Refat Moamen S., Salman Mahmoud, El-Didamony Akram M., Fetooh Hammad, Abd El-Maksoud Eman S.E., El-Sayed Mohamed Y.. Spectroscopic and Fluorescence Studies on the Trivalent Ce, Eu, Nd and La Metal Ions Rhodamine C Florescent Dye Complexes[J]. Spectroscopy and Spectral Analysis, 2021, 41(10): 3311
Search by keywords or author
- Spectroscopy and Spectral Analysis
- Vol. 41, Issue 10, 3311 (2021)
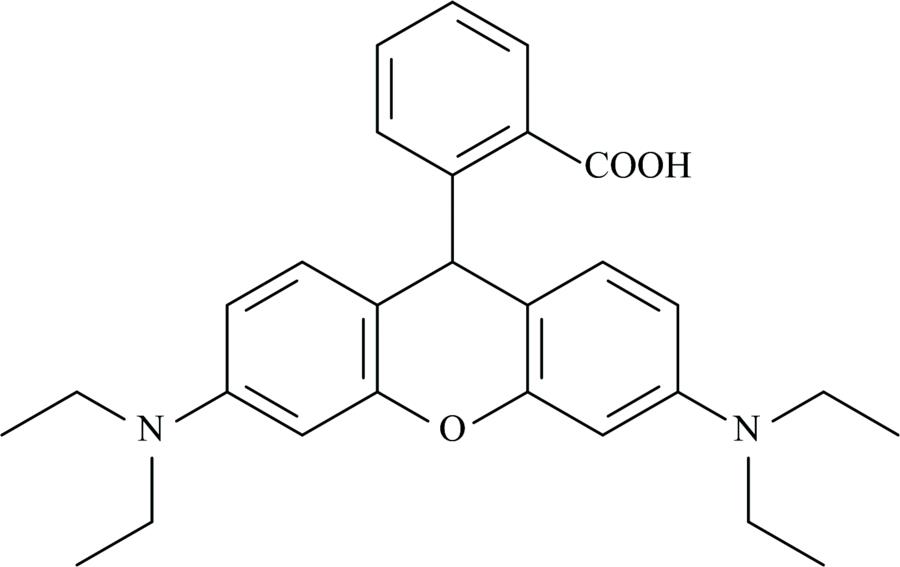
Fig. 1. Structure of Rhodamine C (RHC)
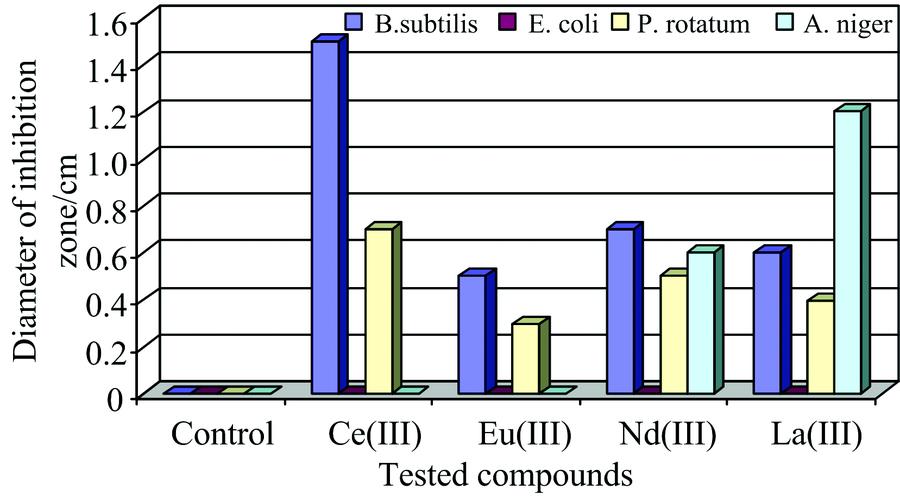
Fig. 2. Statistical representation for antibacterial and antifungal activities of the synthesized complexes
Fig. 3. Modes of the chelation of RHC complexes (where M=Eu(Ⅲ), La(Ⅲ), Nd(Ⅲ) and Ce(Ⅲ), and X =2, 3, 4 and 5, respectively)
Fig. 4. Excitation and emission spectra of rhodamine C
a : Excitation spectrum (λ em=508 nm); b : Emission spectrum (λ ex=550 nm)
Fig. 5. Fluorescence spectra of rhodamine C in absence and presence of varying concentration of Gd3+(λ ex=550 nm)
a : Without Gd3+; b : 2×10-5 mol·L-2; c : 4×10-5 mol·L-1
Fig. 6. Fluorescence spectra of rhodamine C in absence and presence of varying concentration of La3+ (λ ex=550 nm)
a : Without La3+; b : 2×10-5 mol·L-2; c : 4×10-5 mol·L-1
Fig. 7. Fluorescence spectra of rhodamine C in absence and presence of varying concentration of Ce3+ (λ ex=550 nm)
a : Without Ce3+; b : 2×10-5 mol·L-2; c : 4×10-5 mol·L-1
Fig. 8. Fluorescence spectra of rhodamine C in absence and presence of varying concentration of Tb3+ (λ ex=550 nm)
a : Without Tb3+; b : 2×10-5 mol·L-2; c : 4×10-5 mol·L-1
Fig. 9. Fluorescence spectra of rhodamine C in absence and presence of varying concentration of Th4+ (λ ex=550 nm)
a : Without Th3+; b : 2×10-5 mol·L-2; c : 4×10-5 mol·L-1
| |||||||||||||||||||||||||||||||||||||||
Table 1. Elemental and physical data of Rhodamine C metal complexes
|
Table 2. IR Frequencies of RHC and the Ce(Ⅲ), Eu(Ⅲ), Nd(Ⅲ), and La(Ⅲ) complexes
| ||||||||||||||||||||||||||||||||||
Table 3. Antimicrobial activity of the synthesized complexes

Set citation alerts for the article
Please enter your email address



