
A hybrid bright/darkfield transport of intensity (HBDTI) approach for high-throughput quantitative phase microscopy significantly expands the space-bandwidth-product of a conventional microscope, extending the accessible sample spatial frequencies in the Fourier space well beyond the traditional coherent diffraction limit. Image credit: Linpeng Lu, NJUST.
Cell organelles are involved in a variety of cellular life activities. Their dysfunction is closely related to the development and metastasis of cancer. Exploration of subcellular structures and their abnormal states facilitates insights into the mechanisms of pathologies, which may enable early diagnosis for more effective treatment.
The optical microscope, invented more than 400 years ago, has become an indispensable and ubiquitous instrument for the investigation of microscale objects in many areas of science and technology. In particular, fluorescence microscopy has achieved several leaps – from 2D wide-field, to 3D confocal, and then to super-resolution fluorescence microscopy, greatly promoting the development of modern life sciences.
Using conventional microscopes, researchers currently struggle to generate sufficient intrinsic contrast for unstained cells, due to their low absorption or weak scattering properties. Specific dyes or fluorescent labels can help with visualization, but long-term observation of live cells remains difficult to achieve.
Recently, quantitative phase imaging (QPI) has shown promise with its unique ability to quantify the phase delay of unlabeled specimens in a nondestructive way. Yet the throughput of an imaging platform is fundamentally limited by its optical system’s space-bandwidth product (SBP), and the SBP increase of a microscope is fundamentally confounded by the scale-dependent geometric aberrations of its optical elements. This results in a tradeoff between achievable image resolution and field of view (FOV).
An approach to achieving label-free, high-resolution, and large FOV microscopic imaging is needed to enable precise detection and quantitative analysis of subcellular features and events. To this end, researchers from Nanjing University of Science and Technology (NJUST) and the University of Hong Kong recently developed a label-free high-throughput microscopy method based on hybrid bright/darkfield illuminations. As reported in Advanced Photonics, the “hybrid brightfield-darkfield transport of intensity” (HBDTI) approach for high-throughput quantitative phase microscopy significantly expands the accessible sample spatial frequencies in the Fourier space, extending the maximum achievable resolution by approximately fivefold over the coherent imaging diffraction limit.
