Yunke Zhou, Zhiyi Yuan, Xuerui Gong, Muhammad D. Birowosuto, Cuong Dang, Yu-Cheng Chen. Dynamic photonic barcodes for molecular detection based on cavity-enhanced energy transfer[J]. Advanced Photonics, 2020, 2(6): 066002
Search by keywords or author
- Advanced Photonics
- Vol. 2, Issue 6, 066002 (2020)
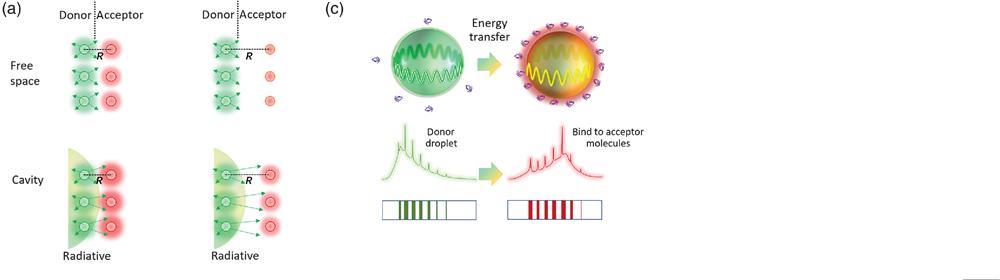
Fig. 1. (a) Comparison of nonradiative FRET and radiative energy transfer, with and without the cavity effect. Left panel: the distance (

Fig. 2. (a) and (b) Comparison between WGM emission spectra obtained from (a) Coumarin 6 microdroplet (with the donor in the cavity) and (b) pure microdroplet (without donor in the cavity) after adding
Fig. 3. MFL spectra and the corresponding photonic barcodes under different cavity diameters of (a) 3.54, (b) 6.53, or (c) Supplementary Material ). The excitation LED wavelength: 430 to 490 nm and
Fig. 4. (a)–(d) Equilibrium WGM spectra of Coumarin 6 microdroplets before (green curve) and after (pink curve) adding (a) 500 nM, (b)
Fig. 5. (a) Schematic illustration of the Biotin-Atto 550 molecules binding to SA-coated microdroplet. (b) Normalized excitation (dashed line) and emission (solid line) spectra of BODIPY-R6G (green) and Atto 550 (red). (c) Comparison of the WGM spectra before and after adding 500 nM Biotin-Atto 550. The inset shows details of the spectral line. The corresponding photonic barcodes are plotted below. Excitation wavelength: 430 to 490 nm; droplet

Set citation alerts for the article
Please enter your email address



