- Spectroscopy and Spectral Analysis
- Vol. 41, Issue 9, 2975 (2021)
Abstract
Keywords
Introduction
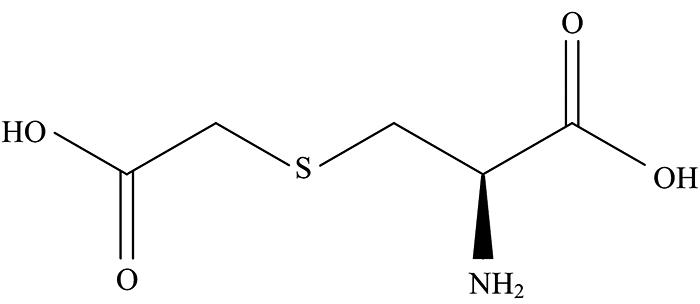
Carbocysteine or carbocisteine having the empirical formula C5H9NO4S, (Fig.1) is one of the most therapeutically prescribed expectorants, sold under the brand name viz., Mucodyne, Rhinathiol and Mucolite[
A new silver-carbocysteine (Ccy-Ag) complex [Ag2(Ccy)2(H2O)2] has been synthesized and characterized by using a combination of FTIR, Raman, molar conductivity, 1HNMR, electronic spectra, thermal analyses, X-ray powder diffraction (XRD) and scanning electron microscopy (SEM)[
![]()
Figure 1.Structure of carbocysteine drug
Carboxylates are a very important class of ligand in bioinorganic chemistry. This can be accredited to the versatility of the RCOO-ligand and the wide range of coordination modes that it can adopt. Many carboxylate complexes have been characterized and the coordination chemistry of carboxylic acids is well documented [
Carbocysteine is an antioxidant and mucolytic agent, is effective in reducing the severity and the rate of exacerbations in COPD patients[
1 Experimental
1.1 Synthesis of Sr(Ⅱ), Ba(Ⅱ) and Pb(Ⅱ) carbocysteine complexes
All chemicals used (strontium(Ⅱ) chloride hexahydrate, barium(Ⅱ) chloride dihydrate and lead(Ⅱ) acetate trihydrate) were of the purest laboratory grade (Aldrich Chemical Company) and the carbocysteine drug was received from Egyptian International Pharmaceutical Industrial Company (EIPICo.). Doubly distilled water and methanol were employed as solvents. All used reagents were of analytical grade and used without further purifications. Strontium(Ⅱ) chloride hexahydrate, barium(Ⅱ) chloride dihydrate and lead(Ⅱ) acetate trihydrate (1 mmol) were dissolved in 20 mL of water/methanol (50/50 w/w) and then the prepared solutions were slowly added to (1 mmol, 30 mL) of water/methanol (50/50 w/w) with of HCcy ligand solution under magnetic stirring. The pH of solution adjusted to 7~8 by addition of few drops of 10% ammonium hydroxide solution. The resulting solutions heated at 70 ℃ for 2 hrs and left to evaporate slowly at room temperature overnight. The obtained precipitates were filtered-off, wash with hot water and then dried at 80 ℃ and stored under vacuum over anhydrous CaCl2.
1.2 Instruments
The C, H and N percentage determined using Vario EL Fab. CHNS. Metal content and water percentage were determined by gravimetrically technique. IR data for Sr(Ⅱ), Ba(Ⅱ) and Pb(Ⅱ) carbocysteine complexes were measured using infrared Bruker spectrophotometer ranged between 400~4 000 cm-1. The conductance measurements with concentration of 10-3 mol·L-1 for complexity in dimethyl sulfoxide solvent measured using HACH conductivity meter model. 1H-NMR was recorded as dimethyl sulfoxide solutions on a Bruker 600 MHz spectrometer using tetramethyl silane as the internal standard. The electronic absorption spectra were recorded in DMSO solvent within 900~200 nm range using a UV2 Unicam UV/Vis Spectrophotometer fitted with a quartz cell of 1.0 cm path length. The X-ray diffraction patterns were recorded on X 'Pert PRO PAN analytical X-ray powder diffraction, target copper with secondary monochromate.
