Cheng Jin, Chi Liu, Lingjie Kong. High-axial-resolution optical stimulation of neurons in vivo via two-photon optogenetics with speckle-free beaded-ring patterns[J]. Photonics Research, 2022, 10(6): 1367
Search by keywords or author
- Photonics Research
- Vol. 10, Issue 6, 1367 (2022)
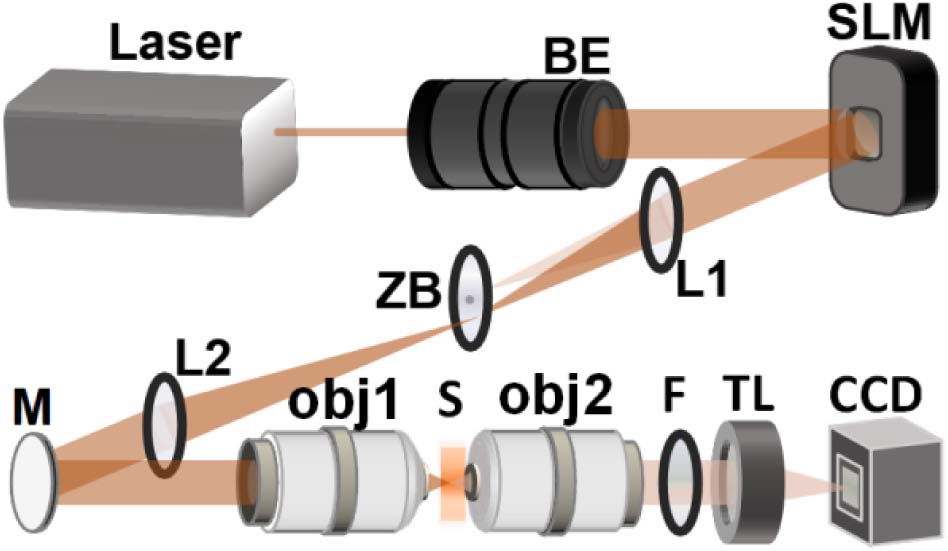
Fig. 1. Detection system with opposite-facing objectives. BE, beam expander; ZB, zero-order blocker; L, lens; M, mirror; obj, objective; S, sample; F, filter; TL, tube lens.
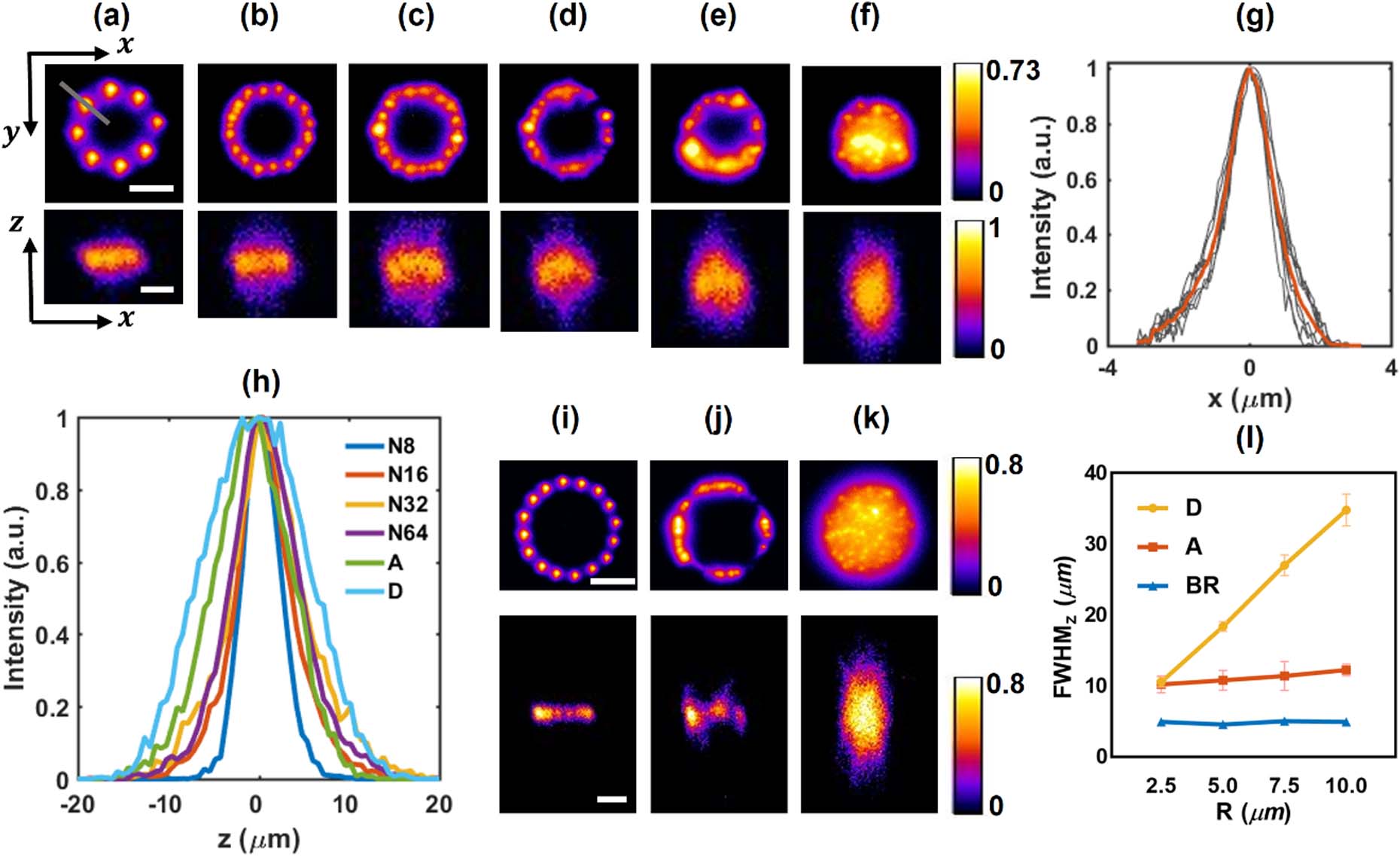
Fig. 2. Characterization of generated holographic patterns for a single target stimulation. (a)–(f) Two-photon intensity distributions in x y x z x y x z
Fig. 3. Characterization of generated holographic patterns for multiple target stimulation in a plane. (a)–(f) Two-photon intensity distributions in x y x z x y − 50 x z − 100 x y x z
Fig. 4. Characterization of generated holographic patterns for multiple target stimulation in 3D stack. (a)–(c) Intensity distributions (upper) and maximum intensity projections in x y 120 μm × 120 μm × 240 μm
Fig. 5. All-optical physiology test with different stimulation patterns on a single neuron in vivo . (a) Schematic diagram of the all-optical physiology system. Green: beam for two-photon calcium imaging. Red: beam for two-photon optogenetics. Diagram at lower right: excitation pattern distribution tested by the all-optical physiology system with 1 μm fluorescent beads. D, disk pattern; BR, beaded-ring