping to take advantage of the technical advantages of SIM with low light dose and low phototoxicity, and has good application potential in dynamic imaging of living cells and long-term monitoring.
Acta Photonica Sinica
- Publication Date: Nov. 25, 2022
- Vol. 51, Issue 11, 1118002 (2022)
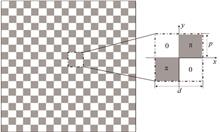
Quadriwave Lateral Shearing Interferometry Quantitative Phase Imaging Based on 2D Ronchi Phase Grating
Jinwei SONG, Junwei MIN, Xun YUAN, Yuge XUE, and Baoli YAO
Quantitative Phase Imaging (QPI) is a technique that can measure the phase map of the light field. It has the characteristics of label-free, non-invasive and three-dimensional observation and has been widely used in bioimaging and industrial inspection. A number of techniques have been developed to measure phase information of objects, including the interferometric method such as Digital Holographic Microscopy (DHM), and the non-interferometric method such as the Fourier Ptychography Microscopy (FPM), Transport of Intensity Equation (TIE) method and so on. The interferometric method has high measurement accuracy but a complex experimental setup sensitive to the environmental disturbance. The non-interferometric method recovers phase from the intensity patterns of objects, but requires iterative calculation or multiple images recorded at different positions, which makes the imaging speed slow and unsuitable for real-time observation. The quantitative phase imaging based on Quadriwave Lateral Shearing Interferometry (QLSI) has the advantages of the referenceless beam, simple configuration, high stability and fast imaging speed. In the existing studies, Cross Grating (CG), Modified Hartmann Mask (MHM), Randomly Encoded Hybrid Grating (REHG) and other splitter elements were used for QLSI. The cross grating has low diffraction efficiency and energy utilization rate (~10%) for the four beams of first-order diffraction. The MHM and REHG can concentrate the diffracted light energy on the four first-order diffraction beams. But the MHM still has a low energy utilization rate (~37%), and the REHG has a complex structure for fabrication.This paper proposes a quantitative phase imaging method based on QLSI using a two-dimensional (2D) Ronchi phase grating. The light incident to the 2D Ronchi phase grating is diffracted mainly with energy concentrated on the four first-order diffraction beams, occupying 65.7% of the total incident energy. The object light carrying the sample's phase information is imprinted to the 2D Ronchi phase grating and then copied into four beams, which interfere with each other to produce the quadriwave lateral shearing interferogram. The quantitative phase image of the sample is reconstructed by Fourier analysis of the interferogram. The influence of the grating period on the QLSI imaging is analyzed theoretically, and the optimal grating period is determined to be six times of the pixel size of the detector. This match can make the best use of the spatial bandwidth product of detector and achieve high resolution image. The influence of the illumination wavelength on the phase reconstruction is theoretically analyzed, which shows that the proposed method is insensitive to the illumination wavelength. The feasibility of quantitative phase imaging under wide spectral light illumination source is demonstrated. The compact QLSI module is constructed with the pixel size of 9 μm×9 μm of the detector and the period of 54 μm of the 2D Ronchi grating. The grating period is precisely six times of the pixel size, meeting the requirement of the optimal condition. The QLSI module is directly connected to a conventional optical microscope to implement the QPI imaging of e.g., Polymethyl Methacrylate (PMMA) microspheres, microlens arrays and staphylococcus section. The relative error of phase experimentally measured is about 1.8%, proving that the method has a high precision of phase measurement. The experimental results also show that the method can be used for quantitative phase imaging with a wide-spectrum light source, making it easily combined with conventional optical microscopes to have a great application potential in biomedicine, three-dimensional topography measurement and other related fields. Quantitative Phase Imaging (QPI) is a technique that can measure the phase map of the light field. It has the characteristics of label-free, non-invasive and three-dimensional observation and has been widely used in bioimaging and industrial inspection. A number of techniques have been developed to measure phase information of objects, including the interferometric method such as Digital Holographic Microscopy (DHM), and the non-interferometric method such as the Fourier Ptychography Microscopy (FPM), Transport of Intensity Equation (TIE) method and so on. The interferometric method has high measurement accuracy but a complex experimental setup sensitive to the environmental disturbance. The non-interferometric method recovers phase from the intensity patterns of objects, but requires iterative calculation or multiple images