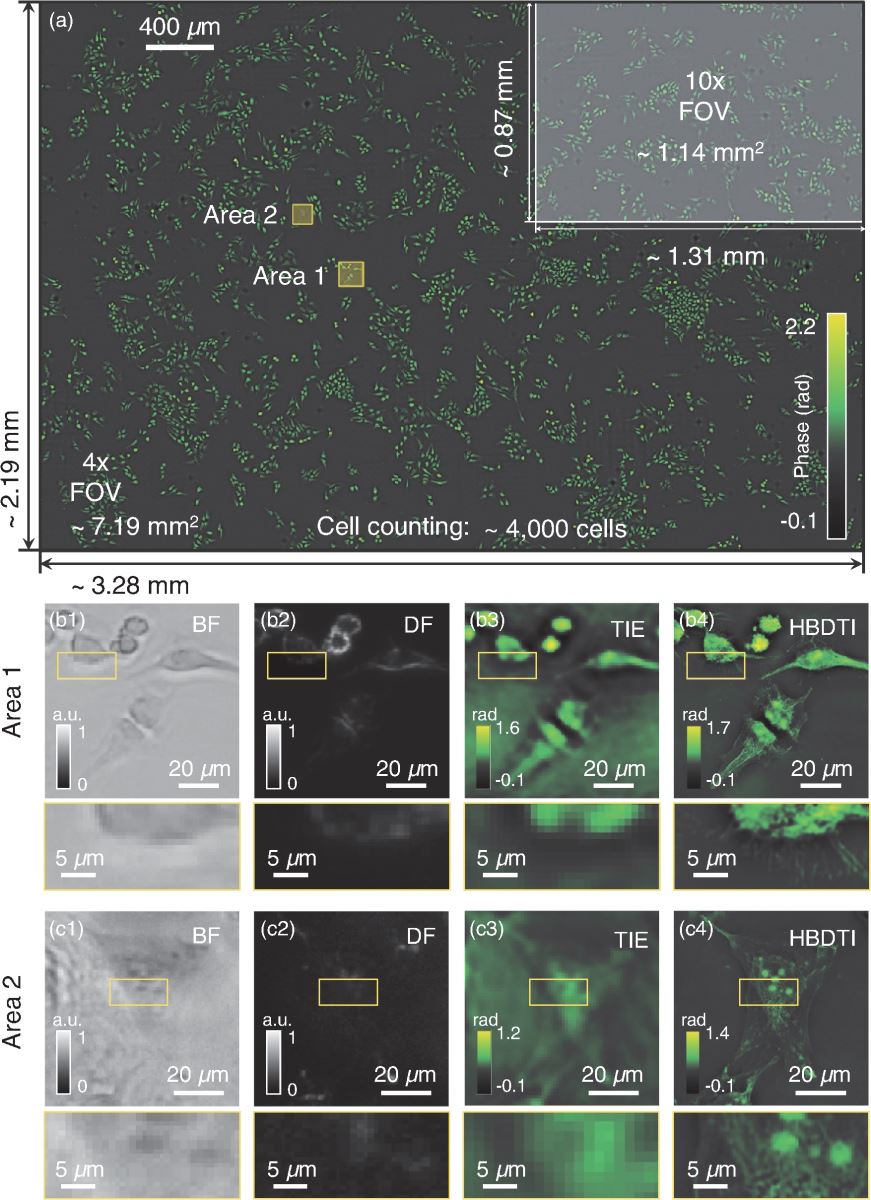
QPI results of unlabeled HeLa cells. (a) Approximately 4000 HeLa cells on a ∼7.19 mm2 FOV. (b1) and (c1) Low-resolution brightfield (BF) in-focus intensity images of areas 1 and 2 in (a), respectively. (b2) and (c2) Low-resolution darkfield (DF) in-focus intensity images of (b1) and (c1), respectively. (b3) and (c3) Retrieval phase results of (b1) and (c1) using the FFT-based traditional transport of intensity equation (TIE) phase retrieval method, respectively. (b4) and (c4) Retrieval phase results of (b1) and (c1) utilizing the novel HBDTI method, respectively. Image credit: Lu et al., doi 10.1117/1.AP.4.5.056002.
Based on the principle of illumination multiplexing and synthetic aperture, they establish a forward imaging model of nonlinear brightfield and darkfield intensity transport. This model endows HBDTI with the ability to provide features beyond the coherent diffraction limit. Using a commercial microscope with a 4x, 0.16NA objective lens, the team demonstrated HBDTI high-throughput imaging, attaining 488-nm half-width imaging resolution within an FOV of approximately 7.19 mm2, yielding a 25× increase in SBP over the case of coherent illumination.
Noninvasive high-throughput imaging enables delineation of subcellular structures in large-scale cell studies. According to corresponding author Chao Zuo, principal investigator of the Smart Computational Imaging Laboratory (SCILab) at NJUST, “HBDTI offers a simple, high-performance, low-cost, and universal imaging tool for quantitative analysis in life sciences and biomedical research. Given its capability for high-throughput QPI, HBDTI is expected to provide a powerful solution for cross-scale detection and analysis of subcellular structures in a large number of cell clusters.” Zuo notes that further efforts are needed to promote the high-speed implementation of HBDTI in large-group live cell analysis.
Lead author Linpeng Lu, a PhD student in the SCILab, provides a vivid hand-painted animation as a helpful summary of the report:
Read the Gold Open Access article by Lu et al., “Hybrid brightfield and darkfield transport of intensity approach for high-throughput quantitative phase microscopy,” Adv. Photon. 4(5), 056002 (2022), doi 10.1117/1.AP.4.5.056002.