2 Results and Discussion
2.1 Microanalytical and Molar conductance data
The elemental analysis results of solid white colour of non-transition Sr(Ⅱ), Ba(Ⅱ) and Pb(Ⅱ) carbocysteine complexes are summarized in Table 1. The melting points of the divalent metal(Ⅱ) Ccy complexes are higher than that of the free HCcy drug ligand, revealing that the chelations are much more stable than ligand. The molar conductance values of the Ccy-M(Ⅱ) complexes found to be 20~30 Ω-1·cm2·mol-1 at 25 ℃, which indicate that all the complexes have a non-electrolytic nature. The low conductivity values agree with the absence of anions inside or outside coordination sphere[
2.2 Electronic spectra
The formation of the Sr(Ⅱ), Ba(Ⅱ) and Pb(Ⅱ) carbocysteine complexes was confirmed using electronic UV-Vis absorption spectra. It can see that the electronic spectrum of free carbocysteine drug has a characteristic band observed at 290~310 nm assigned to n→π* electronic transition. In case of the spectra of the synthesized divalent carbocysteine complexes, the band are blue shifted to lower wave length within 260~280 nm, this result clearly indicate that the Ccy ligand coordinate to central metal ions via the deprotonated carboxylic groups which are in accordance with the results of the FTIR assignments.
2.3 Infrared spectra
The position of the strong ν(C=O) stretching vibration band in case of the carboxylic acid of the free HCcy drug ligand exhibit at ~1 760 cm-1. Upon complexation to the central metal ions, this ν(C=O) band is absent and the two new bands regarding the asymmetric (νasym(OCO)) and symmetric (νsym(OCO)) stretches appear in the regions of 1 565~1 540 and 1 415~1 390 cm-1, respectively[
![]()
Figure 2.FTIR spectrum of Pb(Ⅱ) Ccy complex
![]()
Figure 3.Speculated structures of Ba(Ⅱ), Sr(Ⅱ) and Pb(Ⅱ) Ccy complexes
![]()
Figure 4.XRD spectrum of Pb(Ⅱ) Ccy complex
Table Infomation Is Not EnableImage Infomation Is Not Enable2.4 1H-NMR spectra
The 1H-NMR spectrum of Ba(Ⅱ) Ccy complex was carried out in DMSO-d6 as a suitable solvent. Upon comparison with the Ccy free ligand, the signal observed at 11 ppm can assigned to the proton of carboxylate group. This signal disappears in the spectrum of the Ccy-Ba(Ⅱ) complex, which confirms the coordination of carbocysteine ligand to the barium(Ⅱ) ion through the deprotonated carboxylic group. On the other hand, the signals of NH2 group which observed at 6.9~7.25 ppm did not affected, which mean that it does not contributed in coordination mode. The band present at 2.50 ppm assigned to the coordinated water molecules, which participate in coordination mode. The results clearly indicate that the ligand coordinate to metal ions via carboxylic (Fig.3) which is in accordance with the results of the FTIR and 1HNMR spectra.
2.5 X-ray powder diffraction patterns spectra
X-ray analysis of Ccy complexes X-ray analysis for Pb(Ⅱ)-Ccy complex was carried out by using X 'Pert PRO PANanalytical diffractometer type, tube anode cupper, and PC-APD, diffraction software indicates that Pb(Ⅱ)-Ccy complex formed crystalline structure (Fig.4), but Ba(Ⅱ)-Ccy and Sr(Ⅱ)-Ccy complexes formed amorphous structure.
References
[1] E Bichona, P Sitthisacka, L Sérée-Vattiera et al. Food Chemistry, 260, 283(2018).
[2] K Rajeshwaran, M Manivannan, R Govindhan et al. Journal of Molecular Structure, 1144, 432(2017).
[3] Z Hamza R, M El-Megharbel S, S Refat M. Chemico-Biological Interactions, 220, 169(2014).
[4] S Refat M. J. Mol. Struct., 842, 24(2007).
[5] M Hannon. J. Pure Appl. Chem., 79, 2243(2007).
[6] R C Mehrotra. Metal Carboxylates; Academic Press Inc.: London(1983).
[7] S J Lippard, L Rardin R, B Tolman W. New J. Chem., 15, 417(1991).
[8] P Zheng J, J Kang, G Huang S et al. Lancet, 371, 2013(2008).
[9] W MacNee, I Rahman. Curr. Opin. Pharmacol., 12, 256(2012).
[10] R Nakon, J Angelici R, M Beadle E. J. Am. Chem. Soc., 96, 719(1974).
[11] M Venturini, M Filella, V Brumas et al. J. Inorg. Biochem., 37, 309(1989).
[12] M Claude, M Cromer-Morin, P Scharff J et al. Anal. Chim. Acta, 104, 299(1979).
[13] B Deacon G, R J Phillips. Coord. Chem. Rev, 33, 227(1980).
[14] K Nakamoto. Infrared and Raman Spectra of Inorganic and Coordination Compounds. Wiely, New York(1978).
Set citation alerts for the article
Please enter your email address