recorded at different positions, which makes the imaging speed slow and unsuitable for real-time observation. The quantitative phase imaging based on Quadriwave Lateral Shearing Interferometry (QLSI) has the advantages of the referenceless beam, simple configuration, high stability and fast imaging speed. In the existing studies, Cross Grating (CG), Modified Hartmann Mask (MHM), Randomly Encoded Hybrid Grating (REHG) and other splitter elements were used for QLSI. The cross grating has low diffraction efficiency and energy utilization rate (~10%) for the four beams of first-order diffraction. The MHM and REHG can concentrate the diffracted light energy on the four first-order diffraction beams. But the MHM still has a low energy utilization rate (~37%), and the REHG has a complex structure for fabrication.This paper proposes a quantitative phase imaging method based on QLSI using a two-dimensional (2D) Ronchi phase grating. The light incident to the 2D Ronchi phase grating is diffracted mainly with energy concentrated on the four first-order diffraction beams, occupying 65.7% of the total incident energy. The object light carrying the sample's phase information is imprinted to the 2D Ronchi phase grating and then copied into four beams, which interfere with each other to produce the quadriwave lateral shearing interferogram. The quantitative phase image of the sample is reconstructed by Fourier analysis of the interferogram. The influence of the grating period on the QLSI imaging is analyzed theoretically, and the optimal grating period is determined to be six times of the pixel size of the detector. This match can make the best use of the spatial bandwidth product of detector and achieve high resolution image. The influence of the illumination wavelength on the phase reconstruction is theoretically analyzed, which shows that the proposed method is insensitive to the illumination wavelength. The feasibility of quantitative phase imaging under wide spectral light illumination source is demonstrated. The compact QLSI module is constructed with the pixel size of 9 μm×9 μm of the detector and the period of 54 μm of the 2D Ronchi grating. The grating period is precisely six times of the pixel size, meeting the requirement of the optimal condition. The QLSI module is directly connected to a conventional optical microscope to implement the QPI imaging of e.g., Polymethyl Methacrylate (PMMA) microspheres, microlens arrays and staphylococcus section. The relative error of phase experimentally measured is about 1.8%, proving that the method has a high precision of phase measurement. The experimental results also show that the method can be used for quantitative phase imaging with a wide-spectrum light source, making it easily combined with conventional optical microscopes to have a great application potential in biomedicine, three-dimensional topography measurement and other related fields.
Acta Photonica Sinica
- Publication Date: Nov. 25, 2022
- Vol. 51, Issue 11, 1118001 (2022)
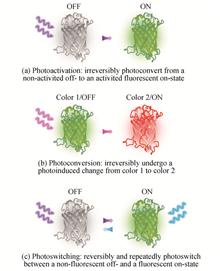
Progress and Prospect of Research on Single-molecule Localization Super-resolution Microscopy(Invited Review)
Sha AN, Dan DAN, Xiang-hua YU, Tong PENG, and Bao-li YAO
Super-resolution optical microscopy breaks through the diffraction limit and becomes a powerful tool for the modern biomedical research with the development of novel fluorescence probes, advanced lasers, high sensitivity photodetectors, etc. Single-molecule Localization Microscopy (SMLM), as one of the super-resolution technologies, can resolve the subcellular structures in nanoscale by using the photoswitching effect of certain fluorescence probes. In this paper, the principle and implementation of SMLM are introduced, the applications in the fields of cytobiology, tissue biology and neuroscience are presented, furthermore, the development trends and the futher investigated directions of this technique are discussed, providing references for the relevant research fields. The continuous innovation of super-resolution microscopy will promote the development of life science. Super-resolution optical microscopy breaks through the diffraction limit and becomes a powerful tool for the modern biomedical research with the development of novel fluorescence probes, advanced lasers, high sensitivity photodetectors, etc. Single-molecule Localization Microscopy (SMLM), as one of the super-resolution technologies, can resolve the subcellular structures in nanoscale by using the photoswitching effect of certain fluorescence probes. In this paper, the principle and implementation of SMLM are introduced, the applications in the fields of cytobiology, tissue biology and neuroscience are presented, furthermore, the development trends and the futher investigated directions of this technique are discussed, providing references for the relevant research fields. The continuous innovation of super-resolution microscopy will promote the development of life science.
Acta Photonica Sinica
- Publication Date: Jan. 01, 2020
- Vol. 49, Issue 9, 0918001 (2020)
Topics