Bo Gu. High temperature magnetic semiconductors: narrow band gaps and two-dimensional systems[J]. Journal of Semiconductors, 2019, 40(8): 081504
Search by keywords or author
- Journal of Semiconductors
- Vol. 40, Issue 8, 081504 (2019)
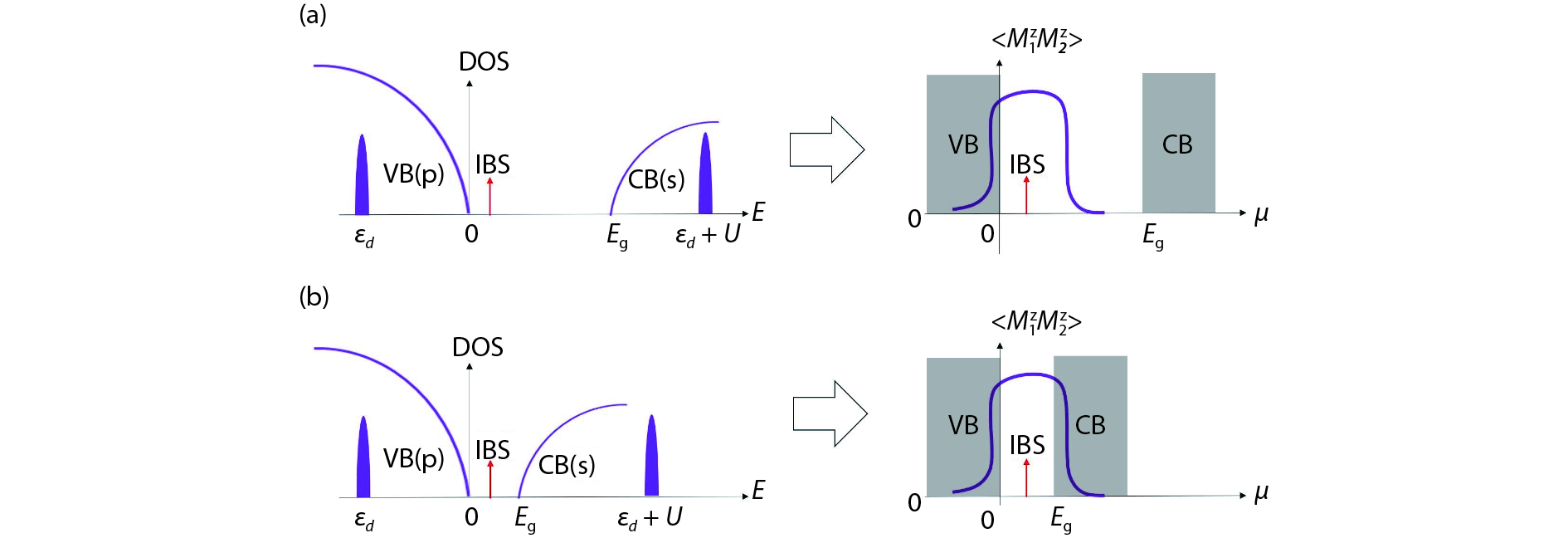
![(Color online) Schematic pictures of magnetic semiconductors with (a) wide band gaps and (b) narrow band gaps. The band gap is . The top of valence band (VB) is dominated by p orbitals, and the bottom of conduction band (CB) is dominated by s orbitals. For the impurity with d orbitals, is impurity level of d orbitals, and is the on-site Coulomb interaction. Impurity bound state (IBS) is also developed due to the doping of impurity into the host. The density of state (DOS) as a function of energy, and the magnetic correlation between two impurities as a function of the chemical potential are depicted. (a) Due to strong mixing between the impurity and the VB, the position of the IBS (arrow) is close to the top of the VB. Due to weak mixing between the impurity and the CB, usually no IBS appears below the bottom of the CB[19–21]. Thus, we have 0 for the wide band gap case. By the condition , positive (FM coupling) can develop[22–24]. For p-type carriers (), ferromagnetic coupling can be obtained as the condition can be satisfied. For n-type carriers (), no magnetic coupling is obtained between impurities because the condition cannot be satisfied[19–21]. (b) Case for narrow band gap . By choosing suitable host semiconductors and impurities, the condition 0 can be obtained. For both p-type and n-type carriers, ferromagnetic coupling can be obtained because the condition is satisfied.](/richHtml/jos/2019/40/8/081504/img_1.jpg)
Fig. 1. (Color online) Schematic pictures of magnetic semiconductors with (a) wide band gaps and (b) narrow band gaps. The band gap is
d orbitals, and
19 –21 ]. Thus, we have 0
22 –24 ]. For p-type carriers (
19 –21 ]. (b) Case for narrow band gap
![(Color online) Band structure of the ZnO host with wurtzite, zincblende, and rocksalt crystal structures. Adapted from Ref. [19].](/richHtml/jos/2019/40/8/081504/img_2.jpg)
Fig. 2. (Color online) Band structure of the ZnO host with wurtzite, zincblende, and rocksalt crystal structures. Adapted from Ref. [19 ].
Fig. 3. (Color online) For Mn impurity in ZnO, hybridization parameter
19 ].
Fig. 4. (Color online) For Mn impurity in ZnO, square of the magnetic moment at the impurity site
19 ].
Fig. 5. (Color online) For Mn impurity in ZnO, impurity-impurity magnetic correlation function
19 ].
Fig. 6. (Color online) For N impurity in MgO, host band and hybridization. (a) MgO bands structure, where an direct band gap of 7.5 eV was obtained. Hybridization between 2
21 ].
Fig. 7. (Color online) For N impurity in MgO, square of magnetic moment
21 ].
Fig. 8. (Color online) For N impurity in MgO, impurity-impurity magnetic correlation
21 ].
Fig. 9. (Color online) For Mn impurity in BaZn2As2, host band and impurity-host hybridization. (a) Energy bands off host BaZn2As2. Band gap of 0.2 eV was obtained by DFT calculations, consistent with experiment[11 ]. The hybridization parameter between the 3d orbitals of Mn and (b) valence bands and (c) conduction bands of BaZn2As2. Adapted from Ref. [17 ].
Fig. 10. (Color online) For Mn impurity in BaZn
17 ].
Fig. 11. (Color online) For Mn impurities in BaZn
17 ].
Fig. 12. (Color online) Cr impurity versus Mn impurity in host BaZn
18 ].
Fig. 13. (Color online) For Cr impurity in BaZn
18 ].
Fig. 14. (Color online) Crystal structure of two-dimensional Cr
Fig. 15. (Color online) Electron band structure of two-dimensional Cr
39 ].
Fig. 16. (Color online) For two-dimensional Cr
39 ].
Fig. 17. (Color online) Crystal structure of two-dimensional PtBr
Fig. 18. (Color online) The band structure of two-dimensional PdBr
C of the nontrivial band near Fermi energy
40 ].
Fig. 19. (Color online) For two-dimensional PtBr
40 ].
Set citation alerts for the article
Please enter your email address