pattern. Scale bar: 5 μm. (b) Two-photon image of a neuron in L2/3 of mouse S1 cortex, infected with a ChRmine/GCaMP6m virus. Scale bar: 10 μm. (c) Calcium signal of a neuron under different stimulation patterns at the optimal stimulation depth. (d) Changes in calcium signal caused by two-photon optogenetics at different stimulation depths under different stimulation patterns. The fitted response intensity curves are shown with solid lines. (e) Statistical analysis of axial resolutions under different stimulation schemes. Data are from 10 neurons in three mice. * * p = 0.0017 t
Fig. 6. All-optical physiology test with different stimulation patterns on multiple neurons simultaneously in vivo . (a) Left: typical two-photon image of neurons in L2/3 of mouse S1 cortex, infected with a ChRmine/GCaMP6m virus. Locations of gray spots are the center of two excitation patterns. Right: distribution of excitation patterns to stimulate target neurons. D, disk pattern; BR, beaded-ring pattern. Scale bar: 10 μm. (b) Mean axial resolutions of calcium signals of multiple neurons under different stimulation schemes. Data are from eight groups, and each group has two neurons to be stimulated. * * p = 0.0086 t p = 0.0638 t
Fig. 7. Calcium signal of neurons with different types of stimulation patterns, at different stimulation powers. BR, beaded-ring pattern; D, disk pattern; ns, not significant. p = 0.30 t

Set citation alerts for the article
Please enter your email address



